Mechanism of Synergistic Photoinactivation Utilizing Curcumin and Lauric Arginate Ethyl Ester against Escherichia coli and Listeria innocua
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Stock Curcumin-LAE Solution
2.3. Stability of Encapsulated Curcumin after Dilution
2.4. Bacterial Culture Conditions
2.5. Bacterial Photoinactivation
2.6. Bacterial Growth after Photoinactivation
2.6.1. Monitoring Growth Using a Plate Reader
2.6.2. Microbial Growth Modeling
2.7. Scanning Electron Microscopy (SEM)
2.8. Transmission Electron Microscopy (TEM)
2.9. Live/Dead Cell Assay
2.10. Data Acquisition and Analysis
3. Results and Discussion
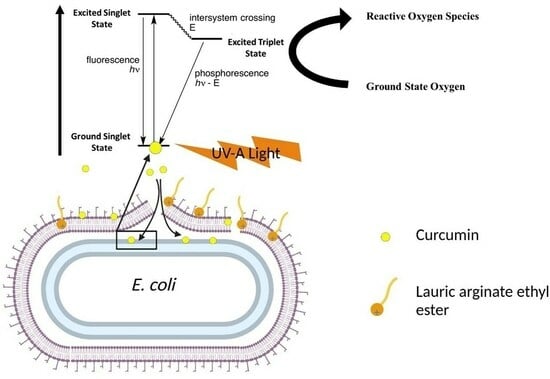
3.1. Possible Mechanisms of Synergistic Photoinactivation
3.2. Recovery of Cells after Photoinactivation
3.3. SEM Micrographs
3.4. TEM Micrographs
3.5. Live/Dead Cell Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rineh, A.; Dolla, N.K.; Ball, A.R.; Magana, M.; Bremner, J.B.; Hamblin, M.R.; Tegos, G.P.; Kelso, M.J. Attaching the NorA efflux pump inhibitor INF55 to methylene blue enhances antimicrobial photodynamic inactivation of methicillin-resistant Staphylococcus aureus in vitro and in vivo. ACS Infect. Dis. 2017, 3, 756–766. [Google Scholar] [CrossRef]
- Kettlitz, B.; Kemendi, G.; Thorgrimsson, N.; Cattoor, N.; Verzegnassi, L.; Le Bail-Collet, Y.; Maphosa, F.; Perrichet, A.; Christall, B.; Stadler, R.H. Why chlorate occurs in potable water and processed foods: A critical assessment and challenges faced by the food industry. Food Addit. Contam. Part A 2016, 33, 968–982. [Google Scholar] [CrossRef]
- Langsrud, S.; Sidhu, M.S.; Heir, E.; Holck, A.L. Bacterial disinfectant resistance—A challenge for the food industry. Int. Biodeterior. Biodegrad. 2003, 51, 283–290. [Google Scholar] [CrossRef]
- Hegstad, K.; Langsrud, S.; Lunestad, B.T.; Scheie, A.A.; Sunde, M.; Yazdankhah, S.P. Does the wide use of quaternary ammonium compounds enhance the selection and spread of antimicrobial resistance and thus threaten our health? Microb. Drug Resist. 2010, 16, 91–104. [Google Scholar] [CrossRef]
- Tong, C.; Hu, H.; Chen, G.; Li, Z.; Li, A.; Zhang, J. Chlorine disinfectants promote microbial resistance in Pseudomonas sp. Environ. Res. 2021, 199, 111296. [Google Scholar] [CrossRef]
- Khan, S.; Beattie, T.K.; Knapp, C.W. Relationship between antibiotic- and disinfectant-resistance profiles in bacteria harvested from tap water. Chemosphere 2016, 152, 132–141. [Google Scholar] [CrossRef]
- Cieplik, F.; Deng, D.; Crielaard, W.; Buchalla, W.; Hellwig, E.; Al-Ahmad, A.; Maisch, T. Antimicrobial photodynamic therapy—What we know and what we don’t. Crit. Rev. Microbiol. 2018, 44, 571–589. [Google Scholar] [CrossRef]
- Vatansever, F.; De Melo, W.C.; Avci, P.; Vecchio, D.; Sadasivam, M.; Gupta, A.; Chandran, R.; Karimi, M.; Parizotto, N.A.; Yin, R.; et al. Antimicrobial strategies centered around reactive oxygen species—Bactericidal antibiotics, photodynamic therapy, and beyond. FEMS Microbiol. Rev. 2013, 37, 955–989. [Google Scholar] [CrossRef]
- EFSA. Refined exposure assessment for curcumin (E 100). EFSA J. 2014, 12, 3876. [Google Scholar] [CrossRef]
- Karlsen, J.; Tønnesen, H.H. Studies on curcumin and curcuminoids. Eur. Food Res. Technol. 1985, 180, 402–404. [Google Scholar] [CrossRef]
- Ryu, V.; Chuesiang, P.; Corradini, M.G.; McLandsborough, L.; Jin, T.; Ngo, H.; Fan, X. Synergistic photoinactivation of Escherichia coli and Listeria innocua by curcumin and lauric arginate ethyl ester micelles. LWT 2023, 173, 114317. [Google Scholar] [CrossRef]
- Ryu, V.; Chuesiang, P.; Ngo, H.; Ashby, R.D.; Fan, X. Sustainable bio-based antimicrobials derived from fatty acids: Synthesis, safety, and efficacy. Crit. Rev. Food Sci. Nutr. 2022, 1–13. [Google Scholar] [CrossRef]
- FDA. GRAS Exemption Claim for Ethyl-N-lauroyl-l-arginate Hydrochloride; US Food and Drug Administration: Bethesda, MD, USA, 2005.
- Asker, D.; Weiss, J.; McClements, D.J. Analysis of the interactions of a cationic surfactant (Lauric arginate) with an anionic biopolymer (pectin): Isothermal titration calorimetry, light scattering, and microelectrophoresis. Langmuir 2009, 25, 116–122. [Google Scholar] [CrossRef]
- Ryu, V.; Ruiz-Ramirez, S.; Chuesiang, P.; McLandsborough, L.A.; McClements, D.J.; Corradini, M.G. Use of Micellar Delivery Systems to Enhance Curcumin’s Stability and Microbial Photoinactivation Capacity. Foods 2021, 10, 1777. [Google Scholar] [CrossRef]
- Corradini, M.; Peleg, M. Estimating non-isothermal bacterial growth in foods from isothermal experimental data. J. Appl. Microbiol. 2005, 99, 187–200. [Google Scholar] [CrossRef]
- Dahl, T.A.; McGowan, W.M.; Shand, M.A.; Srinivasan, V.S. Photokilling of bacteria by the natural dye curcumin. Arch. Microbiol. 1989, 151, 183–185. [Google Scholar] [CrossRef]
- Yang, X.; Rai, R.; Huu, C.N.; Nitin, N. Synergistic antimicrobial activity by light or thermal treatment and lauric arginate: Membrane damage and oxidative stress. Appl. Environ. Microbiol. 2019, 85, e01033-19. [Google Scholar] [CrossRef]
- de Oliveira, E.F.; Tosati, J.V.; Tikekar, R.V.; Monteiro, A.R.; Nitin, N. Antimicrobial activity of curcumin in combination with light against Escherichia coli O157: H7 and Listeria innocua: Applications for fresh produce sanitation. Postharvest Biol. Technol. 2018, 137, 86–94. [Google Scholar] [CrossRef]
- Pattanayaiying, R.; H-Kittikun, A.; Cutter, C.N. Effect of lauric arginate, nisin Z, and a combination against several food-related bacteria. Int. J. Food Microbiol. 2014, 188, 135–146. [Google Scholar] [CrossRef]
- Pokhrel, R.; Shakya, R.; Baral, P.; Chapagain, P. Molecular Modeling and Simulation of the Peptidoglycan Layer of Gram-Positive Bacteria Staphylococcus aureus. J. Chem. Inf. Model. 2022, 62, 4955–4962. [Google Scholar] [CrossRef]
- Jori, G.; Brown, S.B. Photosensitized inactivation of microorganisms. Photochem. Photobiol. Sci. 2004, 3, 403–405. [Google Scholar] [CrossRef]
- Czakaj, A.; Jarek, E.; Krzan, M.; Warszyński, P. Ethyl lauroyl arginate, an inherently multicomponent surfactant system. Molecules 2021, 26, 5894. [Google Scholar] [CrossRef]
- Wilhelm, M.J.; Gh, M.S.; Wu, T.; Li, Y.; Chang, C.-M.; Ma, J.; Dai, H.-L. Determination of bacterial surface charge density via saturation of adsorbed ions. Biophys. J. 2021, 120, 2461–2470. [Google Scholar] [CrossRef]
- Coronel-León, J.; López, A.; Espuny, M.; Beltran, M.; Molinos-Gómez, A.; Rocabayera, X.; Manresa, A. Assessment of antimicrobial activity of Nα-lauroyl arginate ethylester (LAE®) against Yersinia enterocolitica and Lactobacillus plantarum by flow cytometry and transmission electron microscopy. Food Control 2016, 63, 1–10. [Google Scholar] [CrossRef]
- Ghoshal, M.; Ryu, V.; McLandsborough, L. Evaluation of the efficacy of antimicrobials against pathogens on food contact surfaces using a rapid microbial log reduction detection method. Int. J. Food Microbiol. 2022, 373, 109699. [Google Scholar] [CrossRef]
- Garcez, A.S.; Kaplan, M.; Jensen, G.J.; Scheidt, F.R.; Oliveira, E.M.; Suzuki, S.S. Effects of antimicrobial photodynamic therapy on antibiotic-resistant Escherichia coli. Photodiagnosis Photodyn. Ther. 2020, 32, 102029. [Google Scholar] [CrossRef]
- Ehrenberg, B.; Malik, Z.; Nitzan, Y.; Ladan, H.; Johnson, F.M.; Hemmi, G.; Sessler, J.L. The binding and photosensitization effects of tetrabenzoporphyrins and texaphyrin in bacterial cells. Lasers Med. Sci. 1993, 8, 197–203. [Google Scholar] [CrossRef]
- Dolanský, J.; Henke, P.; Malá, Z.; Žárská, L.; Kubát, P.; Mosinger, J. Antibacterial nitric oxide- and singlet oxygen-releasing polystyrene nanoparticles responsive to light and temperature triggers. Nanoscale 2018, 10, 2639–2648. [Google Scholar] [CrossRef]
- Ehrenberg, B.; Malik, Z.; Nitzan, Y. Fluorescence Spectral Changes of Hematoporphyrin Derivative upon Binding to Lipid Vesicles, Staphylococcus aureus and Escherichia coli Cells. Photochem. Photobiol. 1985, 41, 429–435. [Google Scholar] [CrossRef]










| Sample | pH | Irradiated | Parameters | ||
|---|---|---|---|---|---|
| a | k | tc | |||
| (-) | (h−1) | (h) | |||
| Control | 3.5 | NO | 13.7 | 0.63 | 5.24 |
| YES | 11.8 | 1.06 | 5.32 | ||
| 7 | NO | 12 | 0.29 | 7.08 | |
| YES | 12.7 | 0.28 | 7.5 | ||
| 1 μmol/L Curcumin | 3.5 | NO | 12.3 | 0.91 | 4.99 |
| YES | 12.7 | 1.51 | 5.85 | ||
| 7 | NO | 8.31 | 0.48 | 5.54 | |
| YES | 6.06 | 1.49 | 5.05 | ||
| 105 μg/mL LAE | 3.5 | NO | 14.3 | 0.71 | 6.85 |
| YES | 13.5 | 0.72 | 7.46 | ||
| 7 | NO | 4.51 | 1.24 | 18.2 | |
| YES | 0.64 | 3.07 | 1.4 | ||
| Curcumin-LAE solution | 3.5 | NO | 13.8 | 0.77 | 7.59 |
| YES | 0.16 | 5.53 | 0.63 | ||
| 7 | NO | 0.61 | 1.34 | 1.54 | |
| YES | 0.69 | 1.17 | 1.34 | ||
| Sample | pH | Irradiated | Parameters | ||
|---|---|---|---|---|---|
| a | k | tc | |||
| (-) | (h−1) | (h) | |||
| Control | 3.5 | NO | 3.41 | 0.97 | 8.67 |
| YES | 3.96 | 0.79 | 10 | ||
| 7 | NO | 4 | 1.01 | 8.32 | |
| YES | 4.06 | 0.91 | 9.51 | ||
| 1 µmol/L Curcumin | 3.5 | NO | 4.24 | 0.79 | 9.03 |
| YES | - 1 | - | - | ||
| 7 | NO | 4.23 | 0.74 | 8.95 | |
| YES | 4.34 | 0.75 | 18.9 | ||
| 105 μg/mL LAE | 3.5 | NO | 3.98 | 0.23 | 17.8 |
| YES | 1.51 | 0.72 | 17.8 | ||
| 7 | NO | - | - | - | |
| YES | - | - | - | ||
| Curcumin-LAE solution | 3.5 | NO | 2.66 | 0.66 | 17.7 |
| YES | - | - | - | ||
| 7 | NO | 0.02 | 1.59 | 2.47 | |
| YES | 0.01 | 0.25 | 3.2 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryu, V.; Uknalis, J.; Corradini, M.G.; Chuesiang, P.; McLandsborough, L.; Ngo, H.; Jin, T.; Fan, X. Mechanism of Synergistic Photoinactivation Utilizing Curcumin and Lauric Arginate Ethyl Ester against Escherichia coli and Listeria innocua. Foods 2023, 12, 4195. https://doi.org/10.3390/foods12234195
Ryu V, Uknalis J, Corradini MG, Chuesiang P, McLandsborough L, Ngo H, Jin T, Fan X. Mechanism of Synergistic Photoinactivation Utilizing Curcumin and Lauric Arginate Ethyl Ester against Escherichia coli and Listeria innocua. Foods. 2023; 12(23):4195. https://doi.org/10.3390/foods12234195
Chicago/Turabian StyleRyu, Victor, Joseph Uknalis, Maria G. Corradini, Piyanan Chuesiang, Lynne McLandsborough, Helen Ngo, Tony Jin, and Xuetong Fan. 2023. "Mechanism of Synergistic Photoinactivation Utilizing Curcumin and Lauric Arginate Ethyl Ester against Escherichia coli and Listeria innocua" Foods 12, no. 23: 4195. https://doi.org/10.3390/foods12234195