Growth Factor Binding Peptides in Poly (Ethylene Glycol) Diacrylate (PEGDA)-Based Hydrogels for an Improved Healing Response of Human Dermal Fibroblasts
Abstract
:1. Introduction
2. Results
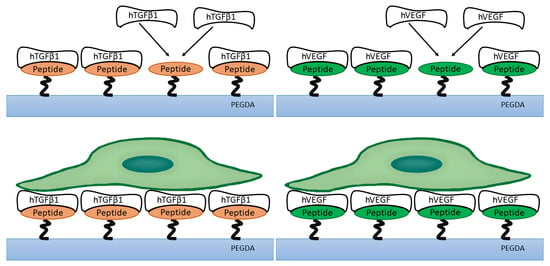
2.1. Peptide Functionalization and Incorporation into PEGDA-Based Hydrogels
2.2. Physical Characterization
2.3. Growth Factor Binding and Retention Capacity on PEGDA-Based Hydrogels
2.4. Bioactivity of Growth Factors Bound to PEGDA-Peptide Hydrogels
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Synthesis of PEGDA and Photoinitiator
5.2. Synthesis of Acrylate-Derived GF Binding Peptides and RGDS
5.3. Fabrication of PEGDA-Based Hydrogels
5.4. Mechanical and Swelling Characterization
5.5. Quantification of PEGDA-Peptide Hydrogel Binding Capacity
5.6. Human Dermal Fibroblasts Cell Culture and Adherence to Hydrogels
5.7. HDFa Proliferation, Surface Coverage, and Average Cell Area
5.8. Gene Expression Analysis
5.9. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stoica, A.E.; Chircov, C.; Grumezescu, A.M. Hydrogel dressings for the treatment of burn wounds: An up-to-date overview. Materials 2020, 13, 2853. [Google Scholar] [CrossRef]
- Aljghami, M.E.; Saboor, S.; Amini-Nik, S. Emerging innovative wound dressings. Ann. Biomed. Eng. 2019, 47, 659–675. [Google Scholar] [CrossRef]
- Monirul Islam, M.; Hemmanahalli Ramesh, V.; Durga Bhavani, P.; Goudanavar, P.S.; Naveen, N.R.; Ramesh, B.; Fattepur, S.; Narayanappa Shiroorkar, P.; Habeebuddin, M.; Meravanige, G. Optimization of process parameters for fabrication of electrospun nanofibers containing neomycin sulfate and Malva sylvestris extract for a better diabetic wound healing. Drug Deliv. 2022, 29, 3370–3383. [Google Scholar] [CrossRef]
- Alven, S.; Buyana, B.; Feketshane, Z.; Aderibigbe, B.A. Electrospun nanofibers/nanofibrous scaffolds loaded with silver nanoparticles as effective antibacterial wound dressing materials. Pharmaceutics 2021, 13, 964. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhao, X. Alginate hydrogel dressings for advanced wound management. Int. J. Biol. Macromol. 2020, 162, 1414–1428. [Google Scholar] [CrossRef] [PubMed]
- Baltzis, D.; Eleftheriadou, I.; Veves, A. Pathogenesis and treatment of impaired wound healing in diabetes mellitus: New insights. Adv. Ther. 2014, 31, 817–836. [Google Scholar] [CrossRef] [PubMed]
- Nardini, M.; Perteghella, S.; Mastracci, L.; Grillo, F.; Marrubini, G.; Bari, E.; Formica, M.; Gentili, C.; Cancedda, R.; Torre, M.L. Growth factors delivery system for skin regeneration: An advanced wound dressing. Pharmaceutics 2020, 12, 120. [Google Scholar] [CrossRef] [Green Version]
- Wang, P.; Huang, S.; Hu, Z.; Yang, W.; Lan, Y.; Zhu, J.; Hancharou, A.; Guo, R.; Tang, B. In situ formed anti-inflammatory hydrogel loading plasmid DNA encoding VEGF for burn wound healing. Acta Biomater. 2019, 100, 191–201. [Google Scholar] [CrossRef]
- Cheng, Y.; Li, Y.; Huang, S.; Yu, F.; Bei, Y.; Zhang, Y.; Tang, J.; Huang, Y.; Xiang, Q. Hybrid freeze-dried dressings composed of epidermal growth factor and recombinant human-like collagen enhance cutaneous wound healing in rats. Front. Bioeng. Biotechnol. 2020, 8, 742. [Google Scholar] [CrossRef] [PubMed]
- Andreu, V.; Mendoza, G.; Arruebo, M.; Irusta, S. Smart dressings based on nanostructured fibers containing natural origin antimicrobial, anti-inflammatory, and regenerative compounds. Materials 2015, 8, 5154–5193. [Google Scholar] [CrossRef]
- Peng, J.; Zhao, H.; Tu, C.; Xu, Z.; Ye, L.; Zhao, L.; Gu, Z.; Zhao, D.; Zhang, J.; Feng, Z. In situ hydrogel dressing loaded with heparin and basic fibroblast growth factor for accelerating wound healing in rat. Mater. Sci. Eng. C 2020, 116, 111169. [Google Scholar] [CrossRef] [PubMed]
- Nastyshyn, S.; Stetsyshyn, Y.; Raczkowska, J.; Nastishin, Y.; Melnyk, Y.; Panchenko, Y.; Budkowski, A. Temperature-responsive polymer brush coatings for advanced biomedical applications. Polymers 2022, 14, 4245. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Anseth, K.S. Controlling affinity binding with peptide-functionalized poly (ethylene glycol) hydrogels. Adv. Funct. Mater. 2009, 19, 2325–2331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, E.S. Lung extracellular matrix and fibroblast function. Ann. Am. Thorac. Soc. 2015, 12 (Suppl. S1), S30–S33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cole, M.A.; Quan, T.; Voorhees, J.J.; Fisher, G.J. Extracellular matrix regulation of fibroblast function: Redefining our perspective on skin aging. J. Cell Commun. Signal. 2018, 12, 35–43. [Google Scholar] [CrossRef] [Green Version]
- Chakroborty, D.; Sarkar, C.; Lu, K.; Bhat, M.; Dasgupta, P.S.; Basu, S. Activation of dopamine D1 receptors in dermal fibroblasts restores vascular endothelial growth factor-A production by these cells and subsequent angiogenesis in diabetic cutaneous wound tissues. Am. J. Pathol. 2016, 186, 2262–2270. [Google Scholar] [CrossRef] [Green Version]
- Shingel, K.I.; Di Stabile, L.; Marty, J.P.; Faure, M.P. Inflammatory inert poly (ethylene glycol)–protein wound dressing improves healing responses in partial-and full-thickness wounds. Int. Wound J. 2006, 3, 332–342. [Google Scholar] [CrossRef]
- Sood, A.; Granick, M.S.; Tomaselli, N.L. Wound dressings and comparative effectiveness data. Adv. Wound Care 2014, 3, 511–529. [Google Scholar] [CrossRef] [Green Version]
- Crispim, J.; Fernandes, H.; Fu, S.; Lee, Y.; Jonkheijm, P.; Saris, D.B. TGF-β1 activation in human hamstring cells through growth factor binding peptides on polycaprolactone surfaces. Acta Biomater. 2017, 53, 165–178. [Google Scholar] [CrossRef]
- Adini, A.; Adini, I.; Chi, Z.-l.; Derda, R.; Birsner, A.E.; Matthews, B.D.; D’Amato, R.J. A novel strategy to enhance angiogenesis in vivo using the small VEGF-binding peptide PR1. Angiogenesis 2017, 20, 399–408. [Google Scholar] [CrossRef]
- Salehi-Abari, M.; Koupaei, N.; Hassanzadeh-Tabrizi, S. Synthesis and characterisation of semi-interpenetrating network of polycaprolactone/polyethylene glycol diacrylate/zeolite-CuO as wound dressing. Mater. Technol. 2020, 35, 290–299. [Google Scholar] [CrossRef]
- Chen, S.-L.; Fu, R.-H.; Liao, S.-F.; Liu, S.-P.; Lin, S.-Z.; Wang, Y.-C. A PEG-based hydrogel for effective wound care management. Cell Transplant. 2018, 27, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Zhu, Z.; Wu, D.; Gan, W.; Zhu, S.; Li, W.; Tian, J.; Li, L.; Zhou, C.; Lu, L. Antibacterial poly (ethylene glycol) diacrylate/chitosan hydrogels enhance mechanical adhesiveness and promote skin regeneration. Carbohydr. Polym. 2019, 225, 115110. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Jiang, T.; Guo, R.; Li, C.; Lu, C.; Yang, G.; Nie, J.; Wang, F.; Yang, X.; Chen, Z. Injectable and degradable PEG hydrogel with antibacterial performance for promoting wound healing. ACS Appl. Bio Mater. 2021, 4, 2769–2780. [Google Scholar] [CrossRef] [PubMed]
- Koehler, J.; Wallmeyer, L.; Hedtrich, S.; Goepferich, A.M.; Brandl, F.P. pH-Modulating poly (ethylene glycol)/alginate hydrogel dressings for the treatment of chronic wounds. Macromol. Biosci. 2017, 17, 1600369. [Google Scholar] [CrossRef]
- Lu, J.; Chen, Y.; Ding, M.; Fan, X.; Hu, J.; Chen, Y.; Li, J.; Li, Z.; Liu, W. A 4arm-PEG macromolecule crosslinked chitosan hydrogels as antibacterial wound dressing. Carbohydr. Polym. 2022, 277, 118871. [Google Scholar] [CrossRef]
- Magalhães, L.S.; Andrade, D.B.; Bezerra, R.D.; Morais, A.I.; Oliveira, F.C.; Rizzo, M.S.; Silva-Filho, E.C.; Lobo, A.O. Nanocomposite hydrogel produced from PEGDA and laponite for bone regeneration. J. Funct. Biomater. 2022, 13, 53. [Google Scholar] [CrossRef]
- Ji, Y.; Yang, X.; Ji, Z.; Zhu, L.; Ma, N.; Chen, D.; Jia, X.; Tang, J.; Cao, Y. DFT-calculated IR spectrum amide I, II, and III band contributions of N-methylacetamide fine components. ACS Omega 2020, 5, 8572–8578. [Google Scholar] [CrossRef] [Green Version]
- Shameli, K.; Ahmad, M.B.; Jazayeri, S.D.; Sedaghat, S.; Shabanzadeh, P.; Jahangirian, H.; Mahdavi, M.; Abdollahi, Y. Synthesis and characterization of polyethylene glycol mediated silver nanoparticles by the green method. Int. J. Mol. Sci. 2012, 13, 6639–6650. [Google Scholar] [CrossRef] [Green Version]
- Jimenez-Vergara, A.C.; Lewis, J.; Hahn, M.S.; Munoz-Pinto, D.J. An improved correlation to predict molecular weight between crosslinks based on equilibrium degree of swelling of hydrogel networks. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018, 106, 1339–1348. [Google Scholar] [CrossRef]
- Weber, L.M.; Lopez, C.G.; Anseth, K.S. Effects of PEG hydrogel crosslinking density on protein diffusion and encapsulated islet survival and function. J. Biomed. Mater. Res. Part A Off. J. Soc. Biomater. 2009, 90, 720–729. [Google Scholar] [CrossRef] [Green Version]
- Varanda, F.; Pratas de Melo, M.J.; Caco, A.I.; Dohrn, R.; Makrydaki, F.A.; Voutsas, E.; Tassios, D.; Marrucho, I.M. Solubility of antibiotics in different solvents. 1. Hydrochloride forms of tetracycline, moxifloxacin, and ciprofloxacin. Ind. Eng. Chem. Res. 2006, 45, 6368–6374. [Google Scholar] [CrossRef]
- Munoz-Pinto, D.J.; Jimenez-Vergara, A.C.; Hou, Y.; Hayenga, H.N.; Rivas, A.; Grunlan, M.; Hahn, M.S. Osteogenic potential of poly (ethylene glycol)–poly (dimethylsiloxane) hybrid hydrogels. Tissue Eng. Part A 2012, 18, 1710–1719. [Google Scholar] [CrossRef] [Green Version]
- Martí-Carvajal, A.J.; Gluud, C.; Nicola, S.; Simancas-Racines, D.; Reveiz, L.; Oliva, P.; Cedeño-Taborda, J. Growth factors for treating diabetic foot ulcers. Cochrane Database Syst. Rev. 2015, 2015, CD008548. [Google Scholar] [CrossRef]
- El Gazaerly, H.; Elbardisey, D.M.; Eltokhy, H.M.; Teaama, D. Effect of transforming growth factor Beta 1 on wound healing in induced diabetic rats. Int. J. Health Sci. 2013, 7, 160. [Google Scholar] [CrossRef]
- Rezvani Ghomi, E.; Khalili, S.; Nouri Khorasani, S.; Esmaeely Neisiany, R.; Ramakrishna, S. Wound dressings: Current advances and future directions. J. Appl. Polym. Sci. 2019, 136, 47738. [Google Scholar] [CrossRef] [Green Version]
- Browning, M.; Wilems, T.; Hahn, M.; Cosgriff-Hernandez, E. Compositional control of poly (ethylene glycol) hydrogel modulus independent of mesh size. J. Biomed. Mater. Res. Part A 2011, 98, 268–273. [Google Scholar] [CrossRef]
- Kalra, A.; Lowe, A.; Al-Jumaily, A. Mechanical behaviour of skin: A review. J. Mater. Sci. Eng. 2016, 5, 1000254. [Google Scholar]
- Pawlaczyk, M.; Lelonkiewicz, M.; Wieczorowski, M. Age-dependent biomechanical properties of the skin. Adv. Dermatol. Allergol./Postępy Dermatol. I Alergol. 2013, 30, 302–306. [Google Scholar] [CrossRef]
- Sandford, E.; Chen, Y.; Hunter, I.; Hillebrand, G.; Jones, L. Capturing skin properties from dynamic mechanical analyses. Ski. Res. Technol. 2013, 19, e339–e348. [Google Scholar] [CrossRef] [PubMed]
- Klaesner, J.W.; Hastings, M.K.; Zou, D.; Lewis, C.; Mueller, M.J. Plantar tissue stiffness in patients with diabetes mellitus and peripheral neuropathy. Arch. Phys. Med. Rehabil. 2002, 83, 1796–1801. [Google Scholar] [CrossRef] [PubMed]
- Kwan, R.L.-C.; Zheng, Y.-P.; Cheing, G.L.-Y. The effect of aging on the biomechanical properties of plantar soft tissues. Clin. Biomech. 2010, 25, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Lechner, A.; Akdeniz, M.; Tomova-Simitchieva, T.; Bobbert, T.; Moga, A.; Lachmann, N.; Blume-Peytavi, U.; Kottner, J. Comparing skin characteristics and molecular markers of xerotic foot skin between diabetic and non-diabetic subjects: An exploratory study. J. Tissue Viability 2019, 28, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, M.; Song, W.; Zhang, Y.; Yu, D.-G.; Liu, Y. Electrospun core (HPMC–acetaminophen)–shell (PVP–sucralose) nanohybrids for rapid drug delivery. Gels 2022, 8, 357. [Google Scholar] [CrossRef]
- Punyamoonwongsa, P.; Klayya, S.; Sajomsang, W.; Kunyanee, C.; Aueviriyavit, S. Silk sericin semi-interpenetrating network hydrogels based on PEG-Diacrylate for wound healing treatment. Int. J. Polym. Sci. 2019, 2019, 4740765. [Google Scholar] [CrossRef] [Green Version]
- Cavallo, A.; Madaghiele, M.; Masullo, U.; Lionetto, M.G.; Sannino, A. Photo-crosslinked poly (ethylene glycol) diacrylate (PEGDA) hydrogels from low molecular weight prepolymer: Swelling and permeation studies. J. Appl. Polym. Sci. 2017, 134, 44380. [Google Scholar] [CrossRef]
- Savari, R.; Shafiei, M.; Galehdari, H.; Kesmati, M. Expression of VEGF and TGF-β genes in skin wound healing process induced using phenytoin in male rats. Jundishapur J. Health Sci. 2019, 11, e86041. [Google Scholar] [CrossRef] [Green Version]
- Kant, V.; Gopal, A.; Kumar, D.; Pathak, N.N.; Ram, M.; Jangir, B.L.; Tandan, S.K.; Kumar, D. Curcumin-induced angiogenesis hastens wound healing in diabetic rats. J. Surg. Res. 2015, 193, 978–988. [Google Scholar] [CrossRef]
- Lichtman, M.K.; Otero-Vinas, M.; Falanga, V. Transforming growth factor beta (TGF-β) isoforms in wound healing and fibrosis. Wound Repair Regen. 2016, 24, 215–222. [Google Scholar] [CrossRef]
- Mitchell, A.C.; Briquez, P.S.; Hubbell, J.A.; Cochran, J.R. Engineering growth factors for regenerative medicine applications. Acta Biomater. 2016, 30, 1–12. [Google Scholar] [CrossRef]
- Laiva, A.L.; O’Brien, F.J.; Keogh, M.B. Innovations in gene and growth factor delivery systems for diabetic wound healing. J. Tissue Eng. Regen. Med. 2018, 12, e296–e312. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, X.; Liu, R.; Gong, Y.; Wang, M.; Huang, Q.; Feng, Q.; Yu, B. Zero-order controlled release of BMP2-derived peptide P24 from the chitosan scaffold by chemical grafting modification technique for promotion of osteogenesis in vitro and enhancement of bone repair in vivo. Theranostics 2017, 7, 1072. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Kim, H.; Kwak, G.; Yoon, H.Y.; Jo, S.D.; Lee, J.E.; Cho, D.; Kwon, I.C.; Kim, S.H. Development of biocompatible HA hydrogels embedded with a new synthetic peptide promoting cellular migration for advanced wound care management. Adv. Sci. 2018, 5, 1800852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Rikabi, A.H.; Tobin, D.J.; Riches-Suman, K.; Thornton, M.J. Dermal fibroblasts cultured from donors with type 2 diabetes mellitus retain an epigenetic memory associated with poor wound healing responses. Sci. Rep. 2021, 11, 1474. [Google Scholar] [CrossRef]
- Lu, Y.; Azad, N.; Wang, L.; Iyer, A.K.; Castranova, V.; Jiang, B.-H.; Rojanasakul, Y. Phosphatidylinositol-3-kinase/akt regulates bleomycin-induced fibroblast proliferation and collagen production. Am. J. Respir. Cell Mol. Biol. 2010, 42, 432–441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Hua, W.; Li, X.; Wang, W. Suppression of human tenon fibroblast cell proliferation by lentivirus-mediated VEGF small hairpin RNA. J. Ophthalmol. 2017, 2017, 7982051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brugmans, M.; Cassiman, J.J.; Vanderheydt, L.; Oosterlinck, A.J.; Vlietinck, R.; Van Den Berghe, H. Quantification of the degree of cell spreading of human fibroblasts by semi-automated analysis of the cell perimeter. Cytom. J. Int. Soc. Anal. Cytol. 1983, 3, 262–268. [Google Scholar] [CrossRef]
- Mahdi, S.H.; Cheng, H.; Li, J.; Feng, R. The effect of TGF-beta-induced epithelial–mesenchymal transition on the expression of intracellular calcium-handling proteins in T47D and MCF-7 human breast cancer cells. Arch. Biochem. Biophys. 2015, 583, 18–26. [Google Scholar] [CrossRef]
- Sliogeryte, K.; Gavara, N. Vimentin plays a crucial role in fibroblast ageing by regulating biophysical properties and cell migration. Cells 2019, 8, 1164. [Google Scholar] [CrossRef] [Green Version]
- Qin, Z.; Balimunkwe, R.; Quan, T. Age-related reduction of dermal fibroblast size upregulates multiple matrix metalloproteinases as observed in aged human skin in vivo. Br. J. Dermatol. 2017, 177, 1337–1348. [Google Scholar] [CrossRef]
- Fairbanks, B.D.; Schwartz, M.P.; Bowman, C.N.; Anseth, K.S. Photoinitiated polymerization of PEG-diacrylate with lithium phenyl-2,4,6-trimethylbenzoylphosphinate: Polymerization rate and cytocompatibility. Biomaterials 2009, 30, 6702–6707. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Vergara, A.C.; Van Drunen, R.; Cagle, T.; Munoz-Pinto, D.J. Modeling the effects of hyaluronic acid degradation on the regulation of human astrocyte phenotype using multicomponent interpenetrating polymer networks (mIPNs). Sci. Rep. 2020, 10, 20734. [Google Scholar] [CrossRef] [PubMed]
- Cruise, G.M.; Scharp, D.S.; Hubbell, J.A. Characterization of permeability and network structure of interfacially photopolymerized poly (ethylene glycol) diacrylate hydrogels. Biomaterials 1998, 19, 1287–1294. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Pinto, D.J.; Erndt-Marino, J.D.; Becerra-Bayona, S.M.; Guiza-Arguello, V.R.; Samavedi, S.; Malmut, S.; Reichert, W.M.; Russell, B.; Höök, M.; Hahn, M.S. Evaluation of late outgrowth endothelial progenitor cell and umbilical vein endothelial cell responses to thromboresistant collagen-mimetic hydrogels. J. Biomed. Mater. Res. Part A 2017, 105, 1712–1724. [Google Scholar] [CrossRef]
- Jimenez-Vergara, A.C.; Zurita, R.; Jones, A.; Diaz-Rodriguez, P.; Qu, X.; Kusima, K.L.; Hahn, M.S.; Munoz-Pinto, D.J. Refined assessment of the impact of cell shape on human mesenchymal stem cell differentiation in 3D contexts. Acta Biomater. 2019, 87, 166–176. [Google Scholar] [CrossRef] [PubMed]




| Bond | Wavenumber [cm−1] | Vibration Type |
|---|---|---|
| CH2 | 2889 | Asymmetrical stretching vibrations |
| C=O | 1721 | Symmetrical stretching vibrations |
| C=O | 1650 | Amide I band vibrations |
| C=C | 1623 | Vibrations of the aliphatic double bond |
| C=O | 1520 | Amide II band vibrations |
| C-O | 1110 | Stretching vibrations |
| CH2=CH | 960 | Out-of-plane vibrations symmetrical stretching |
| CH2=CH | 843 | Symmetrical stretching vibrations |
| Concentration [% w/w] | Complex Modulus (E*) [kPa] | Stiffness [kN/m] | q | q′ | Mc [kDa/mol] | Mesh Size (ξ) [nm] |
|---|---|---|---|---|---|---|
| 10 | 47.7 ± 7.5 | 1.9 ± 0.3 | 15.5 ± 0.7 | 10.9 ± 0.4 | 17.5 ± 1.0 | 21.9 ± 1.0 |
| Growth Factor (GF) | Initial GF Concentration in Solution [µg/mL] | GF Bound to PEGDA-Peptides Hydrogels [µg/mL] | Binding Level [ng/mm2] |
|---|---|---|---|
| hTGFβ1 | 10 | 3.80 ± 0.41 | 3.19 ± 0.34 |
| hVEGF | 10 | 0.73 ± 0.07 | 0.62 ± 0.06 |
| Hydrogel | RGDS [mM] | KGLPLGNSH [mM] | DRVQRQTTTVVA [mM] |
|---|---|---|---|
| Control | 1 | 0 | 0 |
| hTGFβ1bp | 1 | 1 | 0 |
| hVEGFbp | 1 | 0 | 1 |
| Gene | Primer Sequence | Brand |
|---|---|---|
| β-actin | F: CACCATTGGCAATGAGCGGTTC | Fisher-Eurofins |
| R: AGGTCTTTGCGGATGTCCACGT | ||
| PCNA | F: GCTCCAGCGGTGTAAACCTGCA | Fisher-Eurofins |
| R: CGTGCAAAT TCACCAGAAGGCA | ||
| COL1A1 | F: GATTCCCTGGACCTAAAGGTGC | Fisher-Eurofins |
| R: AGCCTCTCCATCTTTGCCAGCA | ||
| TGFβ1 | F: TACCTGAACCCGTGTTGCTCTC | Fisher-Eurofins |
| R: GTTGCTGAGGTATCGCCAGGAA | ||
| Vimentin | F: ACGTCTTGACCTTGAACGCA | Fisher-Eurofins |
| R: GGCTGCCTTACCCTCATTCA | ||
| MMP-1 | F: ATGAAGCAGCCCAGATGTGGAG | Fisher-Eurofins |
| R: TGGTCCACATCTGCTCTTGGCA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clevenger, A.J.; Jimenez-Vergara, A.C.; Tsai, E.H.; de Barros Righes, G.; Díaz-Lasprilla, A.M.; Ramírez-Caballero, G.E.; Munoz-Pinto, D.J. Growth Factor Binding Peptides in Poly (Ethylene Glycol) Diacrylate (PEGDA)-Based Hydrogels for an Improved Healing Response of Human Dermal Fibroblasts. Gels 2023, 9, 28. https://doi.org/10.3390/gels9010028
Clevenger AJ, Jimenez-Vergara AC, Tsai EH, de Barros Righes G, Díaz-Lasprilla AM, Ramírez-Caballero GE, Munoz-Pinto DJ. Growth Factor Binding Peptides in Poly (Ethylene Glycol) Diacrylate (PEGDA)-Based Hydrogels for an Improved Healing Response of Human Dermal Fibroblasts. Gels. 2023; 9(1):28. https://doi.org/10.3390/gels9010028
Chicago/Turabian StyleClevenger, Abigail J., Andrea C. Jimenez-Vergara, Erin H. Tsai, Gabriel de Barros Righes, Ana M. Díaz-Lasprilla, Gustavo E. Ramírez-Caballero, and Dany J. Munoz-Pinto. 2023. "Growth Factor Binding Peptides in Poly (Ethylene Glycol) Diacrylate (PEGDA)-Based Hydrogels for an Improved Healing Response of Human Dermal Fibroblasts" Gels 9, no. 1: 28. https://doi.org/10.3390/gels9010028