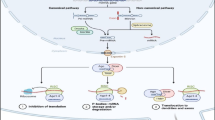
Abstract
Cell senescence genes play a vital role in the pathogenesis of colorectal cancer, a process that may involve the triggering of genetic variations and reversible phenotypes caused by epigenetic modifications. However, the specific regulatory mechanisms remain unclear. Using CellAge and The Cancer Genome Atlas databases and in-house RNA-seq data, DNA methylation-modified cellular senescence genes (DMCSGs) were validated by Support Vector Machine and correlation analyses. In 1150 cases and 1342 controls, we identified colorectal cancer risk variants in DMCSGs. The regulatory effects of gene, variant, and DNA methylation were explored through dual-luciferase and 5-azacytidine treatment experiments, complemented by multiple database analyses. Biological functions of key gene were evaluated via cell proliferation assays, SA-β-gal staining, senescence marker detection, and immune infiltration analyses. The genetic variant rs4558926 in the downstream of TACC3 was significantly associated with colorectal cancer risk (OR = 1.35, P = 3.22 × 10–4). TACC3 mRNA expression increased due to rs4558926 C > G and decreased DNA methylation levels. The CpG sites in the TACC3 promoter region were regulated by rs4558926. TACC3 knockdown decreased proliferation and senescence in colorectal cancer cells. In addition, subjects with high-TACC3 expression presented an immunosuppressive microenvironment. These findings provide insights into the involvement of genetic variants of cellular senescence genes in the development and progression of colorectal cancer.





Similar content being viewed by others
Data availability
Raw data supporting the conclusions of this article will be made available by the authors, without undue reservation, to any qualified researcher. Supplementary material including antibody, primer sequence and small interfering RNA sequence is included with the article. TCGA, GEO (GSE39582), GTEx and HPA datasets were used to analyze the expression of TACC3 and other genes in multiple tissues and cell types. Additional data are available upon request from the corresponding author.
Abbreviations
- 5-Aza:
-
5-Azacytidine
- CCK-8:
-
Cell Counting Kit-8
- CIs:
-
Confidence intervals
- CSGs:
-
Cell senescence genes
- CTCF:
-
CCCTC-binding factor
- DEGs:
-
Differentially expressed genes
- DMCSGs:
-
DNA methylation-related cell senescence genes
- DNMT:
-
DNA methyltransferase
- EdU:
-
5-Ethynyl-2'-deoxyuridine
- eQTL:
-
Expression quantitative trait locus
- FDR:
-
False discovery rate
- GEO:
-
Gene Expression Omnibus
- GSEA:
-
Gene set enrichment analysis
- GTEx:
-
Genotype-Tissue Expression
- HWE:
-
Hardy–Weinberg equilibrium
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- LD:
-
Linkage disequilibrium
- MAF:
-
Minor allele frequency
- meQTLs:
-
Methylation quantitative trait loci
- MSI:
-
Microsatellite instability
- MSS:
-
Microsatellite stable
- MSI-H:
-
Microsatellite instability-high
- ORs:
-
Odds ratios
- PCA:
-
Principal component analysis
- qRT‒PCR:
-
Quantitative reverse transcription polymerase chain reaction
- SVM:
-
Support Vector Machine
- sQTLs:
-
Splicing quantitative trait loci
- ssGSEA:
-
Single-sample gene set enrichment analysis
- TACC3:
-
Transforming acid coiled coil containing protein 3
- TCGA:
-
The Cancer Genome Atlas
- TMB:
-
Tumor mutational burden
References
Akbulut O, Lengerli D, Saatci O et al (2020) A highly potent TACC3 inhibitor as a novel anticancer drug candidate. Mol Cancer Ther 19(6):1243–1254. https://doi.org/10.1158/1535-7163.Mct-19-0957
Ali HR, Chlon L, Pharoah PD, Markowetz F, Caldas C (2016) Patterns of immune infiltration in breast cancer and their clinical implications: a gene-expression-based retrospective study. PLoS Med 13(12):e1002194. https://doi.org/10.1371/journal.pmed.1002194
Angrisano T, Lembo F, Pero R et al (2006) TACC3 mediates the association of MBD2 with histone acetyltransferases and relieves transcriptional repression of methylated promoters. Nucleic Acids Res 34(1):364–372. https://doi.org/10.1093/nar/gkj400
Avelar RA, Ortega JG, Tacutu R et al (2020) A multidimensional systems biology analysis of cellular senescence in aging and disease. Genome Biol 21(1):91. https://doi.org/10.1186/s13059-020-01990-9
Bai X, Wei H, Liu W et al (2022) Cigarette smoke promotes colorectal cancer through modulation of gut microbiota and related metabolites. Gut 71(12):2439–2450. https://doi.org/10.1136/gutjnl-2021-325021
Bird A (2007) Perceptions of epigenetics. Nature 447(7143):396–398. https://doi.org/10.1038/nature05913
Borodkina AV, Shatrova AN, Deryabin PI, Grukova AA, Nikolsky NN, Burova EB (2016) Tetraploidization or autophagy: The ultimate fate of senescent human endometrial stem cells under ATM or p53 inhibition. Cell Cycle (georgetown, Tex) 15(1):117–127. https://doi.org/10.1080/15384101.2015.1121326
Chen Z, Trotman LC, Shaffer D et al (2005) Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 436(7051):725–730. https://doi.org/10.1038/nature03918
Chen D, Liu J, Zang L et al (2022) Integrated machine learning and bioinformatic analyses constructed a novel stemness-related classifier to predict prognosis and immunotherapy responses for hepatocellular carcinoma patients. Int J Biol Sci 18(1):360–373. https://doi.org/10.7150/ijbs.66913
Collado M, Gil J, Efeyan A et al (2005) Tumour biology: senescence in premalignant tumours. Nature 436(7051):642. https://doi.org/10.1038/436642a
De Blander H, Morel AP, Senaratne AP, Ouzounova M, Puisieux A (2021) Cellular plasticity: a route to senescence exit and tumorigenesis. Cancers 13(18):4561.
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB (2019) Colorectal cancer. Lancet (london, England) 394(10207):1467–1480. https://doi.org/10.1016/s0140-6736(19)32319-0
Ding ZM, Huang CJ, Jiao XF, Wu D, Huo LJ (2017) The role of TACC3 in mitotic spindle organization. Cytoskeleton (hoboken, NJ) 74(10):369–378. https://doi.org/10.1002/cm.21388
Gacci M, Corona G, Vignozzi L et al (2015) Metabolic syndrome and benign prostatic enlargement: a systematic review and meta-analysis. BJU Int 115(1):24–31. https://doi.org/10.1111/bju.12728
Gama-Sosa MA, Slagel VA, Trewyn RW et al (1983) The 5-methylcytosine content of DNA from human tumors. Nucleic Acids Res 11(19):6883–6894. https://doi.org/10.1093/nar/11.19.6883
Gu D, Li S, Ben S et al (2018) Circadian clock pathway genes associated with colorectal cancer risk and prognosis. Arch Toxicol 92(8):2681–2689. https://doi.org/10.1007/s00204-018-2251-7
Hamidi T, Singh AK, Chen T (2015) Genetic alterations of DNA methylation machinery in human diseases. Epigenomics 7(2):247–265. https://doi.org/10.2217/epi.14.80
Hanahan D (2022) Hallmarks of cancer: new dimensions. Cancer Discov 12(1):31–46. https://doi.org/10.1158/2159-8290.Cd-21-1059
Hawe JS, Wilson R, Schmid KT et al (2022) Genetic variation influencing DNA methylation provides insights into molecular mechanisms regulating genomic function. Nat Genet 54(1):18–29. https://doi.org/10.1038/s41588-021-00969-x
Hernandez-Segura A, Nehme J, Demaria M (2018) Hallmarks of cellular senescence. Trends Cell Biol 28(6):436–453. https://doi.org/10.1016/j.tcb.2018.02.001
Heyn H, Sayols S, Moutinho C et al (2014) Linkage of DNA methylation quantitative trait loci to human cancer risk. Cell Rep 7(2):331–338. https://doi.org/10.1016/j.celrep.2014.03.016
Huang W, Hickson LJ, Eirin A, Kirkland JL, Lerman LO (2022) Cellular senescence: the good, the bad and the unknown. Nat Rev Nephrol 18(10):611–627. https://doi.org/10.1038/s41581-022-00601-z
Huyghe JR, Bien SA, Harrison TA et al (2019) Discovery of common and rare genetic risk variants for colorectal cancer. Nat Genet 51(1):76–87. https://doi.org/10.1038/s41588-018-0286-6
Jeltsch A, Ehrenhofer-Murray A, Jurkowski TP et al (2017) Mechanism and biological role of Dnmt2 in Nucleic Acid Methylation. RNA Biol 14(9):1108–1123. https://doi.org/10.1080/15476286.2016.1191737
Jiang F, Kuang B, Que Y et al (2016) The clinical significance of transforming acidic coiled-coil protein 3 expression in non-small cell lung cancer. Oncol Rep 35(1):436–446. https://doi.org/10.3892/or.2015.4373
Lai AY, Wade PA (2011) Cancer biology and NuRD: a multifaceted chromatin remodelling complex. Nat Rev Cancer 11(8):588–596. https://doi.org/10.1038/nrc3091
Lee S, Schmitt CA (2019) The dynamic nature of senescence in cancer. Nat Cell Biol 21(1):94–101. https://doi.org/10.1038/s41556-018-0249-2
L’Espérance S, Popa I, Bachvarova M et al (2006) Gene expression profiling of paired ovarian tumors obtained prior to and following adjuvant chemotherapy: molecular signatures of chemoresistant tumors. Int J Oncol 29(1):5–24
Li S, Du M, Xu K et al (2023a) A germline variant in the BET1L 3’-UTR confers colorectal cancer susceptibility by reducing miRNA binding and m6A modification. Can Res. https://doi.org/10.1158/0008-5472.Can-22-0065
Li S, Xu S, Chen Y et al (2023b) Metal exposure promotes colorectal tumorigenesis via the aberrant N(6)-methyladenosine modification of ATP13A3. Environ Sci Technol 57(7):2864–2876. https://doi.org/10.1021/acs.est.2c07389
Llosa NJ, Cruise M, Tam A et al (2015) The vigorous immune microenvironment of microsatellite instable colon cancer is balanced by multiple counter-inhibitory checkpoints. Cancer Discov 5(1):43–51. https://doi.org/10.1158/2159-8290.Cd-14-0863
Maksimainen M, Paavilainen S, Hakulinen N, Rouvinen J (2012) Structural analysis, enzymatic characterization, and catalytic mechanisms of β-galactosidase from Bacillus circulans sp. alkalophilus. FEBS J 279(10):1788–1798. https://doi.org/10.1111/j.1742-4658.2012.08555.x
Milanovic M, Fan DNY, Belenki D et al (2018) Senescence-associated reprogramming promotes cancer stemness. Nature 553(7686):96–100. https://doi.org/10.1038/nature25167
Morgan E, Arnold M, Gini A et al (2023) Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut 72(2):338–344. https://doi.org/10.1136/gutjnl-2022-327736
Murphy SJ, Hart SN, Lima JF et al (2013) Genetic alterations associated with progression from pancreatic intraepithelial neoplasia to invasive pancreatic tumor. Gastroenterology 145(5):1098-1109.e1. https://doi.org/10.1053/j.gastro.2013.07.049
Petrova NV, Velichko AK, Razin SV, Kantidze OL (2016) Small molecule compounds that induce cellular senescence. Aging Cell 15(6):999–1017. https://doi.org/10.1111/acel.12518
Salama R, Sadaie M, Hoare M, Narita M (2014) Cellular senescence and its effector programs. Genes Dev 28(2):99–114. https://doi.org/10.1101/gad.235184.113
Schmit SL, Schumacher FR, Edlund CK et al (2016) Genome-wide association study of colorectal cancer in Hispanics. Carcinogenesis 37(6):547–556. https://doi.org/10.1093/carcin/bgw046
Schmit SL, Edlund CK, Schumacher FR et al (2019) Novel common genetic susceptibility loci for colorectal cancer. J Natl Cancer Inst 111(2):146–157. https://doi.org/10.1093/jnci/djy099
Schübeler D (2015) Function and information content of DNA methylation. Nature 517(7534):321–326. https://doi.org/10.1038/nature14192
Seale K, Horvath S, Teschendorff A, Eynon N, Voisin S (2022) Making sense of the ageing methylome. Nat Rev Genet 23(10):585–605. https://doi.org/10.1038/s41576-022-00477-6
Serrano M, Lin AW, McCurrach ME, Beach D, Lowe SW (1997) Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88(5):593–602. https://doi.org/10.1016/s0092-8674(00)81902-9
Shriver M, Stroka KM, Vitolo MI et al (2015) Loss of giant obscurins from breast epithelium promotes epithelial-to-mesenchymal transition, tumorigenicity and metastasis. Oncogene 34(32):4248–4259. https://doi.org/10.1038/onc.2014.358
Siegel RL, Wagle NS, Cercek A, Smith RA, Jemal A (2023) Colorectal cancer statistics, 2023. CA: a cancer journal for clinicians doi:https://doi.org/10.3322/caac.21772
Song H, Liu C, Shen N et al (2018) Overexpression of TACC3 in breast cancer associates with poor prognosis. Applied Immunohistochemistry & Molecular Morphology : AIMM 26(2):113–119. https://doi.org/10.1097/pai.0000000000000392
Sun J, Shi R, Zhang X et al (2021) Characterization of immune landscape in papillary thyroid cancer reveals distinct tumor immunogenicity and implications for immunotherapy. Oncoimmunology 10(1):e1964189. https://doi.org/10.1080/2162402x.2021.1964189
Wang L, Lankhorst L, Bernards R (2022) Exploiting senescence for the treatment of cancer. Nat Rev Cancer 22(6):340–355. https://doi.org/10.1038/s41568-022-00450-9
Wu T, Zhang X, Liu X et al (2023) Single-cell sequencing reveals the immune microenvironment landscape related to anti-PD-1 resistance in metastatic colorectal cancer with high microsatellite instability. BMC Med 21(1):161. https://doi.org/10.1186/s12916-023-02866-y
Xu C, Li F, Liu Z, Yan C, Xiao J (2022) A novel cell senescence-related IncRNA survival model associated with the tumor immune environment in colorectal cancer. Front Immunol 13:1019764. https://doi.org/10.3389/fimmu.2022.1019764
Zheng CC, Liao L, Liu YP, et al. (2022) Blockade of nuclear β-catenin signaling via direct targeting of RanBP3 with NU2058 induces cell senescence to suppress colorectal tumorigenesis. Adv Sci (Weinheim, Baden-Wurttemberg, Germany) 9(34):e2202528 doi:https://doi.org/10.1002/advs.202202528
Acknowledgements
We sincerely thank Professor Meilin Wang of Nanjing Medical University for his selfless help and valuable advice. His expertise and guidance played a key role in our research, enabling us to complete this work successfully.
Funding
This work was supported by the National Natural Science Foundation of China (82172956, 82103915), and the Natural Science Foundation of Jiangsu Province (BK20210535).
Author information
Authors and Affiliations
Contributions
SL, ZF were the overall principal investigators in this study who conceived the study and obtained financial support, SL, ZF were responsible for the study design and supervised the entire study. HS, YC, and MX performed statistical analyses, interpreted the results, and drafted the initial manuscript. HS, YC, MX, JZ, CH, and ZW performed laboratory experiments, JZ, CH, ZW, YS, HZ, and YL were responsible for patient recruitment and sample preparation. All authors approved the final report for publication. HS, YC, and MX were contribute equally to this work.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shen, H., Chen, Y., Xu, M. et al. Cellular senescence gene TACC3 associated with colorectal cancer risk via genetic and DNA methylated alteration. Arch Toxicol 98, 1499–1513 (2024). https://doi.org/10.1007/s00204-024-03702-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-024-03702-9