Abstract
Objective
To investigate the role of environmental factor—temperature in the regulation of aging process by unc-13 and sbt-1 in Caenorhabditis elegans.
Methods
The lifespan, the speed of pharynx pumping, and the intestinal autofluorescence of unc-13 and sbt-1 mutants were examined at different temperature conditions. In addition, to exclude the possible influences from other factors in unc-13 and sbt-1 mutants, the dauer formation, the thermotaxis, the brood size and the population percentage of the mutants expressing hsp16.2-gfp were further investigated.
Results
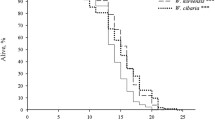
Mutations of unc-13 and sbt-1 significantly increased the mean and the maximum lifespans of nematodes cultured at 20 °C and 25 °C, while no noticeable increase was found at 15 °C in either the mean or the maximum lifespan. Investigations on the speed of pharynx pumping and the intestinal autofluorescence suggested that at 20 °C and 25 °C, mutations of unc-13 and sbt-1 could slow the aging process and delay the accumulation of aging-related cellular damage. Meanwhile, mutations of unc-13 or sbt-1 did not affect the dauer formation or the thermotaxis to different temperatures in nematodes. In contrast, at 20 °C and 25 °C conditions, mutations of unc-13 and sbt-1 significantly decreased the brood size and the percentage of nematodes expressing hsp16.2-gfp, while no such differences were detected at 15 °C. Moreover, the thermotolerance of unc-13 and sbt-1 mutants could be greatly strengthened after the 16-h heat shock at 35 °C.
Conclusion
The regulation of aging by unc-13 and sbt-1 is temperaturedependent. And the alterations in reproduction capability and stress response may be associated with the formation of this temperature-dependent property.
摘要
目的
研究环境因素—温度对秀丽线虫 unc-13 和 sbt-1 基因调控衰老作用的影响。
方法
在不同培养温度下,测定 unc-13 和 sbt-1 突变线虫的寿命、 咽泵运动速率和肠道荧光。 此外, 为排除其他可能的影响因素, 对 unc-13 和 sbt-1 突变体的永久性幼虫形成率、 对培养温度的趋向性、 子代数目以及表达 hsp16.2-gfp 线虫在群体中的比例进行了测定。
结果
在20 °C 和25 °C 培养条件下, unc-13 和 sbt-1 突变能显著提高线虫的平均寿命和最高寿命; 然而在15 °C 条件下, unc-13 和 sbt-1 突变体的平均寿命和最高寿命与野生型相似。 咽泵运动速率和肠道脂褐质自发荧光分析进一步验证了在20 °C 和25 °C 培养条件下, unc-13 和 sbt-1 突变可以减慢线虫衰老或延缓衰老相关的细胞损伤累积。 同时, unc-13 和 sbt-1 突变不影响永久性幼虫发生率以及动物对不同温度的趋向性。 相反, 在20 °C和25 °C 培养条件下, unc-13 和 sbt-1 突变能显著降低秀丽线虫的子代数目和群体中表达 hsp16.2-gfp 的线虫比率, 而在15 °C 条件下未出现此种差异。 此外, unc-13 和 sbt-1 突变体经35 °C 热激16 h 后, 其耐热性得到增强。
结论
unc-13 和 sbt-1 对衰老的调节具有温度依赖性。 unc-13 和 sbt-1 突变体的生殖功能和应激反应的改变可能与此温度依赖性的形成有关。
Similar content being viewed by others
References
Braeckman BP, Vanfleteren JR. Genetic control of longevity in C. elegans. Exp Gerontol 2007, 42: 90–98.
Johnson TE. Advantages and disadvantages of Caenorhabditis elegans for aging research. Exp Gerontol 2003, 38: 1329–1332.
Wolkow CA, Kimura KD, Lee M, Ruvkun G. Regulation of C. elegans life-span by insulinlike signaling in the nervous system. Science 2000, 290: 147–150.
Libina N, Berman JR, Kenyon C. Tissue-specific activities of C. elegans DAF-16 in the regulation of lifespan. Cell 2003, 115: 489–502.
Apfeld J, Kenyon C. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature 1999, 402: 804–809.
Mukhopadhyay A, Deplancke B, Walhout AJM, Tissenbaum HA. C. elegans tubby regulates life span and fat storage by two independent mechanisms. Cell Metab 2005, 2: 35–42.
Tissenbaum HA, Hawdon J, Perregaux M, Hotez P, Guarente L, Ruvkun G. A common muscarinic pathway for diapause recovery in the distantly related nematode species Caenorhabditis elegans and Ancylostoma caninum. Proc Natl Acad Sci U S A 2000, 97: 460–465.
Ailion M, Inoue T, Weaver CI, Holdcraft RW, Thomas JH. Neurosecretory control of aging in Caenorhabditis elegans. Proc Natl Acad Sci U S A 1999, 96: 7394–7397.
Shen LL, Wang Y, Wang DY. Involvement of genes required for synaptic function in aging control in C. elegans. Neurosci Bull 2007, 23: 21–29.
Brenner S. The genetics of Caenorhabditis elegans. Genetics 1974, 77: 71–94.
Donkin S, Williams PL. Influence of developmental stage, salts and food presence on various end points using Caenorhabditis elegans for aquatic toxicity testing. Environ Toxicol Chem 1995, 14: 2139–2147.
Rui Q, Lu Q, Wang DY. Administration of Bushenkangshuai Tang alleviates the UV irradiation- and oxidative stress-induced lifespan defects in nematode Caenorhabditis elegans. Front Med China 2009, 3: 76–90.
Huang C, Xiong C, Kornfeld K. Measurements of age-related changes of physiological processes that predict lifespan of Caenorhabditis elegans. Proc Natl Acad Sci U S A 2004, 101: 8084–8089.
Wilson MA, Shukitt-Hale B, Kalt W, Ingram DK, Joseph JA, Wolkow CA. Blueberry polyphenols increase lifespan and thermotolerance in Caenorhabditis elegans. Aging Cell 2006, 5: 59–68.
Garigan D, Hsu AL, Fraser AG, Kamath RS, Ahringer J, Kenyon C. Genetic analysis of tissue aging in Caenorhabditis elegans: a role for heat-shock factor and bacterial proliferation. Genetics 2002, 161: 1101–1112.
Wang DY, Shen LL, Wang Y. The phenotypic and behavioral defects can be transferred from zinc exposed nematodes to their progeny. Environ Toxicol Pharmacol 2007, 24: 223–230.
Wang DY, Wang Y. Phenotypic and behavioral defects caused by barium exposure in nematode Caenorhabditis elegans. Arch Environ Contam Toxicol 2008, 54: 447–453.
Gomez M, de Castro E, Guarin E, Sasakura H, Kuhara A, Mori I, et al. Ca2+ signaling via the neuronal calcium sensor-1 regulates associative learning and memory in C. elegans. Neuron 2001, 30: 241–248.
Ye HY, Ye BP, Wang DY. Trace administration of vitamin E can retrieve and prevent UV-irradiation- and metal exposureinduced memory deficits in nematode Caenorhabditis elegans. Neurobiol Learn Mem 2008, 90: 10–18.
Sampayo JN, Olsen A, Lithgow GJ. Oxidative stress in Caenorhabditis elegans: protective effects of superoxide dismutase/catalase mimetics. Aging Cell 2003, 2: 319–326.
Gems D, Sutton AJ, Sundermeyer ML, Albert PS, King KV, Eddley ML, et al. Two pleiotropic classes of daf-2 mutations affect larval arrest, adult behavior, reproduction and longevity in Caenorhabditis elegans. Genetics 1998, 150: 129–155.
Ishii N, Fujii M, Hartman PS, Tsuda M, Yasuda K, Senoo-Matsuda N, et al. A mutation in succinate dehydrogenase cytochrome b causes oxidative stress and ageing in nematodes. Nature 1998, 394: 694–697.
Wang Y, Xie W, Wang DY. Transferable properties of multibiological toxicity caused by cobalt exposure in Caenorhabditis elegans. Environ Toxicol Chem 2007, 26: 2405–2412.
Lithgow GJ, White TM, Melov S, Johnson TE. Thermotolerance and extended life span conferred by single-gene mutation and induced by thermal stress. Proc Natl Acad Sci U S A 1995, 92: 7540–7544.
Richmond JE, Davis WS, Jorgensen EM. UNC-13 is required for synaptic vesicle fusion in C. elegans. Nat Neurosci 1999, 2: 959–964.
Sieburth D, Ch’ng Q, Dybbs M, Tavazoie M, Kennedy S, Wang D, et al. Systematic analysis of genes required for synapse structure and function. Nature 2005, 436: 510–517.
Hammarlund M, Palfreyman MT, Watanabe S, Olsen S, Jorgensen EM. Open syntaxin docks synaptic vesicles. PLoS Biol 2007, 5: e198.
McEwen JM, Madison JM, Dybbs M, Kaplan JM. Antagonistic regulation of synaptic vesicle priming by Tomosyn and UNC-13. Neuron 51: 303–315.
Tissenbaum HA, Ruvkun G. An insulin-like signaling pathway affects both longevity and reproduction in Caenorhabditis elegans. Genetics 1998, 148: 703–717.
Johnson TE, de Castro E, Hegi de Castro S, Cypser J, Henderson S, Tedesco P. Relationship between longevity and stress resistance as assessed through gerontogene mutat ions in Caenorhabditis elegans. Exp Gerontol 2001, 36: 1609–1617.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contribute equally to this work
Rights and permissions
About this article
Cite this article
He, KW., Shen, LL., Zhou, WW. et al. Regulation of aging by unc-13 and sbt-1 in Caenorhabditis elegans is temperature-dependent. Neurosci. Bull. 25, 335–342 (2009). https://doi.org/10.1007/s12264-009-6123-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12264-009-6123-2