Abstract
Purpose
Endometriosis, a gynecological disease, is difficult to be cured. Currently, to identify more potential biomarkers for the early diagnosis of endometriosis is urgently needed. Insulin like growth factor 2 (IGF2) has been revealed to correlate with endometriosis. This research aimed to further explore the role of IGF2 and its up-stream mechanism in endometriosis.
Methods
Primary ectopic endometrial stromal cells (EESCs) were extracted from ectopic endometrial tissues which were pathological endometrial tissues resected from three patients with II-III endometriosis. Primary normal endometrial stromal cells (NESCs) were extracted from normal endometrial tissues of two patients with grade III cervical dysplasia and one patient with uterine leiomyoma III. Four endometriotic cell lines (EEC145T, hEM15A, hEM5B2, and 12Z) and normal human endometrial epithelial cells (hEECs) were purchased. Cell proliferation, migration, and invasion were evaluated through functional assays. The molecular interaction between RNAs was investigated through mechanistic analyses.
Results
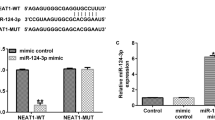
We discovered that IGF2 was upregulated in purchased endometriotic cells and primary EESC. Suppression of IGF2 hampered cell proliferation, migration, and invasion. Furthermore, insulin-like growth factor 2 antisense RNA (IGF2-AS) was uncovered to positively regulate IGF2 expression and enhanced proliferative, migratory, and invasive abilities of endometriotic cells. Mechanistically, miR-370-3p was found to bind with IGF2-AS and IGF2. IGF2-AS competitively bind with miR-370-3p to upregulate IGF2. Furthermore, IGF2-AS was revealed to activate the PI3K/AKT/mTOR signaling pathway through targeting miR-370-3p/IGF2 axis.
Conclusion
IGF2-AS promotes endometriotic cell growth via regulating IGF2/miR-370-3p axis and further activating PI3K/AKT/mTOR signaling pathway.




Similar content being viewed by others
Data availability
Research data are not shared.
Abbreviations
- lncRNA:
-
Long non-coding RNA
- IGF2-AS:
-
Insulin like growth factor 2 antisense RNA
- ceRNA:
-
Competing endogenous RNA
- miRNA:
-
MicroRNA
- IGF2:
-
Insulin like growth factor 2
- qRT-PCR:
-
Quantitative real-time PCR
- CCK-8:
-
Cell counting kit-8
- EdU:
-
5-Ethynyl-2′-deoxyuridine
- RIP:
-
RNA immunoprecipitation
- shRNA:
-
Short hairpin RNA
- FBS:
-
Fetal bovine serum
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- PVDF:
-
Polyvinylidene fluoride
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- SD:
-
Standard deviation
- NC:
-
Negative control
- ANOVA:
-
Analysis of variance
- ATCC:
-
American Type Culture Collection
- OD:
-
Optical density
References
Zondervan KT, Becker CM, Koga K, Missmer SA, Taylor RN, Viganò P. Endometriosis. Nat Rev Dis Primers. 2018;4(1):9. https://doi.org/10.1038/s41572-018-0008-5.
Laganà AS, Salmeri FM, Ban Frangež H, Ghezzi F, Vrtačnik-Bokal E, Granese R. Evaluation of M1 and M2 macrophages in ovarian endometriomas from women affected by endometriosis at different stages of the disease. Gynecol Endocrinol. 2020;36(5):441–4. https://doi.org/10.1080/09513590.2019.1683821.
Bulun SE, Yilmaz BD, Sison C, Miyazaki K, Bernardi L, Liu S, et al. Endometriosis. Endocr Rev. 2019;40(4):1048–79. https://doi.org/10.1210/er.2018-00242.
Laganà AS, Salmeri FM, Vitale SG, Triolo O, Götte M. Stem cell trafficking during endometriosis: may epigenetics play a pivotal role? Reproductive sciences (Thousand Oaks, Calif). 2018;25(7):978–9. https://doi.org/10.1177/1933719116687661.
Giudice LC, Kao LC. Endometriosis. Lancet (London, England). 2004;364(9447):1789–99. https://doi.org/10.1016/s0140-6736(04)17403-5.
Benagiano G, Guo SW, Puttemans P, Gordts S, Brosens I. Progress in the diagnosis and management of adolescent endometriosis: an opinion. Reprod Biomed Online. 2018;36(1):102–14. https://doi.org/10.1016/j.rbmo.2017.09.015.
Vercellini P, Vigano P, Somigliana E, Fedele L. Endometriosis: pathogenesis and treatment. Nat Rev Endocrinol. 2014;10(5):261–75. https://doi.org/10.1038/nrendo.2013.255.
Czyzyk A, Podfigurna A, Szeliga A, Meczekalski B. Update on endometriosis pathogenesis. Minerva Ginecol. 2017;69(5):447–61. https://doi.org/10.23736/s0026-4784.17.04048-5.
Kobayashi H. Imprinting genes associated with endometriosis. EXCLI J. 2014;13:252–64.
Kim H, Park JH, Ku SY, Kim SH, Choi YM, Kim JG. Association between endometriosis and polymorphisms in insulin-like growth factors (IGFs) and IGF-I receptor genes in Korean women. Eur J Obstet Gynecol Reprod Biol. 2011;156(1):87–90. https://doi.org/10.1016/j.ejogrb.2010.12.018.
Wang J, Su Z, Lu S, Fu W, Liu Z, Jiang X, et al. LncRNA HOXA-AS2 and its molecular mechanisms in human cancer. Clin Chim Acta. 2018;485:229–33. https://doi.org/10.1016/j.cca.2018.07.004.
Huang Y. The novel regulatory role of lncRNA-miRNA-mRNA axis in cardiovascular diseases. J Cell Mol Med. 2018;22(12):5768–75. https://doi.org/10.1111/jcmm.13866.
Xing C, Sun SG, Yue ZQ, Bai F. Role of lncRNA LUCAT1 in cancer. Biomed Pharmacother. 2021;134:111158. https://doi.org/10.1016/j.biopha.2020.111158.
Wang L, Xing Q, Feng T, He M, Yu W, Chen H. SNP rs710886 A>G in long noncoding RNA PCAT1 is associated with the risk of endometriosis by modulating expression of multiple stemness-related genes via microRNA-145 signaling pathway. J Cell Biochem. 2019. https://doi.org/10.1002/jcb.29406.
Yu J, Chen LH, Zhang B, Zheng QM. The modulation of endometriosis by lncRNA MALAT1 via NF-kappaB/iNOS. Eur Rev Med Pharmacol Sci. 2019;23(10):4073–80. https://doi.org/10.26355/eurrev_201905_17908.
Zhang C, Wu W, Ye X, Ma R, Luo J, Zhu H, et al. Aberrant expression of CHL1 gene and long non-coding RNA CHL1-AS1, CHL1-AS2 in ovarian endometriosis. Eur J Obstet Gynecol Reprod Biol. 2019;236:177–82. https://doi.org/10.1016/j.ejogrb.2019.03.020.
Sha L, Huang L, Luo X, Bao J, Gao L, Pan Q, et al. Long non-coding RNA LINC00261 inhibits cell growth and migration in endometriosis. J Obstet Gynaecol Res. 2017;43(10):1563–9. https://doi.org/10.1111/jog.13427.
Liu J, Wang Y, Chen P, Ma Y, Wang S, Tian Y et al. AC002454.1 and CDK6 synergistically promote endometrial cell migration and invasion in endometriosis. Reproduction (Cambridge, England). 2019;157(6):535–43. https://doi.org/10.1530/rep-19-0005.
Qiu JJ, Lin XJ, Zheng TT, Tang XY, Zhang Y, Hua KQ. The exosomal long noncoding RNA aHIF is upregulated in serum from patients with endometriosis and promotes angiogenesis in endometriosis. Reproductive sciences (Thousand Oaks, Calif). 2019;26(12):1590–1602. https://doi.org/10.1177/1933719119831775.
Xu Z, Zhang L, Yu Q, Zhang Y, Yan L, Chen ZJ. The estrogen-regulated lncRNA H19/miR-216a-5p axis alters stromal cell invasion and migration via ACTA2 in endometriosis. Mol Hum Reprod. 2019;25(9):550–61. https://doi.org/10.1093/molehr/gaz040.
Shi Y, Zha J, Zuo M, Yan Q, Song H. Long noncoding RNA CHL1-AS1 promotes cell proliferation and migration by sponging miR-6076 to regulate CHL1 expression in endometrial cancer. J Cell Biochem. 2019. https://doi.org/10.1002/jcb.29486.
Wang Y, Huang T, Sun X, Wang Y. Identification of a potential prognostic lncRNA-miRNA-mRNA signature in endometrial cancer based on the competing endogenous RNA network. J Cell Biochem. 2019;120(11):18845–53. https://doi.org/10.1002/jcb.29200.
Zhou CF, Liu MJ, Wang W, Wu S, Huang YX, Chen GB, et al. miR-205-5p inhibits human endometriosis progression by targeting ANGPT2 in endometrial stromal cells. Stem Cell Res Ther. 2019;10(1):287. https://doi.org/10.1186/s13287-019-1388-5.
Wu W, Gao H, Li X, Zhu Y, Peng S, Yu J, et al. LncRNA TPT1-AS1 promotes tumorigenesis and metastasis in epithelial ovarian cancer by inducing TPT1 expression. Cancer Sci. 2019;110(5):1587–98. https://doi.org/10.1111/cas.14009.
Long J, Bai Y, Yang X, Lin J, Yang X, Wang D, et al. Construction and comprehensive analysis of a ceRNA network to reveal potential prognostic biomarkers for hepatocellular carcinoma. Cancer Cell Int. 2019;19:90. https://doi.org/10.1186/s12935-019-0817-y.
Li JH, Liu S, Zhou H, Qu LH, Yang JH. starBase v2.0: decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA interaction networks from large-scale CLIP-Seq data. Nucleic Acids Res. 2014;42(Database issue):D92-7. https://doi.org/10.1093/nar/gkt1248.
Tian B, Zhao Y, Liang T, Ye X, Li Z, Yan D, et al. Curcumin inhibits urothelial tumor development by suppressing IGF2 and IGF2-mediated PI3K/AKT/mTOR signaling pathway. J Drug Target. 2017;25(7):626–36. https://doi.org/10.1080/1061186x.2017.1306535.
Xu X, Yu Y, Zong K, Lv P, Gu Y. Up-regulation of IGF2BP2 by multiple mechanisms in pancreatic cancer promotes cancer proliferation by activating the PI3K/Akt signaling pathway. J Exp Clin Cancer Res. 2019;38(1):497. https://doi.org/10.1186/s13046-019-1470-y.
Mu Q, Wang L, Yu F, Gao H, Lei T, Li P, et al. Imp2 regulates GBM progression by activating IGF2/PI3K/Akt pathway. Cancer Biol Ther. 2015;16(4):623–33. https://doi.org/10.1080/15384047.2015.1019185.
Wang Y, Kuramitsu Y, Baron B, Kitagawa T, Tokuda K, Akada J, et al. PI3K inhibitor LY294002, as opposed to wortmannin, enhances AKT phosphorylation in gemcitabine-resistant pancreatic cancer cells. Int J Oncol. 2017;50(2):606–12. https://doi.org/10.3892/ijo.2016.3804.
Angioni S. New insights on endometriosis. Minerva Ginecol. 2017;69(5):438–9. https://doi.org/10.23736/s0026-4784.17.04089-8.
Borghese B, Santulli P, Marcellin L, Chapron C. Definition, description, clinicopathological features, pathogenesis and natural history of endometriosis: CNGOF-HAS Endometriosis Guidelines. Gynecologie, Obstetrique, Fertilite & Senologie. 2018;46(3):156–67. https://doi.org/10.1016/j.gofs.2018.02.017.
Imesch P, Fink D. Endometriosis Update 2016. Praxis. 2016;105(5):253–7. https://doi.org/10.1024/1661-8157/a002295.
Chen ZH, Hu HK, Zhang CR, Lu CY, Bao Y, Cai Z, et al. Down-regulation of long non-coding RNA FOXD3 antisense RNA 1 (FOXD3-AS1) inhibits cell proliferation, migration, and invasion in malignant glioma cells. Am J Transl Res. 2016;8(10):4106–19.
Song C, Song C, Chen K, Zhang X. Inhibition of long non-coding RNA IGF2AS protects apoptosis and neuronal loss in anesthetic-damaged mouse neural stem cell derived neurons. Biomed Pharmacother. 2017;85:218–24. https://doi.org/10.1016/j.biopha.2016.10.094.
Hu Z, Mamillapalli R, Taylor HS. Increased circulating miR-370-3p regulates steroidogenic factor 1 in endometriosis. Am J Physiol Endocrinol Metab. 2019;316(3):E373–82. https://doi.org/10.1152/ajpendo.00244.2018.
Hagstrom AD, Denham J. microRNAs in high and low responders to resistance training in breast cancer survivors. Int J Sports Med. 2018;39(6):482–9. https://doi.org/10.1055/a-0592-7691.
Zhao JR, Cheng WW, Wang YX, Cai M, Wu WB, Zhang HJ. Identification of microRNA signature in the progression of gestational trophoblastic disease. Cell Death Dis. 2018;9(2):94. https://doi.org/10.1038/s41419-017-0108-2.
Li J, Huang Y, Deng X, Luo M, Wang X, Hu H, et al. Long noncoding RNA H19 promotes transforming growth factor-beta-induced epithelial-mesenchymal transition by acting as a competing endogenous RNA of miR-370-3p in ovarian cancer cells. Onco Targets Ther. 2018;11:427–40. https://doi.org/10.2147/ott.S149908.
Liu S, Qiu J, Tang X, Cui H, Zhang Q, Yang Q. LncRNA-H19 regulates cell proliferation and invasion of ectopic endometrium by targeting ITGB3 via modulating miR-124-3p. Exp Cell Res. 2019;381(2):215–22. https://doi.org/10.1016/j.yexcr.2019.05.010.
Liu Z, Liu L, Zhong Y, Cai M, Gao J, Tan C, et al. LncRNA H19 over-expression inhibited Th17 cell differentiation to relieve endometriosis through miR-342-3p/IER3 pathway. Cell Biosci. 2019;9:84. https://doi.org/10.1186/s13578-019-0346-3.
Acknowledgements
We appreciate the support of all lab members.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All clinical samples were acquired from the First People’s Hospital of Wenling following the ethical and legal standards. All patients have signed a statement of consent.
Consent for publication
The paper has been proofread by all authors enrolled in this study, and they have permitted the submission and publication of this paper.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jin, X., Feng, J. & Cheng, X. LncRNA IGF2-AS promotes endometriosis progression through targeting miR-370-3p/IGF2 axis and activating PI3K/AKT/mTOR signaling pathway. J Assist Reprod Genet 39, 2699–2710 (2022). https://doi.org/10.1007/s10815-022-02638-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-022-02638-2