Abstract
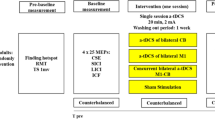
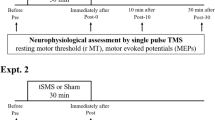
This study investigates the effect of corticospinal excitability during sham stimulation on the individual response to transcranial non-invasive brain stimulation (tNIBS). Thirty healthy young adults aged 24.2 ± 2.8 S.D. participated in the study. Sham, as well as 1 mA of tRNS and 140 Hz tACS stimulation were applied for 10 min each at different sessions. The effect of each stimulation type was quantified by recording TMS-induced, motor evoked potentials (MEPs) before (baseline) and at fixed time points after stimulation (T0, T30, T60 min.). According to the individual response to sham stimulation at T0 in comparison to baseline MEPs, subjects were regarded as responder or non-responder to sham. Following, MEPs at T0, T30 and T60 after verum or sham stimulation were assessed with a repeated measures ANOVA with the within-subject factor stimulation (sham, tRNS, 140 Hz tACS) and the between-subjects factor group (responder vs non-responder). We found that individuals who did not show immediately changes in excitability in sham stimulation sessions were the ones who responded to active stimulation conditions. On the other hand, individuals who responded to sham condition, by either increases or decreases in MEPS, did not respond to active verum stimulation. This result suggests that the presence or lack of responses to sham stimulation can provide a marker for how individuals will respond to tRNS/tACS and thus provide an explanation for the variability in interindividual response. The results of this study draw attention to the general reactivity of the brain, which can be taken into account when planning future studies using tNIBS.



Similar content being viewed by others
References
Ambrus GG, Pisoni A, Primassin A, Turi Z, Paulus W, Antal A (2015) Bi-frontal transcranial alternating current stimulation in the ripple range reduced overnight forgetting. Front Cell Neurosci 9:374. https://doi.org/10.3389/fncel.2015.00374
Ammann C, Lindquist MA, Celnik PA (2017) Response variability of different anodal transcranial direct current stimulation intensities across multiple sessions. Brain Stimul 10:757–763. https://doi.org/10.1016/j.brs.2017.04.003
Antal A, Herrmann CS (2016) Transcranial alternating current and random noise stimulation: possible mechanisms. Neural Plast 2016:3616807. https://doi.org/10.1155/2016/3616807
Antal A, Terney D, Poreisz C, Paulus W (2007) Towards unravelling task-related modulations of neuroplastic changes induced in the human motor cortex. Eur J Neurosci 26:2687–2691. https://doi.org/10.1111/j.1460-9568.2007.05896.x
Antal A, Chaieb L, Moliadze V et al (2010) Brain-derived neurotrophic factor (BDNF) gene polymorphisms shape cortical plasticity in humans. Brain Stimul 3:230–237. https://doi.org/10.1016/j.brs.2009.12.003
Batsikadze G, Moliadze V, Paulus W, Kuo MF, Nitsche M (2013) Partially non-linear stimulation intensity-dependent effects of direct current stimulation on motor cortex excitability in humans. J Physiol 591:1987–2000
Bienenstock EL, Cooper LN, Munro PW (1982) Theory for the development of neuron selectivity: orientation specificity and binocular interaction in visual cortex. J Neurosci 2:32–48
Bocci T, Caleo M, Tognazzi S et al (2014) Evidence for metaplasticity in the human visual cortex. J Neural Transm (Vienna) 121:221–231. https://doi.org/10.1007/s00702-013-1104-z
Brauer H, Kadish NE, Pedersen A, Siniatchkin M, Moliadze V (2018) No modulatory effects when stimulating the right inferior frontal gyrus with continuous 6 Hz tACS and tRNS on response inhibition: a behavioral study. Neural Plast 2018:3156796. https://doi.org/10.1155/2018/3156796
Brignani D, Ruzzoli M, Mauri P, Miniussi C (2013) Is transcranial alternating current stimulation effective in modulating brain oscillations? PLoS One 8:e56589. https://doi.org/10.1371/journal.pone.0056589
Cabral-Calderin Y, Williams KA, Opitz A, Dechent P, Wilke M (2016) Transcranial alternating current stimulation modulates spontaneous low frequency fluctuations as measured with fMRI. Neuroimage 141:88–107. https://doi.org/10.1016/j.neuroimage.2016.07.005
Chew T, Ho KA, Loo CK (2015) Inter- and intra-individual variability in response to transcranial direct current stimulation (tDCS) at varying current intensities. Brain Stimul 8:1130–1137. https://doi.org/10.1016/j.brs.2015.07.031
Dissanayaka TD, Zoghi M, Farrell M, Egan GF, Jaberzadeh S (2018) Sham transcranial electrical stimulation and its effects on corticospinal excitability: a systematic review and meta-analysis. Rev Neurosci 29:223–232. https://doi.org/10.1515/revneuro-2017-0026
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G* Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Fertonani A, Miniussi C (2016) Transcranial electrical stimulation: what we know and do not know about mechanisms. Neuroscientist. https://doi.org/10.1177/1073858416631966
Fertonani A, Pirulli C, Miniussi C (2011) Random noise stimulation improves neuroplasticity in perceptual learning. J Neurosci 31:15416–15423. https://doi.org/10.1523/jneurosci.2002-11.2011
Feurra M, Pasqualetti P, Bianco G, Santarnecchi E, Rossi A, Rossi S (2013) State-dependent effects of transcranial oscillatory currents on the motor system: what you think matters. J Neurosci 33:17483–17489. https://doi.org/10.1523/jneurosci.1414-13.2013
Fierro B, Brighina F, Vitello G et al (2005) Modulatory effects of low- and high-frequency repetitive transcranial magnetic stimulation on visual cortex of healthy subjects undergoing light deprivation. J Physiol 565:659–665. https://doi.org/10.1113/jphysiol.2004.080184
Fonteneau C, Mondino M, Arns M et al (2019) Sham tDCS: a hidden source of variability? Reflections for further blinded, controlled trials. Brain Stimul 12:668–673. https://doi.org/10.1016/j.brs.2018.12.977
Furubayashi T, Terao Y, Arai N et al (2008) Short and long duration transcranial direct current stimulation (tDCS) over the human hand motor area. Exp Brain Res 185:279–286. https://doi.org/10.1007/s00221-007-1149-z
Guerra A, Lopez-Alonso V, Cheeran B, Suppa A (2017) Variability in non-invasive brain stimulation studies: reasons and results. Neurosci Lett. https://doi.org/10.1016/j.neulet.2017.12.058
Hamada M, Murase N, Hasan A, Balaratnam M, Rothwell JC (2013) The role of interneuron networks in driving human motor cortical plasticity. Cereb Cortex 23:1593–1605. https://doi.org/10.1093/cercor/bhs147
Hordacre B, Goldsworthy MR, Vallence AM et al (2016) Variability in neural excitability and plasticity induction in the human cortex: a brain stimulation study. Brain Stimul. https://doi.org/10.1016/j.brs.2016.12.001
Hordacre B, Moezzi B, Ridding MC (2018) Neuroplasticity and network connectivity of the motor cortex following stroke: a transcranial direct current stimulation study. Hum Brain Mapp 39:3326–3339. https://doi.org/10.1002/hbm.24079
Horvath JC, Carter O, Forte JD (2014) Transcranial direct current stimulation: five important issues we aren’t discussing (but probably should be). Front Syst Neurosci 8:2
Horvath JC, Forte JD, Carter O (2015) Evidence that transcranial direct current stimulation (tDCS) generates little-to-no reliable neurophysiologic effect beyond MEP amplitude modulation in healthy human subjects: a systematic review. Neuropsychologia 66:213–236
Horvath JC, Vogrin SJ, Carter O, Cook MJ, Forte JD (2016) Effects of a common transcranial direct current stimulation (tDCS) protocol on motor evoked potentials found to be highly variable within individuals over 9 testing sessions. Exp Brain Res 234(9):2629–2642. https://doi.org/10.1007/s00221-016-4667-8
Javadi AH, Cheng P, Walsh V (2012) Short duration transcranial direct current stimulation (tDCS) modulates verbal memory. Brain Stimul 5:468–474. https://doi.org/10.1016/j.brs.2011.08.003
Kanai R, Paulus W, Walsh V (2010) Transcranial alternating current stimulation (tACS) modulates cortical excitability as assessed by TMS-induced phosphene thresholds. Clin Neurophysiol 121:1551–1554. https://doi.org/10.1016/j.clinph.2010.03.022
Karabanov A, Ziemann U, Hamada M et al (2015) Consensus paper: probing homeostatic plasticity of human cortex with non-invasive transcranial brain stimulation. Brain Stimul 8:442–454. https://doi.org/10.1016/j.brs.2015.01.404
Krause B, Cohen Kadosh R (2014) Not all brains are created equal: the relevance of individual differences in responsiveness to transcranial electrical stimulation. Front Syst Neurosci 8:25
Labruna L, Jamil A, Fresnoza S et al (2016) Efficacy of anodal transcranial direct current stimulation is related to sensitivity to transcranial magnetic stimulation. Brain Stimul 9:8–15. https://doi.org/10.1016/j.brs.2015.08.014
Lang N, Nitsche MA, Paulus W, Rothwell JC, Lemon RN (2004) Effects of transcranial direct current stimulation over the human motor cortex on corticospinal and transcallosal excitability. Exp Brain Res 156:439–443. https://doi.org/10.1007/s00221-003-1800-2
Li LM, Uehara K, Hanakawa T (2015) The contribution of interindividual factors to variability of response in transcranial direct current stimulation studies. Front Cell Neurosci. https://doi.org/10.3389/fncel.2015.00181
López-Alonso V, Cheeran B, Río-Rodríguez D, Fernández-del-Olmo M (2014) Inter-individual variability in response to non-invasive brain stimulation paradigms. Brain Stimul 7:372–380
Mehta AR, Pogosyan A, Brown P, Brittain JS (2015) Montage matters: the influence of transcranial alternating current stimulation on human physiological tremor. Brain Stimul 8:260–268. https://doi.org/10.1016/j.brs.2014.11.003
Mei F, Nagappan G, Ke Y, Sacktor TC, Lu B (2011) BDNF facilitates L-LTP maintenance in the absence of protein synthesis through PKMzeta. PLoS One 6:e21568. https://doi.org/10.1371/journal.pone.0021568
Miniussi C, Harris JA, Ruzzoli M (2013) Modelling non-invasive brain stimulation in cognitive neuroscience. Neurosci Biobehav Rev 37:1702–1712. https://doi.org/10.1016/j.neubiorev.2013.06.014
Moliadze V, Antal A, Paulus W (2010a) Boosting brain excitability by transcranial high frequency stimulation in the ripple range. J Physiol 588:4891–4904. https://doi.org/10.1113/jphysiol.2010.196998 (jphysiol.2010.196998 [pii])
Moliadze V, Antal A, Paulus W (2010b) Electrode-distance dependent after-effects of transcranial direct and random noise stimulation with extracephalic reference electrodes. Clin Neurophysiol 121:2165–2171. https://doi.org/10.1016/j.clinph.2010.04.033 (S1388-2457(10)00481-5 [pii])
Moliadze V, Atalay D, Antal A, Paulus W (2012) Close to threshold transcranial electrical stimulation preferentially activates inhibitory networks before switching to excitation with higher intensities. Brain Stimul 5:505–511. https://doi.org/10.1016/j.brs.2011.11.004 (S1935-861X(11)00167-7 [pii])
Moliadze V, Schmanke T, Andreas S, Lyzhko E, Freitag CM, Siniatchkin M (2015) Stimulation intensities of transcranial direct current stimulation have to be adjusted in children and adolescents. Clin Neurophysiol 126:1392–1399. https://doi.org/10.1016/j.clinph.2014.10.142
Moliadze V, Lyzhko E, Schmanke T, Andreas S, Freitag CM, Siniatchkin M (2018) 1 mA cathodal tDCS shows excitatory effects in children and adolescents: insights from TMS evoked N100 potential. Brain Res Bull 140:43–51. https://doi.org/10.1016/j.brainresbull.2018.03.018
Moliadze V, Sierau L, Lyzhko E, Stenner T, Werchowski M, Siniatchkin M, Hartwigsen G (2019) After-effects of 10 Hz tACS over the prefrontal cortex on phonological word decisions. Brain Stimul. https://doi.org/10.1016/j.brs.2019.06.021
Muller-Dahlhaus F, Ziemann U (2015) Metaplasticity in human cortex. Neuroscientist 21:185–202. https://doi.org/10.1177/1073858414526645
Nguyen J, Deng Y, Reinhart RMG (2018) Brain-state determines learning improvements after transcranial alternating-current stimulation to frontal cortex. Brain Stimul. https://doi.org/10.1016/j.brs.2018.02.008
Nitsche MA, Paulus W (2000) Excitability changes induced in the human motor cortex by weak transcranial direct current stimulation. J Physiol 527(Pt 3):633–639. doi: PHY_1055 [pii]
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Paulus W (2011) Transcranial electrical stimulation (tES–tDCS; tRNS, tACS) methods. Neuropsychological rehabilitation 21:602–617
Paulus W, Nitsche MA, Antal A (2016) Application of transcranial electric stimulation (tDCS, tACS, tRNS). Eur Psychol 21(1):4–14
Pirulli C, Fertonani A, Miniussi C (2013) The role of timing in the induction of neuromodulation in perceptual learning by transcranial electric stimulation. Brain Stimul 6:683–689. https://doi.org/10.1016/j.brs.2012.12.005
Poreisz C, Boros K, Antal A, Paulus W (2007) Safety aspects of transcranial direct current stimulation concerning healthy subjects and patients. Brain Res Bull 72:208–214
Ridding M, Ziemann U (2010) Determinants of the induction of cortical plasticity by non-invasive brain stimulation in healthy subjects. J Physiol 588:2291–2304
Ruzzoli M, Marzi CA, Miniussi C (2010) The neural mechanisms of the effects of transcranial magnetic stimulation on perception. J Neurophysiol 103:2982–2989. https://doi.org/10.1152/jn.01096.2009
Scheldrup M, Greenwood PM, McKendrick R et al (2014) Transcranial direct current stimulation facilitates cognitive multi-task performance differentially depending on anode location and subtask. Front Hum Neurosci 8:665. https://doi.org/10.3389/fnhum.2014.00665
Schinder AF, Poo M (2000) The neurotrophin hypothesis for synaptic plasticity. Trends Neurosci 23:639–645
Sehm B, Kipping J, Schafer A, Villringer A, Ragert P (2013) A comparison between uni- and bilateral tDCS effects on functional connectivity of the human motor cortex. Front Hum Neurosci 7:183. https://doi.org/10.3389/fnhum.2013.00183
Siebner HR, Lang N, Rizzo V, Nitsche MA, Paulus W, Lemon RN, Rothwell JC (2004) Preconditioning of low-frequency repetitive transcranial magnetic stimulation with transcranial direct current stimulation: evidence for homeostatic plasticity in the human motor cortex. J Neurosci 24:3379–3385. https://doi.org/10.1523/jneurosci.5316-03.2004
Silvanto J, Muggleton N, Walsh V (2008) State-dependency in brain stimulation studies of perception and cognition. Trends Cogn Sci 12:447–454. https://doi.org/10.1016/j.tics.2008.09.004
Stagg CJ, Nitsche MA (2011) Physiological basis of transcranial direct current stimulation. Neuroscientist 17:37–53. https://doi.org/10.1177/1073858410386614
Teo F, Hoy KE, Daskalakis ZJ, Fitzgerald PB (2011) Investigating the role of current strength in tDCS modulation of working memory performance in healthy controls. Front Psychiatry 2:45. https://doi.org/10.3389/fpsyt.2011.00045
Terney D, Chaieb L, Moliadze V, Antal A, Paulus W (2008) Increasing human brain excitability by transcranial high-frequency random noise stimulation. J Neurosci 28:14147–14155. https://doi.org/10.1523/jneurosci.4248-08.2008 (28/52/14147 [pii])
Tremblay S, Larochelle-Brunet F, Lafleur LP, El Mouderrib S, Lepage JF, Theoret H (2016) Systematic assessment of duration and intensity of anodal transcranial direct current stimulation on primary motor cortex excitability. Eur J Neurosci 44:2184–2190. https://doi.org/10.1111/ejn.13321
Turi Z, Mittner M, Paulus W, Antal A (2017) Placebo intervention enhances reward learning in healthy individuals. Sci Rep 7:41028. https://doi.org/10.1038/srep41028
Wach C, Krause V, Moliadze V, Paulus W, Schnitzler A, Pollok B (2013) Effects of 10 Hz and 20 Hz transcranial alternating current stimulation (tACS) on motor functions and motor cortical excitability. Behav Brain Res 241:1–6. https://doi.org/10.1016/j.bbr.2012.11.038 (S0166-4328(12)00768-1 [pii])
Wiethoff S, Hamada M, Rothwell JC (2014) Variability in response to transcranial direct current stimulation of the motor cortex. Brain Stimul 7:468–475. https://doi.org/10.1016/j.brs.2014.02.003
Ziemann U, Paulus W, Nitsche MA et al (2008) Consensus: motor cortex plasticity protocols. Brain Stimul 1:164–182. https://doi.org/10.1016/j.brs.2008.06.006
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this paper. All the authors have read the manuscript and have approved this submission.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kortuem, V., Kadish, N.E., Siniatchkin, M. et al. Efficacy of tRNS and 140 Hz tACS on motor cortex excitability seemingly dependent on sensitivity to sham stimulation. Exp Brain Res 237, 2885–2895 (2019). https://doi.org/10.1007/s00221-019-05640-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-019-05640-w