Abstract
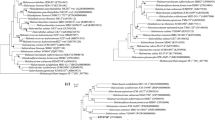
Four halophilic archaea strains, AD-4T, CGA30T, CGA73T, and WLHSJ27T, were isolated from a salt lake and two soda lakes located in different regions of China. The 16S rRNA and rpoB′ gene sequence similarities among strains AD-4T, CGA30T, CGA73T, WLHSJ27T, and the current species of the family Natrialbaceae were 90.9–97.5% and 83.1–91.8%, respectively. The phylogenetic and phylogenomic analyses revealed that these four strains separated from existing genera in the family Natrialbaceae and formed distant branches. The ANI, isDDH, and AAI values among these four strains and the current members of the family Natrialbaceae were 72–79%, 20–25%, and 63–73%, respectively, much lower than the threshold values for species demarcation. Strains AD-4T, CGA73T, and WLHSJ27T may represent three novel genera of the family Natrialbaceae according to the cutoff value of AAI (≤ 76%) proposed to differentiate genera within the family Natrialbaceae. These four strains could be distinguished from the related genera according to differential phenotypic characteristics. The major phospholipids of these four strains were identical while their glycolipid profiles were diverse. DGD-1 is a major glycolipid found in strain AD-4T, trace glycolipids, DGD-1, and S-DGD-1, and (or) S-TGD-1 was found in the other three strains. The major respiratory quinones detected in the four strains were menaquinone MK-8 and MK-8(H2). This polyphasic classification indicated that strains AD-4T, CGA73T, and WLHSJ27T represent three novel species of three new genera with the family Natrialbaceae, and strain CGA30T represents a novel species of Halovivax.


Similar content being viewed by others
Data availability
The sequences determined in this study were deposited and available in the NCBI GenBank.
References
Aziz RK, Bartels D, Best AA, DeJongh M, Disz T, Edwards RA, Formsma K, Gerdes S, Glass EM, Kubal M, Meyer F, Olsen GJ, Olson R, Osterman AL, Overbeek RA, McNeil LK, Paarmann D, Paczian T, Parrello B, Pusch GD, Reich C, Stevens R, Vassieva O, Vonstein V, Wilke A, Zagnitko O (2008) The RAST server: rapid annotations using subsystems technology. BMC Genom 9:75. https://doi.org/10.1186/1471-2164-9-75
Bao C-X, Li S-Y, Xin Y-J, Hou J, Cui H-L (2022) Natrinema halophilum sp. nov., Natrinema salinisoli sp. nov., Natrinema amylolyticum sp. nov. and Haloterrigena alkaliphila sp. nov., four extremely halophilic archaea isolated from salt mine, saline soil and salt lake. Int J Syst Evol Microbiol 72:005385. https://doi.org/10.1099/ijsem.0.005385
Cui H-L, Zhou P-J, Oren A, Liu S-J (2009) Intraspecific polymorphism of 16S rRNA genes in two halophilic archaeal genera, Haloarcula and Halomicrobium. Extremophiles 13:31–37. https://doi.org/10.1007/s00792-008-0194-2
Cui H-L, Gao X, Yang X, Xu X-W (2010) Halorussus rarus gen. nov., sp. nov., a new member of the family Halobacteriaceae isolated from a marine solar saltern. Extremophiles 14:493–499. https://doi.org/10.1007/s00792-010-0329-0
de la Haba RR, Minegishi H, Kamekura M, Shimane Y, Ventosa A (2021) Phylogenomics of Haloarchaea: the controversy of the genera Natrinema-Haloterrigena. Front Microbiol 12:740909. https://doi.org/10.3389/fmicb.2021.740909
Dussault HP (1955) An improved technique for staining red halophilic bacteria. J Bacteriol 70:484–485. https://doi.org/10.1128/jb.70.4.484-485.1955
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/BF01734359
Fitch WM (1971) Toward defining the course of evolution: minimum change for a specific tree topology. Syst Biol 20:406–416. https://doi.org/10.2307/2412116
Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P, Tiedje JM (2007) DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol 57:81–91. https://doi.org/10.1099/ijs.0.64483-0
Gupta RS, Naushad S, Baker S (2015) Phylogenomic analyses and molecular signatures for the class Halobacteria and its two major clades: a proposal for division of the class Halobacteria into an emended order Halobacteriales and two new orders, Haloferacales ord. nov. and Natrialbales ord. nov., containing the novel families Haloferacaceae fam. nov. and Natrialbaceae fam. nov. Int J Syst Evol Microbiol 65:1050–1069. https://doi.org/10.1099/ijs.0.070136-0
Kamekura M, Dyall-Smith ML (1995) Taxonomy of the family Halobacteriaceae and the description of two new genera Halorubrobacterium and Natrialba. J Gen Appl Microbiol 41:333–350. https://doi.org/10.2323/jgam.41.333
Kanehisa M, Goto S, Kawashima S, Okuno Y, Hattori M (2004) The KEGG resource for deciphering the genome. Nucleic Acids Res 32:D277–D280. https://doi.org/10.1093/nar/gkh063
Kim M, Oh HS, Park SC, Chun J (2014) Towards a taxonomic coherence between average nucleotide identity and 16S rRNA gene sequence similarity for species demarcation of prokaryotes. Int J Syst Evol Microbiol 64:346–351. https://doi.org/10.1099/ijs.0.059774-0
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Li S-Y, Xin Y-J, Bao C-X, Hou J, Cui H-L (2022) Haloprofundus salilacus sp. nov., Haloprofundus halobius sp. nov. and Haloprofundus salinisoli sp. nov.: three extremely halophilic archaea isolated from salt lake and saline soil. Extremophiles 26:6. https://doi.org/10.1007/s00792-021-01255-8
Liu B-B, Tang S-K, Zhang Y-G, Lu X-H, Li L, Cheng J, Zhang Y-M, Zhang L-L, Li W-J (2013) Halalkalicoccus paucihalophilus sp. nov., a halophilic archaeon from Lop Nur region in Xinjiang, northwest of China. Antonie Van Leeuwenhoek 103:1007–1014. https://doi.org/10.1007/s10482-013-9880-x
Lobasso S, Pérez-Davó A, Vitale R, Sánchez MM, Corcelli A (2015) Deciphering archaeal glycolipids of an extremely halophilic archaeon of the genus Halobellus by MALDI-TOF/MS. Chem Phys Lipids 186:1–8. https://doi.org/10.1016/j.chemphyslip.2014.11.002
Luo C, Rodriguez-R LM, Konstantinidis KT (2014) MyTaxa: an advanced taxonomic classifier for genomic and metagenomic sequences. Nucleic Acids Res 42:e73. https://doi.org/10.1093/nar/gku169
Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinform 14:60. https://doi.org/10.1186/1471-2105-14-60
Oren A (2008) Microbial life at high salt concentrations: phylogenetic and metabolic diversity. Saline Syst 4:2. https://doi.org/10.1186/1746-1448-4-2
Oren A, Ventosa A, Grant WD (1997) Proposed minimal standards for description of new taxa in the order Halobacteriales. Int J Syst Evol Microbiol 47:233–238. https://doi.org/10.1099/00207713-47-1-233
Richter M, Rosselló-Móra R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci USA 106:19126–19131. https://doi.org/10.1073/pnas.0906412106
Richter M, Rosselló-Móra R, Oliver Glöckner F, Peplies J (2016) JSpeciesWS: a web server for prokaryotic species circumscription based on pairwise genome comparison. Bioinformatics 32:929–931. https://doi.org/10.1093/bioinformatics/btv681
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Sorokin DY, Khijniak TV, Kostrikina NA, Elcheninov AG, Toshchakov SV, Bale NJ, Damsté JSS, Kublanov IV (2018) Natronobiforma cellulositropha gen. nov., sp. nov., a novel haloalkaliphilic member of the family Natrialbaceae (class Halobacteria) from hypersaline alkaline lakes. Syst Appl Microbiol 41:355–362. https://doi.org/10.1016/j.syapm.2018.04.002
Sun Y-P, Wang B-B, Zheng X-W, Wu Z-P, Hou J, Cui H-L (2022) Description of Halosolutus amylolyticus gen. nov., sp. nov., Halosolutus halophilus sp. nov. and Halosolutus gelatinilyticus sp. nov., and genome-based taxonomy of genera Natribaculum and Halovarius. Int J Syst Evol Microbiol 72:005598. https://doi.org/10.1099/ijsem.0.005598
Wainø M, Tindall BJ, Ingvorsen K (2000) Halorhabdus utahensis gen. nov., sp. nov., an aerobic, extremely halophilic member of the Archaea from Great Salt Lake, Utah. Int J Syst Evol Microbiol 50:183–190. https://doi.org/10.1099/00207713-50-1-183
Wang B-B, Sun Y-P, Wu Z-P, Zheng X-W, Hou J, Cui H-L (2022) Halorientalis salina sp. nov., Halorientalis marina sp. nov., Halorientalis litorea sp. nov.: three extremely halophilic archaea isolated from a salt lake and coarse sea salt. Extremophiles 26:26. https://doi.org/10.1007/s00792-022-01275-y
Xu L, Dong Z, Fang L, Luo Y, Wei Z, Guo H, Zhang G, Gu YQ, Coleman-Derr D, Xia Q, Wang Y (2019) OrthoVenn2: a web server for whole-genome comparison and annotation of orthologous clusters across multiple species. Nucleic Acids Res 47(W1):W52–W58. https://doi.org/10.1093/nar/gkz333
Xue Q, Zuo Z, Zhou H, Zhou J, Zhang S, Han J, Zhao D, Xiang H (2021) Salinadaptatus halalkaliphilus gen. nov., sp. nov., a haloalkaliphilic archaeon isolated from salt pond in Inner Mongolia autonomous region, China. Int J Syst Evol Microbiol 71:004584. https://doi.org/10.1099/ijsem.0.004584
Yin X-Q, Liu B-B, Chu X, Salam N, Li X, Yang Z-W, Zhang Y, Xiao M, Li W-J (2017) Saliphagus infecundisoli gen. nov., sp. nov., an extremely halophilic archaeon isolated from a saline soil. Int J Syst Evol Microbiol 67:4154–4160. https://doi.org/10.1099/ijsem.0.002270
Yoon SH, Ha SM, Kwon S, Lim J, Kim Y, Seo H, Chun J (2017) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67:1613–1617. https://doi.org/10.1099/ijsem.0.001755
Zheng X-W, Wu Z-P, Sun Y-P, Wang B-B, Hou J, Cui H-L (2022) Halorussus vallis sp. nov., Halorussus aquaticus sp. nov., Halorussus gelatinilyticus sp. nov., Halorussus limi sp. nov., Halorussus salilacus sp. nov., Halorussus salinisoli sp. nov.: six extremely halophilic archaea isolated from solar saltern, salt lake and saline soil. Extremophiles 26:32. https://doi.org/10.1007/s00792-022-01280-1
Acknowledgements
We are grateful to the graduated students (Qin Xu, Yang-Jie Zhao, and Cong-Qi Tao, Master’s degree), for their primary contribution to this study.
Funding
This work was financially supported by the National Natural Science Foundation of China (No. 32070003), and the National Science and Technology Fundamental Resources Investigation Program of China (No. 2019FY100700; No. 2021FY100900).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that no competing interests exist.
Ethical approval
The article does not contain any studies related to human participants or animals.
Additional information
Communicated by Oren.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, XX., Tan, S., Cheng, M. et al. Salinilacihabitans rarus gen. nov., sp. nov., Natrononativus amylolyticus gen. nov., sp. nov., Natronobeatus ordinarius gen. nov., sp. nov., and Halovivax gelatinilyticus sp. nov., halophilic archaea, isolated from a salt lake and soda lakes. Extremophiles 27, 15 (2023). https://doi.org/10.1007/s00792-023-01303-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00792-023-01303-5