Abstract
Background
Coix seed oil (CSO) has a wide range of anticancer effects. However, the mechanism of action against pancreatic cancer (PC) and regulation of mitochondria in vitro is still unclear.
Materials and results
This research investigated the possible mechanism of CSO induction of PC cell apoptosis and regulating mitochondrial functional damage. Proliferation of PC cells, mitochondrial membrane potential (MMP), qualitative and quantitative analysis of PC cell apoptosis, openness of mitochondrial permeability transition pore, related protein expression, generation of reactive oxygen species (ROS), and gene expression were determined by cell counting kit-8, JC-1 staining, acridine orange and ethidium bromide staining, flow cytometry, calcein-AM/cobalt staining, western blotting, dichlorofluorescein diacetate probe, and quantitative real-time reverse transcription-polymerase chain reaction, respectively. We confirmed that PTEN protein was involved in CSO-induced PANC-1 cell apoptosis and mitochondrial functional damage. CSO induced depolarization of MMP, increased opening of the mitochondrial permeability transition pore, increased ROS production, and further increased mitochondrial damage. Additionally, CSO downregulated expression of p-AKT and p-PI3K proteins; upregulated protein expression of cleaved caspase-9, Bax, cleaved caspase-3 and cytochrome c; and downregulated expression of Bcl-2 by upregulating the PTEN gene. The corresponding protein expression was consistent with the gene expression level. Furthermore, the loss of function of PTEN protein reduces the ability of CSO to induce apoptosis of PANC-1 cells and damage to mitochondrial function.
Conclusions
CSO induces apoptosis of PANC-1 PC cells by modulating mitochondrial functional impairment and related apoptotic molecules via PTEN, which may be closely related to the PI3K/AKT signaling pathway.
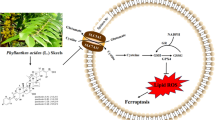
Graphical abstract







Similar content being viewed by others
Data Availability
All datasets for the analysis in the present study are available upon reasonable request to the corresponding author.
References
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70(1):7–30. https://doi.org/10.3322/caac.21590
Panji M, Behmard V, Zare Z, Malekpour M, Nejadbiglari H, Yavari S, Nayerpour Dizaj T, Safaeian A, Maleki N, Abbasi M, Abazari O, Shabanzadeh M, Khanicheragh P (2021) Suppressing effects of green tea extract and Epigallocatechin-3-gallate (EGCG) on TGF-β- induced Epithelial-to-mesenchymal transition via ROS/Smad signaling in human cervical cancer cells. Gene 794:145774. https://doi.org/10.1016/j.gene.2021.145774
Rana P, Shrama A, Mandal CC (2021) Molecular insights into phytochemicals-driven break function in tumor microenvironment. J Food Biochem 45(9):e13824. https://doi.org/10.1111/jfbc.13824
Zhan YP, Huang XE, Cao J, Lu YY, Wu XY, Liu J, Xu X, Xiang J, Ye LH (2012) Clinical safety and efficacy of Kanglaite® (Coix Seed Oil) injection combined with chemotherapy in treating patients with gastric cancer. Asian Pac J Cancer Prev 13(10):5319–5321. https://doi.org/10.7314/apjcp.2012.13.10.5319
Mao W, Fan Y, Cheng C, Yuan X, Lan T, Mao K, Wang J (2020) Efficacy and safety of Kanglaite injection combined with chemotherapy for colorectal cancer: A protocol for systematic review and meta-analysis. Med (Baltim) 99(39):e22357. https://doi.org/10.1097/md.0000000000022357
Liu J, Yu L, Ding W (2019) Efficacy and safety of Kanglaite injection combined with radiochemotherapy in the treatment of advanced pancreatic cancer: A PRISMA-compliant meta-analysis. Med (Baltim) 98(32):e16656. https://doi.org/10.1097/md.0000000000016656
Liu J, Liu X, Ma J, Li K, Xu C (2019) The clinical efficacy and safety of kanglaite adjuvant therapy in the treatment of advanced hepatocellular carcinoma: A PRISMA-compliant meta-analysis. Biosci Rep 39(11):23. https://doi.org/10.1042/bsr20193319
Wen J, Yang T, Wang J, Ma X, Tong Y, Zhao Y (2020) Kanglaite Injection Combined with Chemotherapy versus Chemotherapy Alone for the Improvement of Clinical Efficacy and Immune Function in Patients with Advanced Non-Small-Cell Lung Cancer: A Systematic Review and Meta-Analysis. Evid Based Complement Alternat Med 2020:8586596. https://doi.org/10.1155/2020/8586596
Falasca M, Selvaggi F, Buus R, Sulpizio S, Edling CE (2011) Targeting phosphoinositide 3-kinase pathways in pancreatic cancer–from molecular signalling to clinical trials. Anticancer Agents Med Chem 11(5):455–463. https://doi.org/10.2174/187152011795677382
Ebrahimi S, Hosseini M, Shahidsales S, Maftouh M, Ferns GA, Ghayour-Mobarhan M, Hassanian SM, Avan A (2017) Targeting the Akt/PI3K Signaling Pathway as a Potential Therapeutic Strategy for the Treatment of Pancreatic Cancer. Curr Med Chem 24(13):1321–1331. https://doi.org/10.2174/0929867324666170206142658
Abate M, Festa A, Falco M, Lombardi A, Luce A, Grimaldi A, Zappavigna S, Sperlongano P, Irace C, Caraglia M, Misso G (2020) Mitochondria as playmakers of apoptosis, autophagy and senescence. Semin Cell Dev Biol 98:139–153. https://doi.org/10.1016/j.semcdb.2019.05.022
Yang H, Villani RM, Wang H, Simpson MJ, Roberts MS, Tang M, Liang X (2018) The role of cellular reactive oxygen species in cancer chemotherapy. J Exp Clin Cancer Res 37(1):266. https://doi.org/10.1186/s13046-018-0909-x
Yen YP, Tsai KS, Chen YW, Huang CF, Yang RS, Liu SH (2012) Arsenic induces apoptosis in myoblasts through a reactive oxygen species-induced endoplasmic reticulum stress and mitochondrial dysfunction pathway. Arch Toxicol 86(6):923–933. https://doi.org/10.1007/s00204-012-0864-9
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Gunter TE, Pfeiffer DR (1990) Mechanisms by which mitochondria transport calcium. Am J Physiol 258(5 Pt 1):C755–C786. https://doi.org/10.1152/ajpcell.1990.258.5.C755
Zorov DB, Isaev NK, Plotnikov EY, Zorova LD, Stelmashook EV, Vasileva AK, Arkhangelskaya AA, Khrjapenkova TG (2007) The mitochondrion as janus bifrons. Biochem (Mosc) 72(10):1115–1126. https://doi.org/10.1134/s0006297907100094
Zamzami N, Marchetti P, Castedo M, Decaudin D, Macho A, Hirsch T, Susin SA, Petit PX, Mignotte B, Kroemer G (1995) Sequential reduction of mitochondrial transmembrane potential and generation of reactive oxygen species in early programmed cell death. J Exp Med 182(2):367–377. https://doi.org/10.1084/jem.182.2.367
Kinnally KW, Peixoto PM, Ryu SY, Dejean LM (2011) Is mPTP the gatekeeper for necrosis, apoptosis, or both? Biochim Biophys Acta 1813(4):616–622. https://doi.org/10.1016/j.bbamcr.2010.09.013
Lindsay J, Esposti MD, Gilmore AP (2011) Bcl-2 proteins and mitochondria–specificity in membrane targeting for death. Biochim Biophys Acta 1813(4):532–539. https://doi.org/10.1016/j.bbamcr.2010.10.017
Peña-Blanco A, García-Sáez AJ (2018) Bax, Bak and beyond - mitochondrial performance in apoptosis. Febs j 285(3):416–431. https://doi.org/10.1111/febs.14186
Kulikov AV, Shilov ES, Mufazalov IA, Gogvadze V, Nedospasov SA, Zhivotovsky B (2012) Cytochrome c: the Achilles’ heel in apoptosis. Cell Mol Life Sci 69(11):1787–1797. https://doi.org/10.1007/s00018-011-0895-z
Creagh EM, Martin SJ (2001) Caspases: cellular demolition experts. Biochem Soc Trans 29(Pt 6):696–702. https://doi.org/10.1042/0300-5127:0290696
Rohlenova K, Neuzil J, Rohlena J (2016) The role of Her2 and other oncogenes of the PI3K/AKT pathway in mitochondria. Biol Chem 397(7):607–615. https://doi.org/10.1515/hsz-2016-0130
Liu Y, Zhang W, Wang XJ, Liu S (2014) Antitumor effect of Kanglaite® injection in human pancreatic cancer xenografts. BMC Complement Altern Med 14:228. https://doi.org/10.1186/1472-6882-14-228
Yang C, Hou A, Yu C, Dai L, Wang W, Zhang K, Shao H, Ma J, Xu W (2018) Kanglaite reverses multidrug resistance of HCC by inducing apoptosis and cell cycle arrest via PI3K/AKT pathway. Onco Targets Ther 11:983–996. https://doi.org/10.2147/ott.S153814
Fang T, Jiang YX, Chen L, Huang L, Tian XH, Zhou YD, Nagle DG, Zhang DD (2020) Coix Seed Oil Exerts an Anti-Triple-Negative Breast Cancer Effect by Disrupting miR-205/S1PR1 Axis. Front Pharmacol 11:529962. https://doi.org/10.3389/fphar.2020.529962
Liu Y, Sun XJ, Xiao Y, Liu S, Zhao J, Qin W (2019) PTEN is involved in Kanglaite (R) injection-induced apoptosis of human pancreatic cancer cells. Int J Clin Exp Med 12(2):1658–1665
Adamska A, Elaskalani O, Emmanouilidi A, Kim M, Abdol Razak NB, Metharom P, Falasca M (2018) Molecular and cellular mechanisms of chemoresistance in pancreatic cancer. Adv Biol Regul 68:77–87. https://doi.org/10.1016/j.jbior.2017.11.007
Zeng S, Pöttler M, Lan B, Grützmann R, Pilarsky C, Yang H (2019) Chemoresistance in Pancreatic Cancer. Int J Mol Sci 20(18):4504. https://doi.org/10.3390/ijms20184504
Liu S, Xu ZL, Sun L, Liu Y, Li CC, Li HM, Zhang W, Li CJ, Qin W (2016) (–)–Epigallocatechin–3–gallate induces apoptosis in human pancreatic cancer cells via PTEN. Mol Med Rep 14(1):599–605. https://doi.org/10.3892/mmr.2016.5277
Stambolic V, Suzuki A, de la Pompa JL, Brothers GM, Mirtsos C, Sasaki T, Ruland J, Penninger JM, Siderovski DP, Mak TW (1998) Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell 95(1):29–39. https://doi.org/10.1016/s0092-8674(00)81780-8
Lu Y, Li CS, Dong Q (2008) Chinese herb related molecules of cancer-cell-apoptosis: a minireview of progress between Kanglaite injection and related genes. J Exp Clin Cancer Res 27(1):31. https://doi.org/10.1186/1756-9966-27-31
Zhao YJ, Fan JP (2021) The Effect of Kanglaite Injection on Apoptosis of Colon Cancer SW480 Cells and Expression of Apoptosis-Related Factors. Pakistan J Zool 53(1):379–382. https://doi.org/10.17582/journal.pjz/20191119131142
Fu Y, Ricciardiello F, Yang G, Qiu J, Huang H, Xiao J, Cao Z, Zhao F, Liu Y, Luo W, Chen G, You L, Chiaradonna F, Zheng L, Zhang T (2021) The Role of Mitochondria in the Chemoresistance of Pancreatic Cancer Cells. Cells 10(3):497. https://doi.org/10.3390/cells10030497
Chipuk JE, Green DR (2008) How do BCL-2 proteins induce mitochondrial outer membrane permeabilization? Trends Cell Biol 18(4):157–164. https://doi.org/10.1016/j.tcb.2008.01.007
Danial NN (2007) BCL-2 family proteins: critical checkpoints of apoptotic cell death. Clin Cancer Res 13(24):7254–7263. https://doi.org/10.1158/1078-0432.Ccr-07-1598
Fadeel B, Orrenius S (2005) Apoptosis: a basic biological phenomenon with wide-ranging implications in human disease. J Intern Med 258(6):479–517. https://doi.org/10.1111/j.1365-2796.2005.01570.x
Yokoyama C, Sueyoshi Y, Ema M, Mori Y, Takaishi K, Hisatomi H (2017) Induction of oxidative stress by anticancer drugs in the presence and absence of cells. Oncol Lett 14(5):6066–6070. https://doi.org/10.3892/ol.2017.6931
Gorrini C, Harris IS, Mak TW (2013) Modulation of oxidative stress as an anticancer strategy. Nat Rev Drug Discov 12(12):931–947. https://doi.org/10.1038/nrd4002
Zorov DB, Juhaszova M, Sollott SJ (2014) Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev 94(3):909–950. https://doi.org/10.1152/physrev.00026.2013
Zorov DB, Filburn CR, Klotz LO, Zweier JL, Sollott SJ (2000) Reactive oxygen species (ROS)-induced ROS release: a new phenomenon accompanying induction of the mitochondrial permeability transition in cardiac myocytes. J Exp Med 192(7):1001–1014. https://doi.org/10.1084/jem.192.7.1001
Simon HU, Haj-Yehia A, Levi-Schaffer F (2000) Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 5(5):415–418. https://doi.org/10.1023/a:1009616228304
Acknowledgements
This study was supported by Qiqihar Academy of Medical Sciences (Grant Number QMSI2019M-12), and thanks to the Third Affiliated Hospital of Qiqihar Medical University for funding this research.
Author information
Authors and Affiliations
Contributions
JY: conceptualization, methodology, software, validation, and writing—original draft preparation; YL: formal analysis and resources; S-NL: investigation and data curation; X-JS: software; YY: investigation and data curation; K-FW: conceptualization, formal analysis, and supervision; SL: project administration, funding application, and writing—review & editing and supervision; All authors reviewed the paper and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no confict of interest.
Ethical approval
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, J., Liu, Y., Lu, S. et al. Coix seed oil regulates mitochondrial functional damage to induce apoptosis of human pancreatic cancer cells via the PTEN/PI3K/AKT signaling pathway. Mol Biol Rep 49, 5897–5909 (2022). https://doi.org/10.1007/s11033-022-07371-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07371-8