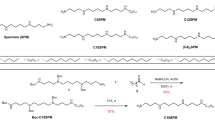
Abstract
Mass spectrometry (MS) of intact soluble protein complexes has emerged as a powerful technique to study the stoichiometry, structure-function and dynamics of protein assemblies. Recent developments have extended this technique to the study of membrane protein complexes, where it has already revealed subunit stoichiometries and specific phospholipid interactions. Here we describe a protocol for MS of membrane protein complexes. The protocol begins with the preparation of the membrane protein complex, enabling not only the direct assessment of stoichiometry, delipidation and quality of the target complex but also the evaluation of the purification strategy. A detailed list of compatible nonionic detergents is included, along with a protocol for screening detergents to find an optimal one for MS, biochemical and structural studies. This protocol also covers the preparation of lipids for protein-lipid binding studies and includes detailed settings for a quadrupole time-of-flight (Q-TOF) mass spectrometer after the introduction of complexes from gold-coated nanoflow capillaries.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Dorsam, R.T. & Gutkind, J.S. G-protein-coupled receptors and cancer. Nat. Rev. Cancer 7, 79–94 (2007).
Lappano, R. & Maggiolini, M. G protein-coupled receptors: novel targets for drug discovery in cancer. Nat. Rev. Drug. Discov. 10, 47–60 (2011).
Barrera, N.P., Di Bartolo, N., Booth, P.J. & Robinson, C.V. Micelles protect membrane complexes from solution to vacuum. Science 321, 243–246 (2008).
Barrera, N.P. & Robinson, C.V. Advances in the mass spectrometry of membrane proteins: from individual proteins to intact complexes. Annu. Rev. Biochem. 80, 247–271 (2011).
Zhou, M. et al. Mass spectrometry of intact V-type ATPases reveals bound lipids and the effects of nucleotide binding. Science 334, 380–385 (2011).
Newby, Z.E. et al. A general protocol for the crystallization of membrane proteins for X-ray structural investigation. Nat. Protoc. 4, 619–637 (2009).
Drew, D., Lerch, M., Kunji, E., Slotboom, D.J. & de Gier, J.W. Optimization of membrane protein overexpression and purification using GFP fusions. Nat. Methods 3, 303–313 (2006).
Drew, D. et al. GFP-based optimization scheme for the overexpression and purification of eukaryotic membrane proteins in Saccharomyces cerevisiae. Nat. Protoc. 3, 784–798 (2008).
Sonoda, Y. et al. Benchmarking membrane protein detergent stability for improving throughput of high-resolution X-ray structures. Structure 19, 17–25 (2011).
Seddon, A.M., Curnow, P. & Booth, P.J. Membrane proteins, lipids and detergents: not just a soap opera. Biochim. Biophys. Acta. 1666, 105–117 (2004).
Jurchen, J.C. & Williams, E.R. Origin of asymmetric charge partitioning in the dissociation of gas-phase protein homodimers. J. Am. Chem. Soc. 125, 2817–2826 (2003).
Light-Wahl, K.J., Schwartz, B.L. & Smith, R.D. Observation of the noncovalent quaternary associations of proteins by electrospray ionization mass spectrometry. J. Am. Chem. Soc. 116, 5271–5278 (1994).
Benesch, J.L. & Robinson, C.V. Mass spectrometry of macromolecular assemblies: preservation and dissociation. Curr. Opin. Struct. Biol. 16, 245–251 (2006).
Ruotolo, B.T., Benesch, J.L., Sandercock, A.M., Hyung, S.J. & Robinson, C.V. Ion mobility-mass spectrometry analysis of large protein complexes. Nat. Protoc. 3, 1139–1152 (2008).
Wang, S.C. et al. Ion mobility mass spectrometry of two tetrameric membrane protein complexes reveals compact structures and differences in stability and packing. J. Am. Chem. Soc. 132, 15468–15470 (2010).
Strop, P. & Brunger, A.T. Refractive index-based determination of detergent concentration and its application to the study of membrane proteins. Protein Sci. 14, 2207–2211 (2005).
Khademi, S. et al. Mechanism of ammonia transport by Amt/MEP/Rh: structure of AmtB at 1.35 A. Science 305, 1587–1594 (2004).
Hernandez, H. & Robinson, C.V. Determining the stoichiometry and interactions of macromolecular assemblies from mass spectrometry. Nat. Protoc. 2, 715–726 (2007).
Sobott, F., Hernandez, H., McCammon, M.G., Tito, M.A. & Robinson, C.V. A tandem mass spectrometer for improved transmission and analysis of large macromolecular assemblies. Anal. Chem. 74, 1402–1407 (2002).
Pedelacq, J.D., Cabantous, S., Tran, T., Terwilliger, T.C. & Waldo, G.S. Engineering and characterization of a superfolder green fluorescent protein. Nat. Biotechnol. 24, 79–88 (2006).
Kirshenbaum, N., Michaelevski, I. & Sharon, M. Analyzing large protein complexes by structural mass spectrometry. J. Vis. Exp. doi:10.3791/1954 (19 June 2010).
Hilf, R.J. & Dutzler, R. X-ray structure of a prokaryotic pentameric ligand-gated ion channel. Nature 452, 375–379 (2008).
Hilf, R.J. et al. Structural basis of open channel block in a prokaryotic pentameric ligand-gated ion channel. Nat. Struct. Mol. Biol. 17, 1330–1336 (2010).
Bantscheff, M., Schirle, M., Sweetman, G., Rick, J. & Kuster, B. Quantitative mass spectrometry in proteomics: a critical review. Anal. Bioanal. Chem. 389, 1017–1031 (2007).
Acknowledgements
We thank R. Dutzler and I. Zimmerman (University of Zurich) for the ELIC expression plasmid, and D. Rees and C. Gandhi (California Institute of Technology) for the oligomer characterization by addition of mass (OCAM) constructs. The Medical Research Council, European Research Council (ERC) IMPRESS and the Wellcome Trust are acknowledged for funding. A.L. is a Nicholas Kurti Junior Research Fellow of Brasenose College, Oxford and C.V.R. is a Royal Society Professor.
Author information
Authors and Affiliations
Contributions
A.L., E.R., J.T.S.H. and C.V.R. designed the project. A.L. and E.R. performed all experiments, with the exception of the mass spectrometry optimization experiments using ELIC performed by J.T.S.H. A.L. and E.R. prepared the figures. A.L. and C.V.R. wrote the manuscript and coordinated contributions by other authors.
Corresponding author
Ethics declarations
Competing interests
University of Oxford has filed a provisional patent on mass spectrometry mediated drug discovery for membrane proteins using polyoxyethylene glycol detergents.
Supplementary information
Supplementary Methods
Membrane protein expression and purification (PDF 327 kb)
Supplementary Figure 1
Mass spectra dilution series of Ammonium channel, AmtB Purified AmtB in DDM was diluted in Mem MS Buffer containing 2× CMC DDM. Protein concentration of the trimeric complex was determined using a BCA Protein Assay Kit (Pierce, Thermo Scientific) and shown in the upper right corner of each mass spectrum. These results highlight the sensitivity of mass spectrometry. (PDF 571 kb)
Supplementary Figure 2
Mass spectra of the pentameric ligand-gated ion channel from Erwinia chrysanthemi (ELIC) at various purification steps. ELIC was expressed as a HRV3C protease cleavable N-terminal fusion to a maltose binding protein preceded by a secretion signal peptide (pelB) and 10× His-tag22. (a) Mass spectrum of ELIC purified by IMAC and gel filtration chromatography. Peaks are clearly resolved for the pentameric complex and disassociation products poorly resolved. (b) Mass spectrum of ELIC after HRV3C protease treatment and reverse IMAC. Fusion removal has led to increased disassociation products along with resolved spectra all species. (PDF 504 kb)
Supplementary Figure 3
The preparation of lipids can influence membrane protein-lipid interactions. ELIC was titrated with POPE from various preparations to a final concentration of 12.5 μg/mL. Comparison of desodiated (top, purple spectra) and sodiated (middle, blue spectra) lipid preparations prepared as described in BOX 3, and incorrectly using organic solvent, chloroform (bottom, brown spectra). At this resolution desodiated and sodiated lipid preparations are comparable. In contrast, significantly less bound POPE is observed when lipid stocks in chloroform are used. (PDF 339 kb)
Rights and permissions
About this article
Cite this article
Laganowsky, A., Reading, E., Hopper, J. et al. Mass spectrometry of intact membrane protein complexes. Nat Protoc 8, 639–651 (2013). https://doi.org/10.1038/nprot.2013.024
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.024
This article is cited by
-
Regulation of membrane protein structure and function by their lipid nano-environment
Nature Reviews Molecular Cell Biology (2023)
-
Structure and dynamics of endogenous cardiac troponin complex in human heart tissue captured by native nanoproteomics
Nature Communications (2023)
-
Polyamine detergents tailored for native mass spectrometry studies of membrane proteins
Nature Communications (2023)
-
Initial recommendations for performing, benchmarking and reporting single-cell proteomics experiments
Nature Methods (2023)
-
Emergence of mass spectrometry detergents for membrane proteomics
Analytical and Bioanalytical Chemistry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.