Abstract
Background
Grip strength predicts long-term morbidity and mortality in adults. We compared grip strength in adults born with extremely low birth weight (ELBW; under 1 kg) and a normal birth weight control group (NBW) and describe change in grip strength over a 10-year period in a longitudinal cohort study of preterm birth.
Methods
Grip strength, body composition, and device-measured physical activity were assessed in 95 mature adults (MA) born ELBW (age 31.6 (1.6) mean (SD) years, 59 females) and 88 born NBW (age 31.9 (1.4) years, 52 females). Regression models were used to examine the effect of perinatal factors, body composition, physical activity, and physical self-efficacy on grip strength.
Results
Grip strength was lower in MA born ELBW compared to NBW (31.8 (10.0) vs. 39.8 (11.2) kg; p < 0.001). Birth weight group was associated with grip strength independent of sex, height, and lean mass index, but device-measured physical activity was not. The change in grip strength from mid-20s to MA was similar in ELBW and NBW participants.
Discussion
Grip strength in MA born ELBW is low and is similar to a reference group 25–30 years older, suggesting higher risk for cardiovascular and all-cause mortality.
Impact
-
Adults born extremely preterm have reduced grip strength compared to control participants born at full term.
-
Reduced grip strength is a predictor of frailty and increased cardiovascular disease risk.
-
Change in grip strength from age in mid-20s to mid-30s is similar in those born preterm and full-term-born controls.
-
Grip strength is related to lean mass and not to device-measured physical activity—and correlates of grip strength are similar in those born preterm and term-born controls.
-
Grip strength is a simple measure that may provide information about the health of adults born preterm.
Similar content being viewed by others
Introduction
Adults born with extreme prematurity have a higher risk of having adverse cardiometabolic health. In multiple cohort studies, hypertension,1 insulin resistance,2 and dysglycemia3 are more prevalent in adults born with prematurity and extremely low or very low birth weight (ELBW—birth weight under 1000 g and VLBW—birth weight under 1500 g, respectively) compared to term-born controls. While the underlying mechanisms explaining these findings are not fully understood, these individuals also have lower lean mass and higher body fat percentage than the control population,3 which may contribute to the aforementioned cardiometabolic disturbances.
Greater attention is being paid to the relationship between muscular mass, function, and adult health, particularly in the elderly. Sarcopenia is an age-related condition of generalized, progressive loss of skeletal muscle mass and strength.4 Adults with sarcopenia have greater challenges with mobility, poorer health-related quality of life and higher risk of premature mortality. In cross-sectional studies, muscle mass is highest in men and women in the third decade of life and then declines.5 In older adults, physical activity interventions can slow this decline in muscle mass.6 A key feature of sarcopenia is low muscle strength. Grip strength is recognized as a simple measure of muscle strength that has been relatively widely studied in adults.4 Grip strength peaks in early adult life, remains relatively stable through mid-life and then declines.7 In a large, multinational study, reduced grip strength predicted all-cause and cardiovascular-specific mortality in adults.8 Given the findings of lower lean mass and poorer cardiometabolic health indicators in adults born extremely preterm, we were interested in evaluating grip strength in adults born ELBW.
The McMaster ELBW cohort, comprised of people born between 1978 and 1982, is one of the oldest known cohorts of survivors of extreme prematurity. The cohort is comprised of people with birth weight under 1000 g and age and sex-matched normal birth weight (NBW) controls. Periodic assessments of this cohort have contributed substantially to our understanding of the implications of extreme preterm birth.9 As part of the evaluation of this cohort when seen in their early 30s, very close to the predicted peak of lean mass accretion and grip strength, we sought to determine if grip strength differed in those born ELBW compared to control. Furthermore, we sought to determine if early life exposures, adult body composition, and device-measured physical activity were related to grip strength in both groups.
Methods
Participants
The McMaster longitudinal cohort is a population-based study of individuals recruited from a geographically defined region of central-west Ontario, Canada from 1977 to 1982. The participants born ELBW have been followed since birth and health outcomes have been reported at multiple time points from childhood to young adulthood (22–26 years) and, most recently, in their early 30s.3 The age-, gender- and socioeconomic status (SES)-matched NBW group was identified at 8 years of age and they have since been followed at the same time points as the ELBW group.
Ethics
The study was approved by the joint Research Ethics Board of McMaster University and Hamilton Health Sciences. Participants provided informed, signed consent.
Mature adult—visits and measurement
Data collection for the mature adult (MA) visit occurred at a single study visit when the participants were 29–36 years of age. The visit was scheduled in the morning, after an 8–12 h fast (water was permitted). Subsequent to an oral glucose tolerance test, the participants were offered breakfast. Grip strength was measured following a standardized protocol using a Jamar Hand Dynamometer (Model 5030J1, Bolingbrook, IL, USA). While standing, participants were instructed to hold the dynamometer in their dominant hand with the arm extended at the elbow and to squeeze the dynamometer with as much force as possible, sustaining the pressure for a count of three. The assessment was repeated twice more with a rest of at least 30 s between measures. Grip strength measurement was then repeated using the same protocol on the nondominant hand. Using the standardized protocol of Leong et al.,8 our primary measure of grip strength was defined as “combined grip strength” and was calculated from the mean of the maximum values of the nondominant and dominant hand grip strength. If only one hand was measured, the maximum value of that hand was used as the measure of overall grip strength. No participants were missing dominant hand measures, two were missing measurement from the nondominant hand. In addition, in order to compare our cohort with published normative values,7 we utilized the approach suggested by Roberts et al.,10 and defined absolute grip strength as maximum of all values measured (dominant and nondominant).
At the end of the study visit, the study participants were fitted with an Actigraph GTIM (n = 34) or GT3X+ (n = 54) accelerometer to measure device-measured physical activity and sedentary time. Each participant was also given a diary for tracking physical activities, wear time, and sleep. Participants were asked to wear the monitor on their hip for 7 consecutive days, during waking hours, except during water activities (e.g. swimming or showering). Data were collected in 60-s sampling intervals or epochs. Accelerometer cut points developed by Freedson et al.11 for adults were applied to define average daily time spent sedentary and in light, moderate, and vigorous physical activity. Data were considered valid and included in the analyses if individuals wore the monitor for ≥3 days with ≥10 h per day.12 All those with valid data were included. Self-reported physical activity was also assessed using the International Physical Activity Questionnaire (IPAQ).13
Anthropometric measures and questionnaire data were collected as previously described.14 Briefly, standing height, measured using a Harpenden Stadiometer (London, UK), and weight were measured three times and averaged, and BMI (kg/m2) was calculated. Waist circumference (WC) was measured to the nearest 0.1 cm over an unclothed abdomen at the smallest diameter between the costal margin and the iliac crest. Body composition was assessed using dual energy X-ray absorptiometry on a GE Lunar Prodigy Advance (Model #8743) scanner. Total lean mass (kg) for each participant was adjusted for current adult height by calculating the lean mass index (LMI = total lean mass (kg)/height (m2)) and body fat was presented as total body fat %. BP was measured with the participant seated, using the right arm with an appropriately sized cuff and the BpTRU (Coquitlam, BC, Canada) device. BpTRU is an automated device that takes five serial blood pressure measurements—the average of these measures was utilized. Participants completed questionnaires related to demographic characteristics (education, marital status and income, SES), personal health history including medication use and family history of diabetes and cardiovascular disease. Study personnel were unaware of group assignment prior to completion of the study visit.
Measures collected at earlier study visits
To evaluate earlier life factors that might influence grip strength in the fourth decade of life, and to better understand the trajectory of this health measure, we utilized data collected from previous study visits. Birth weight, gestational age (weeks), birth weight for gestational age, presence or absence of neurosensory impairment, maternal smoking during pregnancy and maternal steroid exposure during pregnancy were available. NBW were known to be born at full term. Select data from the YA visit (22–26 years of age) were included in the current analyses. These measures include grip strength in cohort participants with no neurosensory impairment, assessed as described above.15 Physical self-efficacy and the subscale perceived physical ability were assessed at the YA visit using the validated questionnaire developed by Ryckman et al.16
Statistical analysis
Statistical analyses were conducted using SPSS versions 25 and 26 (IBM SPSS Statistics, IBM Corporation, Armonk, NY). To address our primary question related to differences in grip strength between the ELBW and NBW groups (overall and gender specific), independent t tests were used. Grip strength comparisons were completed including and excluding those with neurosensory impairments. A two-way repeated measures ANOVA was performed for comparison of grip strength overtime (group: ELBW/NBW) by time (YA/MA). Device-measured physical activity (including minutes of sedentary time, light, moderate and vigorous physical activity, and total counts) was compared between the ELBW and NBW groups who had valid physical activity data.
Univariate regression analysis was used to analyze the contribution of predictors of grip strength. Predictors examined included birth weight group, birth weight (g), gestational age, maternal corticosteroid exposure, gender, neurosensory impairment at birth, height, BMI, WC, total percent body fat, LMI, moderate-to-vigorous physical activity (MVPA), and total activity counts. To evaluate the predictors of change in grip strength from YA to MA, grip strength and total physical self-efficacy at YA were also examined. Multiple regression was used to determine the influence of variables significant in the univariate analysis on grip strength. The z-distribution scores for skewness and kurtosis using a conservative alpha level of p = 0.001 were used to assess normality of the scores for each of the variables prior to the regression analyses;17 LMI was the only variable that was not normally distributed; however, transformation was not done given that it was used as a covariate and not an outcome variable. Multicollinearity was evaluated and where present, the measure with the closest relation in univariate analysis was included in the multivariate model. An α of p ≤ 0.05 was considered statistically significant. Variable descriptives for normally distributed data are presented as mean (SD).
Results
Study participants
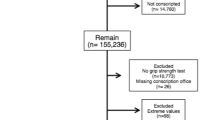
One hundred of the surviving 148 (67.6%) ELBW participants and 89 (66.9%) of the 133 NBW participants recruited at age 8 years and seen at YA participated in this assessment. Completion of questionnaires, grip strength measures, and the availability of valid physical activity data are presented in Fig. 1. Those who attended the MA visit were generally similar to those who did not with the exception that the ELBW that attended the visit had a higher likelihood of being born small for gestational age than those who did not attend (Supplementary Table 1). A total of 27 participants at the most recent visit had a neurosensory impairment including 26 participants in the ELBW group (visual impairment, n = 12; cerebral palsy, n = 10; intellectual development disorder, n = 2; and other, n = 2) and one participant from the NBW group with unilateral deafness.
Clinical outcomes—mature adult visit
The characteristics of the study participants who attended the MA visit are presented in Table 1. When seen at the MA visit (age range 29–36 years), both male and female ELBW participants had lower height (p < 0.001) than the NBW group. The body weight for females was similar between the groups, but males with ELBW and the overall ELBW group weighed less than those born NBW. BMI and WC were similar between groups in both males and females. Male and female ELBW participants had higher total body fat percentage (p = 0.004) and males had lower lean mass (p = 0.003) than the male NBW participants.
Handgrip strength
Handgrip strength, measured either as combined grip strength of the dominant and nondominant hands or as the absolute maximum from three repeated efforts with each hand, was lower in male and female ELBW compared to NBW (see Fig. 2). The published normative mean (SD) values for absolute grip strength for males at 30 years of age is 51.6 (9.6) kg and for females is 31.4 (6.0) kg.7 While the NBW participants had similar mean grip strength (male 53.5 (8.0) kg and female 33.5 (5.2) kg), the mean values in both male and female ELBW participants were well below the normative values (male 43.1 (8.6) and female 27.5 (5.4)). Thus, the mean grip strength of ELBW males was similar to a 65-year-old male in the reference population and grip strength for ELBW females corresponded to a 55–60-year-old female in the reference population. As grip strength is related to height and the ELBW participants were shorter than the NBW participants, we also expressed the combined grip strength for height and had similar findings (ELBW 19.2 (5.2) vs. NBW 23.0 (5.3); p < 0.001). These results are consistent with our previously published findings for this cohort at the YA visit approximately 10 years earlier.15 An important distinction between the studies is that individuals with neurosensory impairments did not have grip strength measured at the YA visit. At MA, the subset of ELBW participants with neurosensory impairments did have lower grip strength than those without (28.0 (8.3) vs. 33.2 (10.2); p = 0.023), but when comparing ELBW and NBW adults without neurosensory impairments, lower grip strength was still noted in the ELBW group (33.2 (10.2) vs. 39.9 (11.2); p < 0.001).
a, b Absolute and combined max grip strength in ELBW (n = 95) and NBW (n = 88) at MA, *p < 0.001. c, d Absolute and combined max grip strength for height in YA and MA ELBW (n = 65) and NBW (n = 80), data presented as mean (SD), *p < 0.001 between groups and sexes, †p < 0.001 between visit dates. ELBW extremely low birth weight, NBW normal birth weight, YA young adult, MA mature adult.
Grip strength was measured at both the YA and MA visits for a subset of participants with no neurosensory impairments. As shown in Fig. 2, grip strength increased from YA to MA in both birth weight groups. Importantly, the rate of change of grip strength over this time period was similar in the ELBW and NBW groups.
Device-measured physical activity
A subset of participants who had grip strength measured at the MA visit also wore an accelerometer (n = 58 ELBW and n = 52 NBW) and 49 ELBW participants and 39 NBW participants had valid data based on wear time (Table 2). No group differences in total activity counts or in time spent sedentary or in light, moderate, or vigorous physical activity were identified. Similarly, no differences were found when we included the physical activity data from all participants irrespective of whether or not they had valid physical activity measures based on wear time. ELBW participants self-reported similar physical activity compared to the NBW group.
Correlates of combined grip strength
We sought to understand if early life factors and/or current activity behaviors might be related to grip strength and if these factors differed in those born ELBW compared to NBW. In univariate analysis, birth weight group, gestational age, birth weight, sex, and current anthropometric characteristics (height, waist circumference, body fat percentage, and lean mass index) were related to grip strength in both the ELBW and NBW groups (Table 3). Physical activity measures (MVPA and average daily activity counts) were also related to grip strength in univariate analysis. Due to problems of multicollinearity, (1) birth weight group and gestational age, (2) body fat percentage, WC, and LMI, and (3) MVPA and total activity counts could not be in the same model. Thus, we selected the variable with the strongest relation to grip strength, from each of these groups, which were: (1) birth weight group, (2) LMI, and (3) MVPA. These were entered, together with participant sex and height, in the final multivariate models shown in Table 3. Birth weight group was related to grip strength independent of sex, height, and LMI and together these variables explained 81.5% of the variation in grip strength. In the subset with valid device-measured physical activity, the relationship of physical activity to grip strength was not significant when birth weight group, sex, and LMI were included in the model. It is conceivable that LMI mediates the relationship between physical activity and grip strength, but as physical activity was only weakly related to LMI in univariate analysis (r = 0.136; p = 0.084), LMI is clearly influenced by factors beyond current physical activity.
To examine potential early life predictors relevant only to the ELBW group, we also conducted univariate and multivariate analyses in the ELBW group alone. In univariate analysis, early life exposures including birth weight, gestational age, maternal corticosteroid exposure, and the presence of a neurosensory impairment were related to grip strength in the fourth decade of life. In the multivariate model, only sex, height, and the presence of a neurosensory impairment independently predicted grip strength. The overall model was significant, explaining 70% of the variance in grip strength in ELBW participants (R2 adjusted = 0.706, p < 0.001).
As described earlier, grip strength, physical self-efficacy, and perceived physical ability were assessed at the YA visit in study participants with no known neurosensory impairments. We were interested to know if these measures predicted grip strength 8–10 years later (see Table 4). Grip strength in the fourth decade was tightly correlated with grip strength in young adulthood (r = 0.92; p < 0.001). Interestingly, total physical self-efficacy scores and perceived physical ability were both directly related to grip strength a decade later in both males and females. However, they were no longer significant when examined in the multivariate model including birth weight group, sex, height, and LMI (Table 4).
Discussion
In this longitudinal cohort study, we have demonstrated that adults born ELBW have lower muscular fitness, measured as handgrip strength, compared to NBW controls. Further, although the association was attenuated when height, lean mass, and device-measured physical activity were included in the model, it is still present and significant. We had shown a similar difference at young adulthood among study participants without evidence of a neurosensory impairment, and now demonstrate that the change in grip strength from young adulthood to mature adulthood is not different in those born ELBW compared to those born NBW.
Handgrip strength
Reduced handgrip strength is a prognostic marker of both all-cause and cardiovascular mortality in adults.8,18 In a large, multinational study, grip strength had strong predictive value for all-cause mortality (hazard ratio (HR) 1.37, 95%CI 1.28–1.47; p < 0.0001) and cardiovascular mortality (HR 1.45, 95%CI 1.30–1.63; p < 0.0001) per SD lower grip strength. In this study, these HRs exceeded or were comparable to the well-known cardiovascular risk factor, systolic blood pressure.8 On average, grip strength in our cohort was 9.8 kg (19.2%) lower in the adult males born ELBW compared to NBW and the adult females born ELBW had 6 kg (18.8%) lower grip strength compared to NBW. Based on the Leong paper, this would correspond to an approximately 40% higher risk for all-cause mortality.
Low birth weight has been consistently associated with reduced grip strength across the lifecourse as previously reviewed.19 In the meta-analysis including all age groups from childhood to older adults, grip strength was 0.86 kg lower per kg lower birth weight. In our study, the grip strength difference between ELBW and NBW appears to be much greater per kg birth weight (3.76 kg per kg birth weight for males and 2.4 kg per kg birth weight for females). Although most of the studies in the meta-analysis evaluated individuals born full term, one study compared adolescents born extremely preterm to those born full term with comparable findings to the current study.20 In the ESTER study, YA born early preterm, but not those born late preterm, had lower grip strength compared to control.21 Thus, grip strength appears to be consistently lower in adults born early preterm compared to controls.
We examined the longitudinal change in grip strength between the YA and MA visits of our cohort, which corresponds to a time leading up to peak muscle strength. We found no difference in the change in grip strength from YA to MA in those born ELBW compared to NBW. This finding suggests that early-life exposures contribute to muscle strength differences identifiable by early adulthood in those born preterm. Further, our findings also suggest that adults born preterm do not get further behind in muscle strength as they move from the third to the fourth decade of life. Further follow-up of this, and other cohorts will enable the understanding of the influence of subsequent aging on grip strength in those born preterm.
The early life factors that influence grip strength in adulthood in those born preterm are understudied. As adults born ELBW are consistently shorter than controls and height is a known predictor of adult grip strength, it is important to include height as a confounder in examining these factors. Lower lean mass is a key predictor of grip strength and it is known that infants born with extreme prematurity have lower lean mass during infancy22 and beyond. Furthermore, self-reported23,24 and device-measured physical activity25 have been previously associated with grip strength in adults and several studies have suggested that adults born preterm self-report less physical activity.26 Thus, we were interested in determining whether or not these variables could explain the group-based differences in grip strength that we observed. Birth weight group remained a significant predictor of grip strength, independent of device-measured physical activity and lean mass index. Thus, although lean mass index is lower in adults born with ELBW compared to controls and is a significant predictor of grip strength, it does not fully account for the birth weight group differences in grip strength we have identified. No group differences in device-measured physical activity were apparent in our cohort, consistent with findings of device-measured physical activity in several other studies of youth27,28 and adults29 born preterm. Furthermore, physical activity was not related to grip strength in the multivariate model. Thus, birth weight group was a contributor to differences in grip strength in adulthood independent of the covariate height and the potential mediators lean mass index and physical activity.
In a large, family-based analysis evaluating the relationship of birth weight to grip strength in males born near term, the factors contributing to grip strength were primarily those not shared by siblings.30 The authors suggested, therefore, that in utero nutrition may be a key contributor given that the last trimester of pregnancy is critical for muscle fiber development. As the infants born ELBW experience very significant health and nutrition issues during this period of muscle fiber development as a result of their preterm birth, it is conceivable that this may be an important contributor to the deficit in grip strength noted in adulthood. Further research examining nutrition directly would be necessary to better examine this.
We were interested in examining the relationship to grip strength of other early-life exposures specific to infants born ELBW. We evaluated birth weight, gestational age, maternal exposure to corticosteroids, and the presence of a neurosensory impairment. The presence of a neurosensory impairment and gestational age (p = 0.063) were weakly related to grip strength independent of height and sex. In contrast to previous research in which exposure to antenatal corticosteroids was associated with higher aerobic fitness in 14-year-old boys born preterm,31 and to animal-based studies in which antenatal corticosteroid exposure had a detrimental influence on skeletal muscle development,32 we identified no relationship to grip strength in this study.
Age is a well-described determinant of muscle strength, with lifecourse analyses suggesting that peak muscle strength occurs early in the fourth decade followed by an annual decline in grip strength of 0.5–1%.33 This decline is partially related to declining muscle mass with aging, but other related factors have also been identified including ethnicity34 and BMI.35 This is, to our knowledge, the first longitudinal study of grip strength in adults born preterm. ELBW and NBW groups had similar increases in grip strength between the YA and MA visits. Further, in univariate analyses, we found that total physical self-efficacy and perceived physical ability at YA is related to grip strength a decade later. The key positive predictors of grip strength at MA were male sex, lean mass for height, height, and grip strength at baseline. These correlates were similar for both birth weight groups. It is likely that the influence of physical self-efficacy and perceived physical ability may have contributed to grip strength at baseline and to lean mass accretion and thus do not appear as independent predictors of grip strength at MA.
Although our study offers insights into grip strength, an important predictor of long-term health, in adults born with extreme prematurity, it is not without limitations. As in many longitudinal follow-up studies, loss to follow-up has occurred and this may contribute to some bias in our results. Furthermore, not all participants had valid device-measured physical activity. There have been many changes in NICU care since members of this cohort were born; therefore, applicability to infants born today is uncertain and the study of more modern cohorts will be important. However, since grip strength is a well-established predictor of morbidity and mortality, it is important to follow these participants and other cohorts as they move into middle- and late-middle age. Longitudinal investigation of muscle strength in children born more recently will potentially lead to improved understanding of factors influencing the development of muscle strength during childhood in those born preterm, which may assist in better elucidating the underlying mechanisms and ultimately lead to the design of interventions to target impairments in muscle strength.36
Conclusions
Adults born ELBW have reduced grip strength compared to those born NBW at term. This reduced grip strength may be an early marker of increased frailty and all-cause mortality as ELBW adults age. Based on findings from the PURE study, the combined grip strength difference of 8 kg between ELBW and NBW suggests a substantially increased risk of all-cause mortality. Further examination of the early-life factors related to lower muscle strength and consideration of interventions that might target low muscle strength in those born ELBW is warranted in future research.
References
Hovi, P. et al. Blood pressure in young adults born at very low birth weight: adults born preterm international collaboration. Hypertension 68, 880–887 (2016).
Hovi, P. et al. Glucose regulation in young adults with very low birth weight. N. Engl. J. Med. 356, 2053–2063 (2007).
Morrison, K. M. et al. Cardiometabolic health in adults born premature with extremely low birth weight. Pediatrics 138, e20160515 (2016).
Shaw, S. C., Dennison, E. M. & Cooper, C. Epidemiology of sarcopenia: determinants throughout the lifecourse. Calcif. Tissue Int. 101, 229–247 (2017).
Janssen, I., Heymsfield, S. B., Wang, Z. M. & Ross, R. Skeletal muscle mass and distribution in 468 men and women aged 18-88 yr. J. Appl. Physiol. 89, 81–88 (2000).
Lee, S.-Y., Tung, H.-H., Liu, C.-Y. & Chen, L.-K. Physical activity and sarcopenia in the geriatric population: a systematic review. J. Am. Med. Dir. Assoc. 19, 378–383 (2018).
Dodds, R. M. et al. Grip strength across the life course: normative data from twelve British studies. PLoS ONE 9, e113637 (2014).
Leong, D. P. et al. Prognostic value of grip strength: findings from the Prospective Urban Rural Epidemiology (PURE) study. Lancet 386, 266–273 (2015).
Saigal, S. et al. Transition of extremely low-birth-weight infants from adolescence to young adulthood: comparison with normal birth-weight controls. JAMA 295, 667–675 (2006).
Roberts, H. C. et al. A review of the measurement of grip strength in clinical and epidemiological studies: towards a standardised approach. Age Ageing 40, 423–429 (2011).
Freedson, P. S., Melanson, E. & Sirard, J. Calibration of the Computer Science and Applications, Inc. accelerometer. Med. Sci. Sports Exerc. 30, 777–781 (1998).
Tudor-Locke, C., Camhi, S. M. & Troiano, R. P. A catalog of rules, variables, and definitions applied to accelerometer data in the National Health and Nutrition Examination Survey, 2003−2006. Prev. Chronic Dis. 9, E113 (2012).
Hagströmer, M., Oja, P. & Sjöström, M. The International Physical Activity Questionnaire (IPAQ): a study of concurrent and construct validity. Public Health Nutr. 9, 755–762 (2006).
Morrison, K. R. Cardiometabolic health in adults born premature with extremely low birth weight. Pediatrics 138, e20160515 (2016). https://doi.org/10.1542/peds.2016-0515.
Saigal, S. et al. Comparison of current health, functional limitations, and health care use of young adults who were born with extremely low birth weight and normal birth weight. Pediatrics 119, e562–e573 (2007).
Ryckman, R. M., Robbins, M. A., Thornton, B. & Cantrell, P. Development and validation of a physical self-efficacy scale. J. Personal. Soc. Psychol. 42, 891–900 (1982).
Tabachnick, B. G. & Fidell, L. S. Using Multivariate Statistics, 5th edn (Pearson/Allyn & Bacon, Boston, 2007).
Cooper, R., Kuh, D., Hardy, R. & Mortality Review Group, on behalf of the FALCon and HALCyon study teams. Objectively measured physical capability levels and mortality: systematic review and meta-analysis. BMJ 341, c4467 (2010).
Dodds, R. et al. Birth weight and muscle strength: a systematic review and meta-analysis. J. Nutr. Health Aging 16, 609–615 (2012).
Rogers, M., Fay, T. B., Whitfield, M. F., Tomlinson, J. & Grunau, R. E. Aerobic capacity, strength, flexibility, and activity level in unimpaired extremely low birth weight (<or=800 g) survivors at 17 years of age compared with term-born control subjects. Pediatrics 116, e58–e65 (2005).
Tikanmäki, M. et al. Physical fitness in young adults born preterm. Pediatrics 137, e20151289 (2016).
Hamatschek et al. Fat and fat-free mass of preterm and term infants from birth to six months: a review of current evidence. Nutrients 12, 288 (2020).
Cooper, R., Mishra, G. D. & Kuh, D. Physical activity across adulthood and physical performance in midlife. Am. J. Prev. Med. 41, 376–384 (2011).
Kuh, D., Bassey, E. J., Butterworth, S., Hardy, R., Wadsworth, M. E. J. & the Musculoskeletal Study Team. Grip strength, postural control, and functional leg power in a representative cohort of British men and women: associations with physical activity, health status, and socioeconomic conditions. J. Gerontol. Ser. A: Biol. Sci. Med. Sci. 60, 224–231 (2005).
Cooper, A. J. M., Simmons, R. K., Kuh, D., Brage, S., Cooper, R. & NSHD Scientific and Data Collection Team. Physical activity, sedentary time and physical capability in early old age: British Birth Cohort Study. PLoS ONE 10, e0126465 (2015).
Sipola-Leppänen, M. et al. Resting energy expenditure in young adults born preterm—the Helsinki study of very low birth weight adults. PLoS ONE 6, e17700 (2011).
Welsh, L. et al. The EPICure study: maximal exercise and physical activity in school children born extremely preterm. Thorax 65, 165–172 (2010).
Lowe, J. et al. Physical activity in school-age children born preterm. J. Pediatrics 166, 877–883 (2015).
Kaseva, N. et al. Objectively measured physical activity in young adults born preterm at very low birth weight. J. Pediatrics 166, 474–476 (2015).
Ahlqvist, V. H. et al. Birth weight and grip strength in young Swedish males: a longitudinal matched sibling analysis and across all body mass index ranges. Sci. Rep. 9, 9719 (2019).
Nixon, P. A. et al. Antenatal steroid exposure, aerobic fitness, and physical activity in adolescents born preterm with very low birth weight. J. Pediatrics 215, 98–106.e2 (2019).
Gokulakrishnan, G., Chang, X., Fleischmann, R. & Fiorotto, M. L. Precocious glucocorticoid exposure reduces skeletal muscle satellite cells in the fetal rat. J. Endocrinol. 232, 561–572 (2017).
Frederiksen, H. et al. Age trajectories of grip strength: cross-sectional and longitudinal data among 8,342 Danes aged 46 to 102. Ann. Epidemiol. 16, 554–562 (2006).
Araujo, A. B. et al. Lean mass, muscle strength, and physical function in a diverse population of men: a population-based cross-sectional study. BMC Public Health 10, 508 (2010).
Forrest, K. Y. Z. et al. Patterns and correlates of grip strength change with age in Afro-Caribbean men. Age Ageing 41, 326–332 (2012).
Siebel, A. L., Carey, A. L. & Kingwell, B. A. Can exercise training rescue the adverse cardiometabolic effects of low birth weight and prematurity? Clin. Exp. Pharm. Physiol. 39, 944–957 (2012).
Acknowledgements
Funding for this project was provided by the Canadian Institutes of Health Research (Grant # 119386) and McMaster Children’s Hospital Foundation.
Author information
Authors and Affiliations
Contributions
K.M.M., E.G., L.A.S. and S.S. contributed to study design; K.M.M., L.A.S., S.S. and S.G. contributed to acquisition of data, E.G. and K.M.M. contributed to analysis and drafting the article, J.O. contributed to analysis and interpretation of the physical activity data, K.M.M. contributed to interpreting data. All authors critically revised the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Patient consent
Informed written consent was obtained from each study participant.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Morrison, K.M., Gunn, E., Guay, S. et al. Grip strength is lower in adults born with extremely low birth weight compared to term-born controls. Pediatr Res 89, 996–1003 (2021). https://doi.org/10.1038/s41390-020-1012-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41390-020-1012-5
This article is cited by
-
Impact of preterm birth on muscle mass and function: a systematic review and meta-analysis
European Journal of Pediatrics (2024)
-
From premature birth to premature kidney disease: does accelerated aging play a role?
Pediatric Nephrology (2023)