Abstract
Purpose
To analyze the efficacy and tolerability of cabozantinib—a small molecule inhibitor of MET and VEGFR2—alone or with trastuzumab in patients with breast cancer brain metastases (BCBM).
Methods
This single-arm phase II study enrolled patients with new or progressive measurable BCBM into 3 cohorts: Cohort 1 (HER2-positive), Cohort 2 (hormone receptor-positive/HER2-negative), and Cohort 3 (triple-negative). Patients received cabozantinib 60-mg daily on a 21-day cycle. Cohort 1 added trastuzumab every 3 weeks and had a primary objective of central nervous system (CNS) objective response rate (ORR) by RECIST 1.1. Secondary objectives for all cohorts were progression-free survival, overall survival, toxicity, and changes in vascular parameters and circulating biomarkers. Cohorts 2 and 3 also had CNS ORR as a secondary objective.
Results
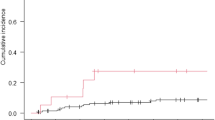
Thirty-six BCBM patients enrolled (cohort 1, n = 21; cohort 2, n = 7; cohort 3, n = 8), with a median age of 50. Patients had a median of 3 prior lines for metastatic disease (range 1–9). Treatments prior to enrollment included craniotomy (n = 4), whole brain radiation (n = 24) and stereotactic radiosurgery (n = 11). CNS ORR was 5% in cohort 1, 14% in cohort 2, and 0% in cohort 3. Most common grade 3/4 adverse events included elevations in lipase (11%), AST (8%), ALT (6%), hyponatremia (8%), and hypertension (6%). Cabozantinib increased plasma concentrations of CA-IX, soluble (s)MET, PlGF, sTIE-2, VEGF, and VEGF-D, and decreased sVEGFR2 and TNF-α and total tumor blood volume.
Conclusions
Cabozantinib had insufficient activity in heavily pretreated BCBM patients. Biomarker analysis showed that cabozantinib had antiangiogenic activity and increased tissue hypoxia.
Trial registration
Clinicaltrial.gov registration: NCT02260531.

Similar content being viewed by others
Data availability
The datasets during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Lin NU, Bellon JR, Winer EP (2004) CNS metastases in breast cancer. J Clin Oncol 22:3608–3617. https://doi.org/10.1200/JCO.2004.01.175
Aversa C, Rossi V, Geuna E, Martinello R, Milani A, Redana S, Valabrega G, Aglietta M, Montemurro F (2014) Metastatic breast cancer subtypes and central nervous system metastases. Breast 23:623–628. https://doi.org/10.1016/j.breast.2014.06.009
Klos KJ, O’Neill BP (2004) Brain metastases. Neurologist 10:31–46. https://doi.org/10.1097/01.nrl.0000106922.83090.71
Yakes FM, Chen J, Tan J, Yamaguchi K, Shi Y, Yu P, Qian F, Chu F, Bentzien F, Cancilla B, Orf J, You A, Laird AD, Engst S, Lee L, Lesch J, Chou YC, Joly AH (2011) Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol Cancer Ther 10:2298–2308. https://doi.org/10.1158/1535-7163.MCT-11-0264
Choueiri TK, Escudier B, Powles T, Mainwaring PN, Rini BI, Donskov F, Hammers H, Hutson TE, Lee JL, Peltola K, Roth BJ, Bjarnason GA, Geczi L, Keam B, Maroto P, Heng DY, Schmidinger M, Kantoff PW, Borgman-Hagey A, Hessel C, Scheffold C, Schwab GM, Tannir NM, Motzer RJ (2015) Cabozantinib versus everolimus in advanced renal-cell carcinoma. N Engl J Med 373:1814–1823. https://doi.org/10.1056/NEJMoa1510016
Elisei R, Schlumberger MJ, Muller SP, Schoffski P, Brose MS, Shah MH, Licitra L, Jarzab B, Medvedev V, Kreissl MC, Niederle B, Cohen EE, Wirth LJ, Ali H, Hessel C, Yaron Y, Ball D, Nelkin B, Sherman SI (2013) Cabozantinib in progressive medullary thyroid cancer. J Clin Oncol 31:3639–3646. https://doi.org/10.1200/JCO.2012.48.4659
Abou-Alfa GK, Meyer T, Cheng AL, El-Khoueiry AB, Rimassa L, Ryoo BY, Cicin I, Merle P, Chen Y, Park JW, Blanc JF, Bolondi L, Klumpen HJ, Chan SL, Zagonel V, Pressiani T, Ryu MH, Venook AP, Hessel C, Borgman-Hagey AE, Schwab G, Kelley RK (2018) Cabozantinib in patients with advanced and progressing hepatocellular carcinoma. N Engl J Med 379:54–63. https://doi.org/10.1056/NEJMoa1717002
Tolaney SM, Nechushtan H, Ron IG, Schoffski P, Awada A, Yasenchak CA, Laird AD, O’Keeffe B, Shapiro GI, Winer EP (2016) Cabozantinib for metastatic breast carcinoma: results of a phase II placebo-controlled randomized discontinuation study. Breast Cancer Res Treat 160:305–312. https://doi.org/10.1007/s10549-016-4001-y
Klempner SJ, Borghei A, Hakimian B, Ali SM, Ou SI (2017) Intracranial Activity of Cabozantinib in MET Exon 14-Positive NSCLC with Brain Metastases. J Thorac Oncol 12:152–156. https://doi.org/10.1016/j.jtho.2016.09.127
Negrier S, Moriceau G, Attignon V, Haddad V, Pissaloux D, Guerin N, Carrie C (2018) Activity of cabozantinib in radioresistant brain metastases from renal cell carcinoma: two case reports. J Med Case Rep 12:351. https://doi.org/10.1186/s13256-018-1875-9
Wen PY, Drappatz J, de Groot J, Prados MD, Reardon DA, Schiff D, Chamberlain M, Mikkelsen T, Desjardins A, Holland J, Ping J, Weitzman R, Cloughesy TF (2018) Phase II study of cabozantinib in patients with progressive glioblastoma: subset analysis of patients naive to antiangiogenic therapy. Neuro Oncol 20:249–258. https://doi.org/10.1093/neuonc/nox154
Eichler AF, Chung E, Kodack DP, Loeffler JS, Fukumura D, Jain RK (2011) The biology of brain metastases-translation to new therapies. Nat Rev Clin Oncol 8:344–356. https://doi.org/10.1038/nrclinonc.2011.58
Kodack DP, Chung E, Yamashita H, Incio J, Duyverman AM, Song Y, Farrar CT, Huang Y, Ager E, Kamoun W, Goel S, Snuderl M, Lussiez A, Hiddingh L, Mahmood S, Tannous BA, Eichler AF, Fukumura D, Engelman JA, Jain RK (2012) Combined targeting of HER2 and VEGFR2 for effective treatment of HER2-amplified breast cancer brain metastases. Proc Natl Acad Sci USA 109:E3119–3127. https://doi.org/10.1073/pnas.1216078109
Ebos JM, Lee CR, Cruz-Munoz W, Bjarnason GA, Christensen JG, Kerbel RS (2009) Accelerated metastasis after short-term treatment with a potent inhibitor of tumor angiogenesis. Cancer Cell 15:232–239. https://doi.org/10.1016/j.ccr.2009.01.021
Lu-Emerson C, Duda DG, Emblem KE, Taylor JW, Gerstner ER, Loeffler JS, Batchelor TT, Jain RK (2015) Lessons from anti-vascular endothelial growth factor and anti-vascular endothelial growth factor receptor trials in patients with glioblastoma. J Clin Oncol 33:1197–1213. https://doi.org/10.1200/JCO.2014.55.9575
Paez-Ribes M, Allen E, Hudock J, Takeda T, Okuyama H, Vinals F, Inoue M, Bergers G, Hanahan D, Casanovas O (2009) Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell 15:220–231. https://doi.org/10.1016/j.ccr.2009.01.027
Shojaei F, Lee JH, Simmons BH, Wong A, Esparza CO, Plumlee PA, Feng J, Stewart AE, Hu-Lowe DD, Christensen JG (2010) HGF/c-Met acts as an alternative angiogenic pathway in sunitinib-resistant tumors. Cancer Res 70:10090–10100. https://doi.org/10.1158/0008-5472.CAN-10-0489
di Tomaso E, Snuderl M, Kamoun WS, Duda DG, Auluck PK, Fazlollahi L, Andronesi OC, Frosch MP, Wen PY, Plotkin SR, Hedley-Whyte ET, Sorensen AG, Batchelor TT, Jain RK (2011) Glioblastoma recurrence after cediranib therapy in patients: lack of “rebound” revascularization as mode of escape. Cancer Res 71:19–28. https://doi.org/10.1158/0008-5472.CAN-10-2602
Bjornerud A, Emblem KE (2010) A fully automated method for quantitative cerebral hemodynamic analysis using DSC-MRI. J Cereb Blood Flow Metab 30:1066–1078. https://doi.org/10.1038/jcbfm.2010.4
Intlekofer AM, Thompson CB (2013) At the Bench: preclinical rationale for CTLA-4 and PD-1 blockade as cancer immunotherapy. J Leukoc Biol 94:25–39. https://doi.org/10.1189/jlb.1212621
Simon R (1989) Optimal two-stage designs for phase II clinical trials. Control Clin Trials 10:1–10
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Series B 57:289–300
Lu YS, Chen TW, Lin CH, Yeh DC, Tseng LM, Wu PF, Rau KM, Chen BB, Chao TC, Huang SM, Huang CS, Shih TT, Cheng AL (2015) Bevacizumab preconditioning followed by Etoposide and Cisplatin is highly effective in treating brain metastases of breast cancer progressing from whole-brain radiotherapy. Clin Cancer Res 21:1851–1858. https://doi.org/10.1158/1078-0432.CCR-14-2075
Lin NU, Gelman RS, Younger WJ, Sohl J, Freedman RA, Sorensen AG, Bullitt E, Harris GJ, Morganstern D, Schneider BP (2013) Phase II trial of carboplatin (C) and bevacizumab (BEV) in patients (pts) with breast cancer brain metastases (BCBM). J Clin Oncol 31(15_Suppl):513–513. https://doi.org/10.1200/jco.2013.31.15_suppl.513
Schoffski P, Gordon M, Smith DC, Kurzrock R, Daud A, Vogelzang NJ, Lee Y, Scheffold C, Shapiro GI (2017) Phase II randomised discontinuation trial of cabozantinib in patients with advanced solid tumours. Eur J Cancer 86:296–304. https://doi.org/10.1016/j.ejca.2017.09.011
Tolaney SM, Ziehr DR, Guo H, Ng MR, Barry WT, Higgins MJ, Isakoff SJ, Brock JE, Ivanova EV, Paweletz CP, Demeo MK, Ramaiya NH, Overmoyer BA, Jain RK, Winer EP, Duda DG (2017) Phase II and biomarker study of cabozantinib in metastatic triple-negative breast cancer patients. Oncologist 22:25–32. https://doi.org/10.1634/theoncologist.2016-0229
Jain RK, Duda DG, Willett CG, Sahani DV, Zhu AX, Loeffler JS, Batchelor TT, Sorensen AG (2009) Biomarkers of response and resistance to antiangiogenic therapy. Nat Rev Clin Oncol 6:327–338. https://doi.org/10.1038/nrclinonc.2009.63
Zhu AX, Ancukiewicz M, Supko JG, Sahani DV, Blaszkowsky LS, Meyerhardt JA, Abrams TA, McCleary NJ, Bhargava P, Muzikansky A, Sheehan S, Regan E, Vasudev E, Knowles M, Fuchs CS, Ryan DP, Jain RK, Duda DG (2013) Efficacy, safety, pharmacokinetics, and biomarkers of cediranib monotherapy in advanced hepatocellular carcinoma: a phase II study. Clin Cancer Res 19:1557–1566. https://doi.org/10.1158/1078-0432.CCR-12-3041
Goyal L, Zheng H, Yurgelun MB, Abrams TA, Allen JN, Cleary JM, Knowles M, Regan E, Reardon A, Khachatryan A, Jain RK, Nardi V, Borger DR, Duda DG, Zhu AX (2017) A phase 2 and biomarker study of cabozantinib in patients with advanced cholangiocarcinoma. Cancer 123:1979–1988. https://doi.org/10.1002/cncr.30571
Kalpathy-Cramer J, Chandra V, Da X, Ou Y, Emblem KE, Muzikansky A, Cai X, Douw L, Evans JG, Dietrich J, Chi AS, Wen PY, Stufflebeam S, Rosen B, Duda DG, Jain RK, Batchelor TT, Gerstner ER (2017) Phase II study of tivozanib, an oral VEGFR inhibitor, in patients with recurrent glioblastoma. J Neurooncol 131:603–610. https://doi.org/10.1007/s11060-016-2332-5
Funding
Exelixis, Rosa’s Pursuit of Hope, and Global Giving Grant from Tripadvisor.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
JPL receives institutional research funding from Merck and Kazia Therapeutics. SMT receives institutional research funding from Novartis, Genentech, Eli Lilly, Pfizer, Merck, Exelixis, Eisai, Bristol Meyers Squibb, AstraZeneca, Cyclacel, Immunomedics, Odenate, and Nektar. SMT has served as an advisor/consultant to Novartis, Eli Lilly, Pfizer, Merck, AstraZeneca, Eisai, Puma, Genentech, Immunomedics, Nektar, Tesaro, and Nanostring. RKJ received honorarium from Amgen and consultant fees from Chugai, Merck, Ophthotech, SPARC, SynDevRx, and XTuit. RKJ owns equity in Enlight, SPARC, and SynDevRx, and serves on the Boards of Trustees of Tekla Healthcare Investors, Tekla Life Sciences Investors, Tekla Healthcare Opportunities Fund, and Tekla World Healthcare Fund. DGD received consultant fees from Bayer, Tilos, twoXAR, and BMS and has received research grants from Bayer, Merrimack, Leap, Exelixis, and BMS. WTB received institutional research funding from Pfizer. NUL receives institutional research funding from Genentech, Seattle Genetics, Novartis, and Pfizer. NUL has served as an advisor/consultant to Puma and Daiichi.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (include name of committee + reference number) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Leone, J.P., Duda, D.G., Hu, J. et al. A phase II study of cabozantinib alone or in combination with trastuzumab in breast cancer patients with brain metastases. Breast Cancer Res Treat 179, 113–123 (2020). https://doi.org/10.1007/s10549-019-05445-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-019-05445-z