Abstract
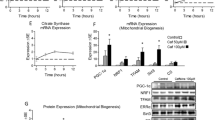
The mitochondrial uncoupling protein-2 (UCP2) can uncouple phosphorylation to subserve several functions. It has been reported that the insulin sensitizers, thiazolidinediones (TZDs), increase UCP2 mRNA levels and, more recently, that TZDs stimulate UCP2 reporter genes but that the sequences involved do not bind peroxisome proliferator-activated receptor γ (PPARγ). We report here that TZDs stimulated UCP2 gene (ucp2) transcription in L6 myotubules involving an indirect mechanism. L6 cells contained comparatively small amounts of PPARγ mRNA but clearly detectable amounts of PPARγ2 protein. UCP2 mRNA levels were increased in a time- and concentration-dependent manner by TZDs. UCP2 mRNA had slow turnover (t 1/2 ≈ 38 h), and this was not affected by TZDs. Bisphenol A diglycidyl ether, a PPARγ antagonist, concentration dependently inhibited the TZD-induced increase in UCP2 mRNA. Blockade of protein synthesis with cycloheximide as well as abrogation of mitogen-activated protein kinase (MAPK) activity with PD98059 or U0126 also prevented the TZD-induced increase in UCP2 mRNA. As with autologous UCP2 mRNA, TZDs stimulated reporter gene expression directed by ucp2 sequences in transiently transfected L6 cells. The effect was enhanced by cotransfection of PPARγ + retinoid X receptor γ and prevented by MEK blockade. TZDs, however, did not increase the activation of MAPK, nor did its activation by other means (change of medium, insulin-like growth factor-1, insulin) increase UCP2 mRNA, indicating that phosphorylation is not limiting. These results suggest that TZDs indirectly stimulate ucp2 transcription by inducing—via PPARγ—limiting amounts of a protein, which must be phosphorylated by MAPK to stimulate the gene.
Similar content being viewed by others
References
Ricquier, D. and Bouillaud, F. (2000). Biochem. J. 345, 161–179.
Adams, S. H. (2000). J. Nutr. 130, 711–714.
Gong, D. W., Monemdjou, S., Gavrilova, O., et al. (2000). J. Biol. Chem. 275, 16251–16257.
Vidal-Puig, A. J., Grujic, D., Zhang, C. Y., et al. (2000). J. Biol. Chem. 275, 16258–16266.
Arsenijevic, D., Onuma, H., Pecqueur, C., et al. (2000). Nat. Genet. 26, 435–439.
Chan, C. B., MacDonald, P. E., Saleh, M. C., Johns, D. C., Marban, E., and Wheeler, M. B. (1999). Diabetes 48, 1482–1486.
Martin, G., Schoonjans, K., Staels, B., and Auwerx, J. (1998). Atherosclerosis 137(Suppl.), S75-S80.
Donnelly, R. and Qu, X. Q. (1998). Clin. Exp. Pharmacol. Physiol. 25, 79–87.
Elbrecht, A., Chen, Y. L., Cullinan, C. A., et al. (1996). Biochem. Biophys. Res. Commun. 224, 431–437.
Yanase, T., Yashiro, T., Takitani, K., et al. (1997). Biochem. Biophys. Res. Commun. 233, 320–324.
Fajas, L., Auboeuf, D., Raspe, E., et al. (1997). J. Biol. Chem. 272, 18779–18789.
Reginato, M. J. and Lazar, M. A. (1999). Trends Endocrinol. Metab. 10, 9–13.
Desvergne, B. and Wahli, W. (1999). Endocr. Rev. 20, 649–688.
Steppan, C. M., Bailey, S. T., Bhat, S., et al. (2001). Nature 409, 307–312.
Vidal-Puig, A. and O’Rahilly, S. (2001). Clin. Endocrinol. (Oxf.) 55, 437–438.
Ballard, F. J. (1994). J. Nutr. 124, 1540S-1545S.
D’Amore, T. and Lo, T. C. (1986). Biochem. Cell Biol. 64, 1081–1091.
Klip, A. and Marette, A. (1992). J. Cell Biochem. 48, 51–60.
Khayat, Z. A., Tsakiridis, T., Ueyama, A., Somwar, R., Ebina, Y., and Klip, A. (1998). Am. Physiol. 275, C1487-C1497.
Aubert, J., Champigny, O., Saintmarc, P., et al. (1997). Biochem. Biophys. Res. Commun. 38, 606–611.
Camirand, A., Marie, V., Rabelo, R., and Silva, J. E. (1998). Endocrinology 139, 428–431.
Shimabukuro, M., Zhou, Y. T., Lee, Y., and Unger, R. H. (1997). Biochem. Biophys. Res. Commun. 237, 359–361.
Strobel, A., Siquier, K., Zilberfarb, V., Strosberg, A. D., and Issad, T. (1999). Diabetologia 42, 527–533.
Viguerie-Bascands, N., Saulnier-Blache, J. S., Dandine, M., Dauzats, M., Daviaud, D., and Langin, D. (1999). Biochem. Biophys. Res. Commun. 256, 138–141.
Zierath, J. R., Ryder, J. W., Doebber, T., et al. (1998). Endocrinology 139, 5034–5041.
Nagase, I., Yoshida, S., Canas, X., et al. (1999). FEBS Lett. 461, 319–322.
Medvedev, A. V., Snedden, S. K., Raimbault, S., Ricquier, D., and Collins, S. (2001). J. Biol. Chem. 276, 10817–10823.
Schoonjans, K. and Auwerx, J. (2000). Lancet 355, 1008–1010.
Braissant, O., Foufelle, F., Scotto, C., Dauca, M., and Wahli, W. (1996). Endocrinology 137, 354–366.
Goke, R., Goke, A., Goke, B., and Chen, Y. (2000). Cell Immunol. 201, 77–82.
Yang, X. Y., Wang, L. H., Chen, T., et al. (2000). J. Biol. Chem. 275, 4541–4544.
Martin, G., Schoonjans, K., Lefebvre, A. M., Staels, B., and Auwerx, J. (1997). J. Biol. Chem. 272, 28210–28217.
Wright, H. M., Clish, C. B., Mikami, T., et al. (2000). J. Biol. Chem. 275, 1873–1877.
Liu, Q. and Linney, E. (1993). Mol. Endocrinol. 7, 651–658.
Hu, E., Kim, J. B., Sarraf, P., and Spiegelman, B. M. (1996). Science 274, 2100–2103.
Shao, D., Rangwala, S. M., Bailey, S. T., Krakow, S. L., Reginato, M. J., and Lazar, M. A. (1998). Nature 396, 377–380.
Brunmair, B., Gras, F., Neschen, S., et al. (2001). Diabetes 50, 2309–2315.
Wang, M., Wise, S. C., Leff, T., and Su, T. Z. (1999). Diabetes 48, 254–260.
Chawla, A., Barak, Y., Nagy, L., Liao, D., Tontonoz, P., and Evans, R. M. (2001). Nat. Med. 7, 48–52.
Palakurthi, S. S., Aktas, H., Grubissich, L. M., Mortensen, R. M., and Halperin, J. A. (2001). Cancer Res. 61, 6213–6218.
Marcus, S. L., Miyata, K. S., Zhang, B., Subramani, S., Rachubinski, R. A., and Capone, J. P. (1993). Proc. Natl. Acad. Sci. USA 90, 5723–5727.
Bishop-Bailey, D., Hla, T., and Warner, T. D. (2000). Br. J. Pharmacol. 131, 651–654.
Burris, T. P., Pelton, P. D., Zhou, L., Osborne, M. C., Cryan, E., and Demarest, K. T. (1999). Mol. Endocrinol. 13, 410–417.
Lehmann, J. M., Moore, L. B., Smith-Oliver, T. A., Wilkison, W. O., Willson, T. M., and Kliewer, S. A. (1995). J. Biol. Chem. 270, 12953–12956.
Cha, B. S., Ciaraldi, T. P., Carter, L., et al. (2001). Diabetologia 44, 444–452.
El-Kebbi, I. M., Roser, S., and Pollet, R. J. (1994). Metab. Clin. Exp. 43, 953–958.
Hattori, Y., Akimoto, K., and Kasai, K. (2000). Biochem. Biophys. Res. Commun. 273, 1144–1149.
Taha, C., Tsakiridis, T., McCall, A., and Klip, A. (1997). Am. J. Physiol. 273, E68-E76.
Miller A. K., Inglis, A. M., Culkin, K. T., Jorkasky, D. K., and Freed, M. I. (2001). Eur. J. Clin. Pharmacol. 57, 105–109.
Prueksaritanont, T., Vega, J. M., Zhao, J., et al. (2001). J. Clin. Pharmacol. 41, 573–581.
Adams, M., Reginato, M. J., Shao, D., Lazar, M. A., and Chatterjee, V. K. (1997). J. Biol. Chem. 272, 5128–5132.
Camp, H. S. and Tafuri, S. R. (1997). J. Biol. Chem. 272, 10811–10816.
Ristow, M., Müller-Wieland, D., Pfeiffer, A., Krone, W., and Kahn, C. R. (1998). N. Engl. J. Med. 339, 953–959.
Zhang, B., Berger, J., Zhou, G. C., et al. (1996). J. Biol. Chem. 271, 31771–31774.
Koo, S. H. and Towle, H. C. (2000). J. Biol. Chem. 275, 5200–5207.
Chomczynski, P. and Sacchi, N. (1987). Anal. Biochem. 162, 156–159.
Boivin, M., Camirand, A., Carli, F., Hoffer, L. J., and Silva, J. E. (2000). J. Clin. Endocrinol. Metab. 85, 1975–1983.
Zhou, Y. T., Shimabukuro, M., Koyama, K., et al. (1997). Proc. Natl. Acad. Sci. USA 94, 6386–6390.
Yoshitomi, H., Yamazaki, K., and Tanaka, I. (1999). Biochem. J. 340, 397–404.
Rabelo, R., Schifman, A., Rubio, A., Sheng, X., and Silva, J. E. (1995). Endocrinology 136, 1003–1013.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
López-Solache, I., Marie, V., Vignault, E. et al. Regulation of uncoupling protein-2 mRNA in L6 myotubules. Endocr 19, 197–208 (2002). https://doi.org/10.1385/ENDO:19:2:197
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/ENDO:19:2:197