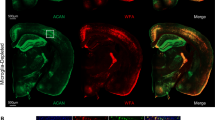
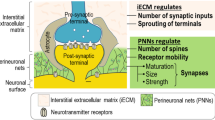
Abstract
The integrity of the central nervous system (CNS) matrix is crucial for its proper function. Loss of the lattice-like structure compromise synaptic stability and can lead to the disruption of the excitatory/inhibitory balance, astrocytosis, maladaptive plasticity and neuronal death. Perineuronal nets (PNNs) in the extracellular matrix (ECM) provide synaptic integration and control the functional wiring between neurons. These nets are significantly modified during CNS disorders, such as neurodegenerative, cerebrovascular and inflammatory diseases. The breakdown or the modification of PNNs could be due to the activity of matrix metalloproteinases (MMPs) or to the deposition of proteoglycans, glycoproteins, and hyaluronic acid. The expression and the activity of ECM-degrading enzymes can be regulated with tissue inhibitors of MMPs or via transcriptional and epigenetic silencing or enhancement (i.e. via histone deacetylases). The identification of molecules and mechanisms able to modify these processes will be essential for a new perspective on brain functioning in health and disease, leading to a target-directed approach with drugs directly interfering with the molecular mechanism underlying neurological disorders.


Similar content being viewed by others
References
Dityatev A, Rusakov DA (2011) Molecular signals of plasticity at the tetrapartite synapse. Curr Opin Neurobiol 21(2):353–359
De Luca C, Savarese L, Colangelo AM, Bianco MR, Cirillo G et al (2016) Astrocytes and microglia-mediated immune response in maladaptive plasticity is differently modulated by NGF in the ventral horn of the spinal cord following peripheral nerve injury. Cell Mol Neurobiol 36(1):37–46.
Dityatev A, Schachner M (2003) Extracellular matrix molecules and synaptic plasticity. Nat Rev Neurosci 4(6):456–468
Papa M, De Luca C, Petta F, Alberghina L, Cirillo G (2014) Astrocyte-neuron interplay in maladaptive plasticity. Neurosci Biobehav Rev 42:35–54
Galtrey CM, Kwok JC, Carulli D, Rhodes KE, Fawcett JW (2008) Distribution and synthesis of extracellular matrix proteoglycans, hyaluronan, link proteins and tenascin-R in the rat spinal cord. Eur J Neurosci 27(6):1373–1390
Wang D, Fawcett J (2012) The perineuronal net and the control of CNS plasticity. Cell Tissue Res 349(1):147–160
Yong VW, Power C, Forsyth P, Edwards DR (2001) Metalloproteinases in biology and pathology of the nervous system. Nat Rev Neurosci 2(7):502–511
Wang D, Ichiyama RM, Zhao R, Andrews MR, Fawcett JW (2011) Chondroitinase combined with rehabilitation promotes recovery of forelimb function in rats with chronic spinal cord injury. J Neurosci 31(25):9332–9344
Gogolla N, Caroni P, Luthi A, Herry C (2009) Perineuronal nets protect fear memories from erasure. Science 325(5945):1258–1261
Cirillo G, Colangelo AM, Bianco MR, Cavaliere C, Zaccaro L et al (2012) BB14, a Nerve Growth Factor (NGF)-like peptide shown to be effective in reducing reactive astrogliosis and restoring synaptic homeostasis in a rat model of peripheral nerve injury. Biotechnol Adv 30(1):223–232
Soleman S, Filippov MA, Dityatev A, Fawcett JW (2013) Targeting the neural extracellular matrix in neurological disorders. Neuroscience 253:194–213
Pollock E, Everest M, Brown A, Poulter MO (2014) Metalloproteinase inhibition prevents inhibitory synapse reorganization and seizure genesis. Neurobiol Dis 70:21–31
Zhang Q, Li J, Liu C, Song C, Li P et al (2015) Protective effects of low molecular weight chondroitin sulfate on amyloid beta (Aβ)-induced damage in vitro and in vivo. Neuroscience 305:169–182
Zinnhardt B, Viel T, Wachsmuth L, Vrachimis A, Wagner S et al (2015) Multimodal imaging reveals temporal and spatial microglia and matrix metalloproteinase activity after experimental stroke. J Cereb Blood Flow Metab 35:1711–1721
Alpár A, Gärtner U, Härtig W, Brückner G (2006) Distribution of pyramidal cells associated with perineuronal nets in the neocortex of rat. Brain Res 1120(1):13–22
Corvetti L, Rossi F (2005) Degradation of chondroitin sulfate proteoglycans induces sprouting of intact purkinje axons in the cerebellum of the adult rat. J Neurosci 25(31):7150–7158
Frischknecht R, Heine M, Perrais D, Seidenbecher CI, Choquet D et al (2009) Brain extracellular matrix affects AMPA receptor lateral mobility and short-term synaptic plasticity. Nat Neurosci 12(7):897–904
Carulli D, Pizzorusso T, Kwok JCF, Putignano E, Poli A et al (2010) Animals lacking link protein have attenuated perineuronal nets and persistent plasticity. Brain 133(8):2331–2347
Golgi C (1893) Intorno all’origine del quarto nervo cerebrale e una questione isto-fisiologica che a questo argomento si collega. Rendiconti della Reale Accademia Dei Lincei 2:379–389
Liu H, Xu H, Yu T, Yao J, Zhao C et al (2013) Expression of perineuronal nets, parvalbumin and protein tyrosine phosphatase sigma in the rat visual cortex during development and after BFD. Curr Eye Res 38(10):1083–1094
Deepa SS, Carulli D, Galtrey C, Rhodes K, Fukuda J et al (2006) Composition of perineuronal net extracellular matrix in rat brain: a different disaccharide composition for the net-associated proteoglycans. J Biol Chem 281(26):17789–17800
Carulli D, Rhodes KE, Brown DJ, Bonnert TP, Pollack SJ et al (2006) Composition of perineuronal nets in the adult rat cerebellum and the cellular origin of their components. J Comp Neurol 494(4):559–577
Rivera S, Khrestchatisky M, Kaczmarek L, Rosenberg G, Jaworski D (2010) Metzincin proteases and their inhibitors: Foes or friends in nervous system physiology? J Neurosci 30:15337–15357
Houghton AM, Hartzell WO, Robbins CS, Gomis-Ruth FX, Shapiro SD (2009) Macrophage elastase kills bacteria within murine macrophages. Nature 460(7255):637–641
Prox J, Arnold P, Becker-Pauly C (2015) Meprin α and meprin β: procollagen proteinases in health and disease. Matrix Biol 44–46:7–13
Vadon-Le Goff S, Hulmes DJS, Moali C (2015) BMP-1/tolloid-like proteinases synchronize matrix assembly with growth factor activation to promote morphogenesis and tissue remodeling. Matrix Biol 44–46:14–23
Ortolano S, Spuch C (2013) tPA in the Central Nervous System: Relations Between tPA and Cell Surface LRPs. Recent Pat Endocr Metab Immune Drug Discov 7(1):65–76
Rosenberg GA (2002) Matrix metalloproteinases and neuroinflammation in multiple sclerosis. Neuroscientist 8(6):586–595
Vargova V, Pytliak M, Mechirova V (2012) Matrix metalloproteinases. EXS 103:1–33
Murphy G (2011) Tissue inhibitors of metalloproteinases. Genome Biol 12(11):233
Chaudhary AK, Pandya S, Ghosh K, Nadkarni A (2013) Matrix metalloproteinase and its drug targets therapy in solid and hematological malignancies: an overview. Mutat Res, Rev Mutat Res 753(1):7–23
Berton A, Rigot V, Huet E, Decarme M, Eeckhout Y et al (2001) Involvement of fibronectin type II repeats in the efficient inhibition of gelatinases A and B by long-chain unsaturated fatty acids. J Biol Chem 276(23):20458–20465
Selvais C, D’Auria L, Tyteca D, Perrot G, Lemoine P et al (2011) Cell cholesterol modulates metalloproteinase-dependent shedding of low-density lipoprotein receptor-related protein-1 (LRP-1) and clearance function. FASEB J 25(8):2770–2781
Bovolenta P, Wandosell F, Nieto-Sampedro M (1993) Neurite outgrowth inhibitors associated with glial cells and glial cell lines. NeuroReport 5(3):345–348
Kwok JCF, Dick G, Wang D, Fawcett JW (2011) Extracellular matrix and perineuronal nets in CNS repair. Dev Neurobiol 71(11):1073–1089
Hill JJ, Jin K, Mao XO, Xie L, Greenberg DA (2012) Intracerebral chondroitinase ABC and heparan sulfate proteoglycan glypican improve outcome from chronic stroke in rats. Proc Natl Acad Sci 109(23):9155–9160
Brown JM, Xia J, Zhuang B, Cho K-S, Rogers CJ et al (2012) A sulfated carbohydrate epitope inhibits axon regeneration after injury. Proc Natl Acad Sci 109(13):4768–4773
Souvenir R, Fathali N, Ostrowski RP, Lekic T, Zhang JH et al (2011) Tissue inhibitor of matrix metalloproteinase-1 mediates erythropoietin-induced neuroprotection in hypoxia ischemia. Neurobiol Dis 44(1):28–37
Xu Y, Tian Y, Wei HJ, Chen J, Dong JF et al (2011) Erythropoietin increases circulating endothelial progenitor cells and reduces the formation and progression of cerebral aneurysm in rats. Neuroscience 181:292–299
Dong X, Song YN, Liu WG, Guo XL (2009) Mmp-9, a potential target for cerebral ischemic treatment. Curr Neuropharmacol 7(4):269–275
Zhan Y, Krafft PR, Lekic T, Ma Q, Souvenir R et al (2015) Imatinib preserves blood–brain barrier integrity following experimental subarachnoid hemorrhage in rats. J Neurosci Res 93(1):94–103
Romero JR, Vasan RS, Beiser AS, Au R, Benjamin EJ et al (2010) Association of matrix metalloproteinases with MRI indices of brain ischemia and aging. Neurobiol Aging 31(12):2128–2135
Yamada J, Jinno S (2013) Spatio-temporal differences in perineuronal net expression in the mouse hippocampus, with reference to parvalbumin. Neuroscience 253:368–379
Tanaka Y, Mizoguchi K (2009) Influence of aging on chondroitin sulfate proteoglycan expression and neural stem/progenitor cells in rat brain and improving effects of a herbal medicine, yokukansan. Neuroscience 164(3):1224–1234
Végh MJ, Rausell A, Loos M, Heldring CM, Jurkowski W et al (2014) Hippocampal extracellular matrix levels and stochasticity in synaptic protein expression increase with age and are associated with age-dependent cognitive decline. Mol Cell Proteom 13(11):2975–2985
Suttkus A, Rohn S, Weigel S, Glockner P, Arendt T et al (2014) Aggrecan, link protein and tenascin-R are essential components of the perineuronal net to protect neurons against iron-induced oxidative stress. Cell Death Dis 5:e1119
Fawcett JW (2015) The extracellular matrix in plasticity and regeneration after CNS injury and neurodegenerative disease. Prog Brain Res 218:213–226
Endres K, Fahrenholz F (2012) Regulation of alpha-secretase ADAM10 expression and activity. Exp Brain Res 217(3–4):343–352
Siller SS, Broadie K (2012) Matrix metalloproteinases and minocycline: therapeutic avenues for fragile X syndrome. Neural Plast 2012:9
Saftig P, Reiss K (2011) The “A Disintegrin And Metalloproteases” ADAM10 and ADAM17: Novel drug targets with therapeutic potential? Eur J Cell Biol 90(6–7):527–535
Cui H, Freeman C, Jacobson GA, Small DH (2013) Proteoglycans in the central nervous system: role in development, neural repair, and Alzheimer’s disease. IUBMB Life 65(2):108–120
Walzer M, Lorens S, Hejna M, Fareed J, Hanin I et al (2002) Low molecular weight glycosaminoglycan blockade of β-amyloid induced neuropathology. Eur J Pharmacol 445(3):211–220
Timmer NM, van Dijk L, van der Zee CE, Kiliaan A, de Waal RM, Verbeek MM (2010) Enoxaparin treatment administered at both early and late stages of amyloid β deposition improves cognition of APPswe/PS1dE9 mice with differential effects on brain Aβ levels. Neurobiol Dis 40(1):340–347
Holmes BB, DeVos SL, Kfoury N, Li M, Jacks R et al (2013) Heparan sulfate proteoglycans mediate internalization and propagation of specific proteopathic seeds. Proc Natl Acad Sci 110(33):E3138–E3147
Zhang X, Wang B, O’Callaghan P, Hjertström E, Jia J et al (2012) Heparanase overexpression impairs inflammatory response and macrophage-mediated clearance of amyloid-β in murine brain. Acta Neuropathol 124(4):465–478
McRae PA, Porter BE (2012) The perineuronal net component of the extracellular matrix in plasticity and epilepsy. Neurochem Int 61(7):963–972
Wilczynski GM, Konopacki FA, Wilczek E, Lasiecka Z, Gorlewicz A et al (2008) Important role of matrix metalloproteinase 9 in epileptogenesis. J Cell Biol 180(5):1021–1035
Park H, Poo MM (2013) Neurotrophin regulation of neural circuit development and function. Nat Rev Neurosci 14(1):7–23
Mizoguchi H, Yamada K, Nabeshima T (2011) Matrix metalloproteinases contribute to neuronal dysfunction in animal models of drug dependence, Alzheimer’s disease, and epilepsy. Biochem Res Int 2011:681385
Nagappan G, Zaitsev E, Senatorov VV, Yang J, Hempstead BL et al (2009) Control of extracellular cleavage of ProBDNF by high frequency neuronal activity. Proc Natl Acad Sci 106(4):1267–1272
Maggio N, Cavaliere C, Papa M, Blatt I, Chapman J et al (2013) Thrombin regulation of synaptic transmission: implications for seizure onset. Neurobiol Dis 50:171–178
Van den Oever MC, Lubbers BR, Goriounova NA, Li KW, Van der Schors RC et al (2010) Extracellular matrix plasticity and GABAergic inhibition of prefrontal cortex pyramidal cells facilitates relapse to heroin seeking. Neuropsychopharmacology 35(10):2120–2133
Nagai T, Yamada K, Yoshimura M, Ishikawa K, Miyamoto Y et al (2004) The tissue plasminogen activator-plasmin system participates in the rewarding effect of morphine by regulating dopamine release. Proc Natl Acad Sci USA 101(10):3650–3655
Nakamoto K, Kawasaki S, Kobori T, Fujita-Hamabe W, Mizoguchi H et al (2012) Involvement of matrix metalloproteinase-9 in the development of morphine tolerance. Eur J Pharmacol 683(1–3):86–92
Bahi A, Dreyer JL (2008) Overexpression of plasminogen activators in the nucleus accumbens enhances cocaine-, amphetamine- and morphine-induced reward and behavioral sensitization. Genes Brain Behav 7(2):244–256
Bahi A, Dreyer J-L (2012) Involvement of tissue plasminogen activator “tPA” in ethanol-induced locomotor sensitization and conditioned-place preference. Behav Brain Res 226(1):250–258
Mizoguchi H, Yamada K, Niwa M, Mouri A, Mizuno T et al (2007) Reduction of methamphetamine-induced sensitization and reward in matrix metalloproteinase-2 and -9-deficient mice. J Neurochem 100(6):1579–1588
Mauney SA, Athanas KM, Pantazopoulos H, Shaskan N, Passeri E et al (2013) Developmental pattern of perineuronal nets in the human prefrontal cortex and their deficit in schizophrenia. Biol Psychiatry 74(6):427–435
Shah A, Lodge DJ (2013) A loss of hippocampal perineuronal nets produces deficits in dopamine system function: relevance to the positive symptoms of schizophrenia. Transl Psychiatry 3:e215
Morawski M, Dityatev A, Hartlage-Rübsamen M, Blosa M, Holzer M et al (2014) Tenascin-R promotes assembly of the extracellular matrix of perineuronal nets via clustering of aggrecan. Philos Trans R Soc Lond B Biol Sci 369(1654):20140046
Takamatsu A, Ohkawara B, Ito M, Masuda A, Sakai T et al (2014) Verapamil protects against cartilage degradation in osteoarthritis by inhibiting Wnt/β-catenin signaling. PLoS ONE 9(3):e92699
Moore CS, Crocker SJ (2012) An alternate perspective on the roles of TIMPs and MMPs in pathology. Am J Pathol 180(1):12–16
Ulrich R, Baumgärtner W, Gerhauser I, Seeliger F, Haist V et al (2006) MMP-12, MMP-3, and TIMP-1 are markedly upregulated in chronic demyelinating theiler murine encephalomyelitis. J Neuropathol Exp Neurol 65(8):783–793
Gur-Wahnon D, Mizrachi T, Maaravi-Pinto F-Y, Lourbopoulos A, Grigoriadis N et al (2013) The plasminogen activator system: involvement in central nervous system inflammation and a potential site for therapeutic intervention. J Neuroinflamm 10(1):124
Candelario-Jalil E, Yang Y, Rosenberg GA (2009) Diverse roles of matrix metalloproteinases and tissue inhibitors of metalloproteinases in neuroinflammation and cerebral ischemia. Neuroscience 158(3):983–994
Verslegers M, Lemmens K, Van Hove I, Moons L (2013) Matrix metalloproteinase-2 and -9 as promising benefactors in development, plasticity and repair of the nervous system. Prog Neurobiol 105:60–78
Thorne M, Moore CS, Robertson GS (2009) Lack of TIMP-1 increases severity of experimental autoimmune encephalomyelitis: EFFECTS of darbepoetin alfa on TIMP-1 null and wild-type mice. J Neuroimmunol 211(1):92–100
Feng S, Cen J, Huang Y, Shen H, Yao L et al (2011) Matrix metalloproteinase-2 and -9 secreted by leukemic cells increase the permeability of blood-brain barrier by disrupting tight junction proteins. PLoS ONE 6(8):e20599
Bourboulia D, Stetler-Stevenson WG (2010) Matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs): positive and negative regulators in tumor cell adhesion. Semin Cancer Biol 20(3):161–168
Iyer RP, Patterson NL, Fields GB, Lindsey ML (2012) The history of matrix metalloproteinases: milestones, myths, and misperceptions. Am J Physiol Heart Circ Physiol 303(8):H919–H930
Stanton H, Melrose J, Little CB (1812) Fosang AJ (2011) Proteoglycan degradation by the ADAMTS family of proteinases. Biochimica et Biophysica Acta (BBA) Mol Basis Dis 12:1616–1629
Levičar N, Nutall RK, Lah TT (2003) Proteases in brain tumour progression. Acta Neurochir 145(9):825–838
Rahmah N, Sakai K, Sano K, Hongo K (2012) Expression of RECK in endothelial cells of glioma: comparison with CD34 and VEGF expressions. J Neuro Oncol 107(3):559–564
Jayanth P, Amith SR, Gee K, Szewczuk MR (2010) Neu1 sialidase and matrix metalloproteinase-9 cross-talk is essential for neurotrophin activation of Trk receptors and cellular signaling. Cell Signal 22(8):1193–1205
Blanco-Mezquita T, Martinez-Garcia C, Proenca R, Zieske JD, Bonini S et al (2013) Nerve growth factor promotes corneal epithelial migration by enhancing expression of matrix metalloprotease-9. Invest Ophthalmol Vis Sci 54(6):3880–3890
Bruno MA, Cuello AC (2006) Activity-dependent release of precursor nerve growth factor, conversion to mature nerve growth factor, and its degradation by a protease cascade. Proc Natl Acad Sci USA 103(17):6735–6740
Romi F, Helgeland G, Gilhus NE (2012) Serum levels of matrix metalloproteinases: implications in clinical neurology. Eur Neurol 67(2):121–128
Casha S, Zygun D, McGowan MD, Bains I, Yong VW et al (2012) Results of a phase II placebo-controlled randomized trial of minocycline in acute spinal cord injury. Brain 135(Pt 4):1224–1236
Tian L, Prabhakaran MP, Ramakrishna S (2015) Strategies for regeneration of components of nervous system: scaffolds, cells and biomolecules. Regen Biomater 2(1):31–45
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
De Luca, C., Papa, M. Looking Inside the Matrix: Perineuronal Nets in Plasticity, Maladaptive Plasticity and Neurological Disorders. Neurochem Res 41, 1507–1515 (2016). https://doi.org/10.1007/s11064-016-1876-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-1876-2