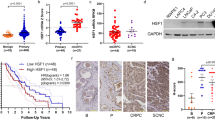
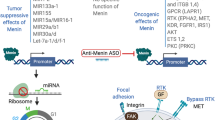
Key Points
-
Heat shock proteins (HSPs) are molecular chaperones that regulate protein homeostasis, signal transduction and transcriptional networks, and are key drivers of adaptive stress-response pathways in many malignancies
-
HSPs are highly expressed in many cancers, and subversion of their chaperone functions enables survival of malignant cells subjected to proteotoxic stress
-
HSPs are important in androgen receptor function (including ligand binding and nuclear trafficking), and many HSP client proteins modulate key signalling and transductional networks in castration-resistant prostate cancer (CRPC) cells
-
Extensive preclinical data have demonstrated the utility of targeting HSPs, including HSP90, HSP70, HSP27 and clusterin, in prostate cancer
-
Clinical development of HSP blockade as a therapeutic strategy in CRPC has been challenging, but antisense oligonucleotides targeting HSP27 and clusterin have proceeded to late-stage clinical trials
-
Integrating HSP blockade into the treatment paradigm for CRPC will require the development of potent, selective inhibitors of HSPs, and identification of rational combination strategies
Abstract
The survival of malignant cells is constantly threatened by a myriad of cellular insults. In the context of such proteotoxic stress, cancer cells activate cytoprotective adaptive pathways. Heat shock proteins (HSPs) are highly conserved molecular chaperones that are expressed at low levels under normal conditions, but upregulated by cellular stress. As molecular chaperones, HSPs control the stability and function of client proteins, preventing aggregation of misfolded proteins, facilitating intracellular protein trafficking, maintaining protein conformation to enable ligand binding, phosphorylating proteins in signalling complexes and degrading severely damaged proteins via the ubiquitin–proteasome pathway. A key client protein of several HSPs is the androgen receptor (AR). HSPs facilitate binding of dihydrotestosterone to the AR, and enhance AR-mediated transcriptional activity. The integral role of HSPs in AR function speaks to their potential utility as therapeutic targets in castration-resistant prostate cancer (CRPC), a disease state characterized by persistent activation of the androgen–AR axis. Inhibition of HSPs has the additional benefit of potentially modulating signalling and transcriptional networks that are associated with HSP client proteins in CRPC cells. As a consequence, HSPs represent highly attractive targets in the development of treatments for CRPC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Calderwood, S. K. & Ciocca, D. R. Heat shock proteins: stress proteins with Janus-like properties in cancer. Int. J. Hyperthermia 24, 31–39 (2008).
Zoubeidi, A. & Gleave, M. Small heat shock proteins in cancer therapy and prognosis. Int. J. Biochem. Cell Biol. 44, 1646–1656 (2012).
Ischia, J. & So, A. I. The role of heat shock proteins in bladder cancer. Nat. Rev. Urol. 10, 386–395 (2013).
Ischia, J., Saad, F. & Gleave, M. The promise of heat shock protein inhibitors in the treatment of castration resistant prostate cancer. Curr. Opin. Urol. 23, 194–200 (2013).
Akerfelt, M., Morimoto, R. I. & Sistonen, L. Heat shock factors: integrators of cell stress, development and lifespan. Nat. Rev. Mol. Cell Biol. 11, 545–555 (2010).
Pirkkala, L., Nykänen, P. & Sistonen, L. Roles of the heat shock transcription factors in regulation of the heat shock response and beyond. FASEB J. 15, 1118–1131 (2001).
Whitesell, L. & Lindquist, S. L. HSP90 and the chaperoning of cancer. Nat. Rev. Cancer 5, 761–772 (2005).
Jego, G., Hazoumé, A., Seigneuric, R. & Garrido, C. Targeting heat shock proteins in cancer. Cancer Lett. 332, 275–285 (2013).
Cano, L. Q., Lavery, D. N. & Bevan, C. L. Mini-review: Foldosome regulation of androgen receptor action in prostate cancer. Mol. Cell. Endocrinol. 369, 52–62 (2013).
Fang, Y., Fliss, A. E., Robins, D. M. & Caplan, A. J. Hsp90 regulates androgen receptor hormone binding affinity in vivo. J. Biol. Chem. 271, 28697–28702 (1996).
Zoubeidi, A. et al. Cooperative interactions between androgen receptor (AR) and heat-shock protein 27 facilitate AR transcriptional activity. Cancer Res. 67, 10455–10465 (2007).
Ryan, C. J. & Tindall, D. J. Androgen receptor rediscovered: the new biology and targeting the androgen receptor therapeutically. J. Clin. Oncol. 29, 3651–3658 (2011).
Powers, M. V. & Workman, P. Targeting of multiple signalling pathways by heat shock protein 90 molecular chaperone inhibitors. Endocr. Relat. Cancer 13 (Suppl. 1), S125–S135 (2006).
Taipale, M., Jarosz, D. F. & Lindquist, S. HSP90 at the hub of protein homeostasis: emerging mechanistic insights. Nat. Rev. Mol. Cell Biol. 11, 515–528 (2010).
Mollapour, M. & Neckers, L. Post-translational modifications of Hsp90 and their contributions to chaperone regulation. Biochim. Biophys. Acta 1823, 648–655 (2012).
Ferraldeschi, R. et al. In vitro and in vivo antitumor activity of the next generation HSP90 inhibitor, AT13387, in both hormone-sensitive and castration-resistant prostate cancer models [abstract 2433]. Cancer Res. http://dx.doi.org/10.1158/1538-7445.AM2013-2433.
Eskew, J. D. et al. Development and characterization of a novel C-terminal inhibitor of Hsp90 in androgen dependent and independent prostate cancer cells. BMC Cancer 11, 468 (2011).
He, S. et al. Potent activity of the Hsp90 inhibitor ganetespib in prostate cancer cells irrespective of androgen receptor status or variant receptor expression. Int. J. Oncol. 42, 35–43 (2013).
Centenera, M. M. et al. Evidence for efficacy of new Hsp90 inhibitors revealed by ex vivo culture of human prostate tumors. Clin. Cancer Res. 18, 3562–3570 (2012).
Plescia, J. et al. Rational design of shepherdin, a novel anticancer agent. Cancer Cell 7, 457–468 (2005).
Solit, D. B. et al. 17-Allylamino-17-demethoxygeldanamycin induces the degradation of androgen receptor and HER-2/neu and inhibits the growth of prostate cancer xenografts. Clin. Cancer Res. 8, 986–993 (2002).
Matthews, S. B. et al. Characterization of a novel novobiocin analogue as a putative C-terminal inhibitor of heat shock protein 90 in prostate cancer cells. Prostate 70, 27–36 (2010).
Lamoureux, F. et al. A novel HSP90 inhibitor delays castrate-resistant prostate cancer without altering serum PSA levels and inhibits osteoclastogenesis. Clin. Cancer Res. 17, 2301–2313 (2011).
Eccles, S. A. et al. NVP-AUY922: a novel heat shock protein 90 inhibitor active against xenograft tumor growth, angiogenesis, and metastasis. Cancer Res. 68, 2850–2860 (2008).
Hance, M. W. et al. Secreted Hsp90 is a novel regulator of the epithelial to mesenchymal transition (EMT) in prostate cancer. J. Biol. Chem. 287, 37732–37744 (2012).
Teng, Y., Ngoka, L., Mei, Y., Lesoon, L. & Cowell, J. K. HSP90 and HSP70 proteins are essential for stabilization and activation of WASF3 metastasis-promoting protein. J. Biol. Chem. 287, 10051–10059 (2012).
Alqawi, O., Moghaddas, M. & Singh, G. Effects of geldanamycin on HIF-1alpha mediated angiogenesis and invasion in prostate cancer cells. Prostate Cancer Prostatic Dis. 9, 126–135 (2006).
Mabjeesh, N. J. et al. Geldanamycin induces degradation of hypoxia-inducible factor 1alpha protein via the proteosome pathway in prostate cancer cells. Cancer Res. 62, 2478–2482 (2002).
Vanaja, D. K., Mitchell, S. H., Toft, D. O. & Young, C. Y. Effect of geldanamycin on androgen receptor function and stability. Cell Stress Chaperones 7, 55–64 (2002).
Saporita, A. J., Ai, J. & Wang, Z. The Hsp90 inhibitor, 17-AAG, prevents the ligand-independent nuclear localization of androgen receptor in refractory prostate cancer cells. Prostate 67, 509–520 (2007).
O'Malley, K. J. et al. Hsp90 inhibitor 17-AAG inhibits progression of LuCaP35 xenograft prostate tumors to castration resistance. Prostate 72, 1117–1123 (2012).
Chandarlapaty, S. et al. SNX2112, a synthetic heat shock protein 90 inhibitor, has potent antitumor activity against HER kinase-dependent cancers. Clin. Cancer Res. 14, 240–248 (2008).
Heath, E. I. et al. A phase II trial of 17-allylamino-17-demethoxygeldanamycin in patients with hormone-refractory metastatic prostate cancer. Clin. Cancer Res. 14, 7940–7946 (2008).
Pacey, S. et al. A phase I study of the heat shock protein 90 inhibitor alvespimycin (17-DMAG) given intravenously to patients with advanced solid tumors. Clin. Cancer Res. 17, 1561–1570 (2011).
Oh, W. K. et al. Multicenter phase II trial of the heat shock protein 90 inhibitor, retaspimycin hydrochloride (IPI-504), in patients with castration-resistant prostate cancer. Urology 78, 626–630 (2011).
Heath, E. I. et al. Phase II trial of single-agent ganetespib (STA-9090), a heat shock protein 90 (Hsp90) inhibitor in heavily pretreated patients with metastatic castration-resistant prostate cancer (mCRPC) post docetaxel-based chemotherapy: Results of a Prostate Cancer Clinical Trials Consortium (PCCTC) study [abstract 5085]. J. Clin. Oncol. 31 (Suppl.) (2013).
Rajan, A. et al. A phase I study of PF-04929113 (SNX-5422), an orally bioavailable heat shock protein 90 inhibitor, in patients with refractory solid tumor malignancies and lymphomas. Clin. Cancer Res. 17, 6831–6839 (2011).
Mattos-Arruda, L. D. et al. Phase I dose-escalation, open-label study of HSP990 administered orally in adult patients with advanced solid malignancies [abstract 2561]. J. Clin. Oncol. 31 (Suppl.) (2013).
Kang, B. H. et al. Preclinical characterization of mitochondria-targeted small molecule hsp90 inhibitors, gamitrinibs, in advanced prostate cancer. Clin. Cancer Res. 16, 4779–4788 (2010).
Lamoureux, F. et al. Suppression of heat shock protein 27 using OGX-427 induces endoplasmic reticulum stress and potentiates heat shock protein 90 inhibitors to delay castrate-resistant prostate cancer. Eur. Urol. 66, 145–155 (2014).
Lamoureux, F. et al. Clusterin inhibition using OGX-011 synergistically enhances Hsp90 inhibitor activity by suppressing the heat shock response in castrate-resistant prostate cancer. Cancer Res. 71, 5838–5849 (2011).
Ciocca, D. R., Arrigo, A. P. & Calderwood, S. K. Heat shock proteins and heat shock factor 1 in carcinogenesis and tumor development: an update. Arch. Toxicol. 87, 19–48 (2013).
Shatkina, L. et al. The cochaperone Bag-1L enhances androgen receptor action via interaction with the NH2-terminal region of the receptor. Mol. Cell. Biol. 23, 7189–7197 (2003).
Ren, A., Yan, G., You, B. & Sun, J. Down-regulation of mammalian sterile 20-like kinase 1 by heat shock protein 70 mediates cisplatin resistance in prostate cancer cells. Cancer Res. 68, 2266–2274 (2008).
Gibbons, N. B., Watson, R. W., Coffey, R. N., Brady, H. P. & Fitzpatrick, J. M. Heat-shock proteins inhibit induction of prostate cancer cell apoptosis. Prostate 45, 58–65 (2000).
Roigas, J., Wallen, E. S., Loening, S. A. & Moseley, P. L. Effects of combined treatment of chemotherapeutics and hyperthermia on survival and the regulation of heat shock proteins in Dunning R3327 prostate carcinoma cells. Prostate 34, 195–202 (1998).
Gabai, V. L., Budagova, K. R. & Sherman, M. Y. Increased expression of the major heat shock protein Hsp72 in human prostate carcinoma cells is dispensable for their viability but confers resistance to a variety of anticancer agents. Oncogene 24, 3328–3338 (2005).
Koya, K. et al. MKT-077, a novel rhodacyanine dye in clinical trials, exhibits anticarcinoma activity in preclinical studies based on selective mitochondrial accumulation. Cancer Res. 56, 538–543 (1996).
Britten, C. D. et al. A phase I and pharmacokinetic study of the mitochondrial-specific rhodacyanine dye analog MKT 077. Clin. Cancer Res. 6, 42–49 (2000).
Parcellier, A. et al. Small heat shock proteins HSP27 and alphaB-crystallin: cytoprotective and oncogenic functions. Antioxid. Redox Signal. 7, 404–413 (2005).
Rocchi, P. et al. Increased Hsp27 after androgen ablation facilitates androgen-independent progression in prostate cancer via signal transducers and activators of transcription 3-mediated suppression of apoptosis. Cancer Res. 65, 11083–11093 (2005).
Andrieu, C. et al. Heat shock protein 27 confers resistance to androgen ablation and chemotherapy in prostate cancer cells through eIF4E. Oncogene 29, 1883–1896 (2010).
Baylot, V. et al. Targeting TCTP as a new therapeutic strategy in castration-resistant prostate cancer. Mol. Ther. 20, 2244–2256 (2012).
Zoubeidi, A. et al. Hsp27 promotes insulin-like growth factor-I survival signaling in prostate cancer via p90Rsk-dependent phosphorylation and inactivation of BAD. Cancer Res. 70, 2307–2317 (2010).
Hayashi, N. et al. Hsp27 silencing coordinately inhibits proliferation and promotes Fas-induced apoptosis by regulating the PEA-15 molecular switch. Cell Death Differ. 19, 990–1002 (2012).
Xu, L., Chen, S. & Bergan, R. C. MAPKAPK2 and HSP27 are downstream effectors of p38 MAP kinase-mediated matrix metalloproteinase type 2 activation and cell invasion in human prostate cancer. Oncogene 25, 2987–2998 (2006).
Shiota, M. et al. Hsp27 regulates epithelial mesenchymal transition, metastasis, and circulating tumor cells in prostate cancer. Cancer Res. 73, 3109–3119 (2013).
Razandi, M., Pedram, A. & Levin, E. R. Heat shock protein 27 is required for sex steroid receptor trafficking to and functioning at the plasma membrane. Mol. Cell. Biol. 30, 3249–3261 (2010).
Bubendorf, L. et al. Hormone therapy failure in human prostate cancer: analysis by complementary DNA and tissue microarrays. J. Natl Cancer Inst. 91, 1758–1764 (1999).
Cornford, P. A. et al. Heat shock protein expression independently predicts clinical outcome in prostate cancer. Cancer Res. 60, 7099–7105 (2000).
Garrido, C. et al. Heat shock protein 27 enhances the tumorigenicity of immunogenic rat colon carcinoma cell clones. Cancer Res. 58, 5495–5499 (1998).
Rocchi, P. et al. Heat shock protein 27 increases after androgen ablation and plays a cytoprotective role in hormone-refractory prostate cancer. Cancer Res. 64, 6595–6602 (2004).
Foster, C. S. et al. Hsp-27 expression at diagnosis predicts poor clinical outcome in prostate cancer independent of ETS-gene rearrangement. Br. J. Cancer 101, 1137–1144 (2009).
Zoubeidi, A. et al. Hsp27 promotes insulin-like growth factor-I survival signaling in prostate cancer via p90Rsk-dependent phosphorylation and inactivation of BAD. Cancer Res. 70, 2307–2317 (2010).
Aloy, M. T. et al. Protective role of Hsp27 protein against gamma radiation-induced apoptosis and radiosensitization effects of Hsp27 gene silencing in different human tumor cells. Int. J. Radiat. Oncol. Biol. Phys. 70, 543–553 (2008).
Zielinski, R. & Chi, K. N. Custirsen (OGX-011): a second-generation antisense inhibitor of clusterin in development for the treatment of prostate cancer. Future Oncol. 8, 1239–1251 (2012).
Kumano, M. et al. Cotargeting stress-activated Hsp27 and autophagy as a combinatorial strategy to amplify endoplasmic reticular stress in prostate cancer. Mol. Cancer Ther. 11, 1661–1671 (2012).
Hotte, S. J. et al. Phase I trial of OGX-427, a 2′methoxyethyl antisense oligonucleotide (ASO), against heat shock protein 27 (Hsp27): Final results [abstract 3077]. J. Clin. Oncol. 28 (Suppl.) (2010).
Chi, K. et al. A randomized phase II study of OGX-427 plus prednisone (P) versus P alone in patients (pts) with metastatic castration resistant prostate cancer (CRPC) [abstract 4514]. J. Clin. Oncol. 30 (Suppl.) (2012).
Loison, F. et al. Up-regulation of the clusterin gene after proteotoxic stress: implication of HSF1-HSF2 heterocomplexes. Biochem. J. 395, 223–231 (2006).
Cochrane, D. R., Wang, Z., Muramaki, M., Gleave, M. E. & Nelson, C. C. Differential regulation of clusterin and its isoforms by androgens in prostate cells. J. Biol. Chem. 282, 2278–2287 (2007).
Shiota, M. et al. Clusterin is a critical downstream mediator of stress-induced YB-1 transactivation in prostate cancer. Mol. Cancer Res. 9, 1755–1766 (2011).
Matsumoto, H. et al. Cotargeting androgen receptor and clusterin delays castrate-resistant prostate cancer progression by inhibiting adaptive stress response and AR stability. Cancer Res. 73, 5206–5217 (2013).
Montpetit, M. L., Lawless, K. R. & Tenniswood, M. Androgen-repressed messages in the rat ventral prostate. Prostate 8, 25–36 (1986).
Miyake, H., Nelson, C., Rennie, P. S. & Gleave, M. E. Testosterone-repressed prostate message-2 is an antiapoptotic gene involved in progression to androgen independence in prostate cancer. Cancer Res. 60, 170–176 (2000).
Zhang, H. et al. Clusterin inhibits apoptosis by interacting with activated Bax. Nat. Cell Biol. 7, 909–915 (2005).
Ammar, H. & Closset, J. L. Clusterin activates survival through the phosphatidylinositol 3-kinase/Akt pathway. J. Biol. Chem. 283, 12851–12861 (2008).
Zhong, B. et al. Induction of clusterin by AKT--role in cytoprotection against docetaxel in prostate tumor cells. Mol. Cancer Ther. 9, 1831–1841 (2010).
Li, N., Zoubeidi, A., Beraldi, E. & Gleave, M. E. GRP78 regulates clusterin stability, retrotranslocation and mitochondrial localization under ER stress in prostate cancer. Oncogene 32, 1933–1942 (2013).
Shiota, M. et al. Clusterin mediates TGF-beta-induced epithelial-mesenchymal transition and metastasis via Twist1 in prostate cancer cells. Cancer Res. 72, 5261–5272 (2012).
Lenferink, A. E. et al. Transcriptome profiling of a TGF-beta-induced epithelial-to-mesenchymal transition reveals extracellular clusterin as a target for therapeutic antibodies. Oncogene 29, 831–844 (2010).
Lee, K. B. et al. Clusterin, a novel modulator of TGF-beta signaling, is involved in Smad2/3 stability. Biochem. Biophys. Res. Commun. 366, 905–909 (2008).
Chou, T. Y. et al. Clusterin silencing in human lung adenocarcinoma cells induces a mesenchymal-to-epithelial transition through modulating the ERK/Slug pathway. Cell. Signal. 21, 704–711 (2009).
Bhutia, S. K. et al. mda-7/IL-24 differentially regulates soluble and nuclear clusterin in prostate cancer. J. Cell. Physiol. 227, 1805–1813 (2012).
Song, H. et al. Loss of Nkx3.1 leads to the activation of discrete downstream target genes during prostate tumorigenesis. Oncogene 28, 3307–3319 (2009).
Steinberg, J. et al. Intracellular levels of SGP-2 (Clusterin) correlate with tumor grade in prostate cancer. Clin. Cancer Res. 3, 1707–1711 (1997).
July, L. V. et al. Clusterin expression is significantly enhanced in prostate cancer cells following androgen withdrawal therapy. Prostate 50, 179–188 (2002).
Miyake, H. et al. Enhanced expression of the secreted form of clusterin following neoadjuvant hormonal therapy as a prognostic predictor in patients undergoing radical prostatectomy for prostate cancer. Oncol. Rep. 14, 1371–1375 (2005).
Girard, F. P. et al. Detecting soluble clusterin in in-vitro and in-vivo models of prostate cancer. Neoplasma 57, 488–493 (2010).
Gleave, M. E. et al. Use of antisense oligonucleotides targeting the antiapoptotic gene, clusterin/testosterone-repressed prostate message 2, to enhance androgen sensitivity and chemosensitivity in prostate cancer. Urology 58, 39–49 (2001).
Miyake, H., Chi, K. N. & Gleave, M. E. Antisense TRPM-2 oligodeoxynucleotides chemosensitize human androgen-independent PC-3 prostate cancer cells both in vitro and in vivo. Clin. Cancer Res. 6, 1655–1663 (2000).
Miyake, H., Nelson, C., Rennie, P. S. & Gleave, M. E. Acquisition of chemoresistant phenotype by overexpression of the antiapoptotic gene testosterone-repressed prostate message-2 in prostate cancer xenograft models. Cancer Res. 60, 2547–2554 (2000).
Sowery, R. D. et al. Clusterin knockdown using the antisense oligonucleotide OGX-011 re-sensitizes docetaxel-refractory prostate cancer PC-3 cells to chemotherapy. BJU Int. 102, 389–397 (2008).
Trougakos, I. P., So, A., Jansen, B., Gleave, M. E. & Gonos, E. S. Silencing expression of the clusterin/apolipoprotein j gene in human cancer cells using small interfering RNA induces spontaneous apoptosis, reduced growth ability, and cell sensitization to genotoxic and oxidative stress. Cancer Res. 64, 1834–1842 (2004).
Zellweger, T. et al. Enhanced radiation sensitivity in prostate cancer by inhibition of the cell survival protein clusterin. Clin. Cancer Res. 8, 3276–3284 (2002).
Chi, K. N. et al. A phase I pharmacokinetic and pharmacodynamic study of OGX-011, a 2′-methoxyethyl antisense oligonucleotide to clusterin, in patients with localized prostate cancer. J. Natl Cancer Inst. 97, 1287–1296 (2005).
Chi, K. N. et al. A phase I study of OGX-011, a 2′-methoxyethyl phosphorothioate antisense to clusterin, in combination with docetaxel in patients with advanced cancer. Clin. Cancer Res. 14, 833–839 (2008).
Chi, K. N. et al. Randomized phase II study of docetaxel and prednisone with or without OGX-011 in patients with metastatic castration-resistant prostate cancer. J. Clin. Oncol. 28, 4247–4254 (2010).
Teva and OncoGenex announce top-line survival results of phase III SYNERGY trial evaluating custirsen in combination with first-line docetaxel and prednisone for metastatic castrate-resistant prostate cancer. Teva Pharmaceutical Industries Ltd [online], (2014).
Saad, F. et al. Randomized phase II trial of Custirsen (OGX-011) in combination with docetaxel or mitoxantrone as second-line therapy in patients with metastatic castrate-resistant prostate cancer progressing after first-line docetaxel: CUOG trial P-06c. Clin. Cancer Res. 17, 5765–5773 (2011).
Kole, R., Krainer, A. R. & Altman, S. RNA therapeutics: beyond RNA interference and antisense oligonucleotides. Nat. Rev. Drug Discov. 11, 125–140 (2012).
Stephanou, A. & Latchman, D. S. Transcriptional modulation of heat-shock protein gene expression. Biochem. Res. Int. 2011, 238601 (2011).
Morimoto, R. I. & Santoro, M. G. Stress-inducible responses and heat shock proteins: new pharmacologic targets for cytoprotection. Nat. Biotechnol. 16, 833–838 (1998).
Tsan, M. F. & Gao, B. Heat shock protein and innate immunity. Cell. Mol. Immunol. 1, 274–279 (2004).
Lanneau, D., de Thonel, A., Maurel, S., Didelot, C. & Garrido, C. Apoptosis versus cell differentiation: role of heat shock proteins HSP90, HSP70 and HSP27. Prion 1, 53–60 (2007).
Author information
Authors and Affiliations
Contributions
A.A.A., A.Z. and K.N.C. researched data for the article. All authors made a substantial contribution to discussion of content. A.A.A. and A.Z. wrote the article. All authors contributed to review and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
A.A.A. declares honoraria from Janssen and receipt of research funding from Astellas. M.E.G. is listed as inventor on patent applications OGX 011 and OGX 427, submitted by the University of British Columbia, and licensed to OncoGenex Technologies, in which M.E.G. is a founding shareholder. K.N.C. declares receipt of research funding from Astellas, Janssen, Novartis and OncoGenex Technologies, and consultancy for Amgen, Astellas, Bayer, Janssen, Millennium, Novartis and Sanofi. A.Z. declares no competing interests.
Rights and permissions
About this article
Cite this article
Azad, A., Zoubeidi, A., Gleave, M. et al. Targeting heat shock proteins in metastatic castration-resistant prostate cancer. Nat Rev Urol 12, 26–36 (2015). https://doi.org/10.1038/nrurol.2014.320
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2014.320
This article is cited by
-
AR antagonists develop drug resistance through TOMM20 autophagic degradation-promoted transformation to neuroendocrine prostate cancer
Journal of Experimental & Clinical Cancer Research (2023)
-
Clinical implications of heat shock protein 70 in oral carcinogenesis and prediction of progression and recurrence in oral squamous cell carcinoma patients: a retrospective clinicopathological study
European Journal of Medical Research (2023)
-
Nanoparticle-mediated TRPV1 channel blockade amplifies cancer thermo-immunotherapy via heat shock factor 1 modulation
Nature Communications (2023)
-
The intersection of COVID-19 and cancer: signaling pathways and treatment implications
Molecular Cancer (2021)
-
ARVib suppresses growth of advanced prostate cancer via inhibition of androgen receptor signaling
Oncogene (2021)