Abstract
Purpose
Does focal cavity radiotherapy after resection of brain metastasis “spare” whole-brain radiotherapy, which is associated with toxicity for patients, through the complete course of their disease without compromising long-term local control of the brain?
Methods
We retrospectively analyzed outcomes of patients who underwent adjuvant focal cavity radiotherapy between 2014 and 2021 at our center.
Results
A total of 83 patients with 86 resected brain metastases were analyzed. 64% had singular, 36% two to four brain metastases. In cases with multiple metastases, omitted lesions were treated with radiosurgery. Median follow-up was 7.3 months (range 0–71.2 months), 1‑year overall survival rate was 57.8% (95% CI 44.9–68.8%). Radiotherapy was administered with a median biologically effective dose (α/β 10) surrounding the planning target volume of 48 Gy (range 23.4–60 Gy). Estimated 1‑year local control rate was 82.7% (95% CI 67.7–91.2%), estimated 1‑year distant brain control rate was 55.7% (95% CI 40.5–68.4%), estimated 1‑year leptomeningeal disease rate was 16.0% (95% CI 7.3–32.9%). Eleven distant brain recurrences could be salvaged with radiosurgery. In the further course of disease, 14 patients (17%) developed disseminated metastatic disease in the brain. Estimated 1‑year free of whole-brain radiotherapy rate was 72.3% (95% CI 57.1–82.9%). All applied treatments led to an estimated 1‑year neuro-control rate of 79.1% (95% CI 65.0–88.0%), estimated 1‑year radionecrosis rate was 23% (95% CI 12.4–40.5%).
Conclusion
In our single-center study, focal cavity radiotherapy was associated with high local control. In three out of four patients, whole-brain radiotherapy could be avoided in the complete course of disease, using radiosurgery as salvage approach without compromising neuro-control.
Similar content being viewed by others
Introduction
Neurosurgical resection is standard of care for patients suffering from large symptomatic brain metastases [1]. Even in highly selected cases, surgical resection alone leads to 1‑year local control rates of only around 40% [2]. Since the 1990s, postoperative radiotherapy has become the gold standard for these patients. Improvement of systemic oncological therapies has meanwhile led to prolonged survival of metastatic cancer patients. Therefore, long-term neurotoxicity resulting from former standard whole-brain radiotherapy becomes more and more relevant and affects patients’ functional outcome. Randomized trials have shown the detrimental effect of whole-brain radiotherapy on neurocognitive functioning [3]. This led to the treatment strategy of focal cavity radiotherapy following surgery [4, 5]. However, this treatment strategy cannot address the risk of tumor recurring in the brain distant from the resection cavity, and coping systemic treatment options are lacking currently. At Augsburg University Medical Center (UKA), the interdisciplinary neuro-oncology board established a treatment strategy in 2014 composed of regular 3‑monthly MRI follow-up and radiosurgery as the basis for salvage treatment of distant “oligo-”metastatic brain failure.
This study presents retrospective data on our strategy to spare whole-brain radiotherapy, which is known to be associated with toxicity for patients, through the complete course of their disease. Furthermore, it provides data on long-term local control of the brain (“neuro-control”).
Methods
Data collection
We scanned the data from our hospital information system ORBIS (Dedalus Healthcare Group GmbH, Bonn, Germany), our radiology information system Deep Unity (Dedalus Healthcare Group GmbH, Bonn, Germany), and our oncology information system MOSAIQ (ELEKTA AB, Stockholm, Sweden) for patients who underwent neurosurgical resection of brain metastases followed by focal percutaneous radiotherapy of the resection cavity and who did not receive whole-brain radiotherapy. Treatments took place between 2014 and 2021 at UKA. The time point for the last follow-up included was December 31, 2021.
Treatment
Treatment of all cases followed the recommendations of the UKA multidisciplinary tumor board. After obtaining informed consent from the patient, neurosurgical brain metastasectomy was performed. Within 72h after surgery, a postoperative MRI was performed to evaluate resection status.
Radiotherapy treatment planning
For radiotherapy treatment planning, all patients were immobilized with an individually customized thermoplastic mask in supine position and scanned with a Somatom Confidence CT (Siemens Healthineers AG, Erlangen, Germany). The dataset was reconstructed in 1‑mm slices. An additional MRI with intravenous gadolinium was obtained on the same or, at the latest, the following day, and reconstructed in 1‑mm slices for image fusion. Board-certified radiation oncologists performed target volume definition and contouring. The gross tumor volume (GTV) was defined as the resection cavity including the contrast-enhancing rim/suspected residual tumor. For all treatments, a clinical target volume (CTV) was created by a 1-mm expansion of the GTV within surrounding brain or meningeal tissue. As the standard neurosurgical resection procedure of brain metastases with meningeal contact in our center includes wide meningeal resection, no additional margins along the meninges were added in these cases. Planning target volume (PTV) was defined as CTV plus 2 mm.
In 2014, our institutional policy changed. With increasing data regarding the advantages of fractioned stereotactic radiotherapy, we moved away from single-fraction treatment in cavity treatment. However, in two patients in this cohort, a single radiation dose of 20 Gy was prescribed to the 80% isodose surrounding the PTV margin, demanding that at least 98% of the PTV should receive 100% of the prescribed dose.
For the vast majority of patients, a radiation dose of five fractions of 6 Gy was prescribed to the PTV margin, demanding that at least 98% of the PTV should receive 100% of the prescribed dose. In case of suspected residual disease in the postoperative MRI [6], the radiation dose was escalated to five fractions of 7 Gy. For two patients with large cavities, the prescribed dose/fractionation had to be changed to 13 × 3 Gy to meet the dose constrains of organs at risk. One of these patients only received 6 × 3 Gy due to a severe pulmonary infection, requiring intensive care unit treatment.
Radiation treatment
Radiotherapy was performed as frameless, image-guided stereotactic radiotherapy with a True Beam linear accelerator (Varian Medical Systems, Palo Alto, USA). Radiotherapy fractions were given on consecutive working days.
Follow-up
After treatment, all patients received standardized follow-up (FU) including 3‑monthly MRI of the brain according to UKA FU policy after focal brain radiotherapy. Without exceptions, all decisions for further treatments followed the recommendations of the UKA multidisciplinary tumor board.
Neuroradiological assessment
Assessment of FU MRIs followed the response assessment in neuro-oncology (RANO) criteria for brain metastases [7].
In the case of a suspected local relapse, MRI perfusion [8] and PET scan with F‑18-fluorethyltyrosin (FET) were used for non-invasive distinction between tumor relapse and radionecrosis. In case of persistent suspicion of local tumor regrowth, neurosurgical resection of the lesion was recommended to the patient.
Statistical analyses
All statistical analyses were performed with EZR (version 3.4.1/the R Foundation for Statistical Computing, Vienna, Austria) [9] using Kaplan–Meier methods, Cox proportional hazard regression, and log-rank tests.
Results
We analyzed the course of 83 patients with a median age of 63 years (range 34–87 years) with 86 resected brain metastases. Fifty-three patients (64%) had a singular brain metastasis, 16 patients (19%) two brain metastases, 12 patients (14%) three, and two patients (2%) four brain metastases at the time of treatment. Three of these metastases were resected; all others were treated with radiosurgery only. At the time of analysis, 36 of all patients had died, 34 were alive, and the status of 13 patients was unknown. Twenty-five of the deceased patients (75%) had no radiologic signs of central nervous system progression on their last cerebral MRI. Main causes of death were systemic progressive disease and infections. Median follow-up and MRI follow-up after radiotherapy was 7.3 months (range 0–71.2 months) and 6.4 months (range 0–64.3 months). Estimated 1‑year overall survival rate and median survival were 57.8% (95% CI 44.9–68.8%) and 1.5 years (95% CI 0.8 year–not applicable (NA)), respectively. Table 1 provides additional patient characteristics.
Postoperative MRI report suspected incomplete resection of 29 lesions (34%). Median interval between brain surgery and start of radiotherapy was 29 days (range 14–78 days). External beam radiotherapy was administered with a median of five fractions (range 1–13 fractions) over a median period of 6 days (range 1–19 days) to a median biological effective dose (α/β 10) of 48 Gy (range 23.4–60 Gy) at the margin of the planning target volume. Ten patients underwent salvage surgery due to suspected regrown tumor in the resection cavity (i.e., local failure) during follow-up in MRI ± FET-PET. Only in five of these cases did histology reveal vital tumor cells.
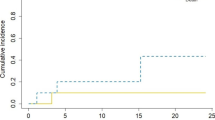
Within our standardized follow-up, we detected eight local recurrences (9.3%) in total. The 1‑year local control rate at the resection site was estimated at 82.7% (95% CI 67.7–91.2%; Fig. 1). There was no statistically significant difference in local control at the resection site between patients with complete and suspected incomplete resection. In 25 patients (30.1%), recurrences occurred in distant areas of the brain, leading to an estimated 1‑year distant brain control rate of 55.7% (95% CI 40.5–68.4%) (Fig. 2). Within the subset of these distant relapses, six cases of leptomeningeal disease (7.2%) were observed. Estimated 1‑year leptomeningeal disease rate was 16.0% (95% CI 7.3–32.9%). Eleven distant brain recurrences could be salvaged with radiosurgery. During the whole course of disease, 14 patients (17%) developed disseminated metastatic disease in the brain, thus demanding whole-brain radiotherapy (WBI). In all these patients, this was the first manifestation of a distant recurrence after focal cavity radiotherapy. Median time between the end of WBI and death was 65 days (9–1232 days). Kaplan–Meier survival analysis showed that 72.3% of all patients (95% CI 57.1–82.9%) were estimated to be free of WBI after 1 year (Fig. 3). The estimated 1‑year WBI-free survival rate was 51.3% (95% CI 38.7–62.5%). In summary the course of all applied treatments led to an estimated 1‑year neuro-control rate of 79.1% (95% CI 65.0–88.0%) in our set of patients after focal cavity radiotherapy with controlled disease in the brain on the last obtained MRI scan (Fig. 4). Five patients (6%) died within 30 days after completion of radiotherapy, mostly due to infections. No radiotherapy-associated cause of death was observed. After brain metastasis resection, 56 patients (67%) received palliative systemic treatment, which started at a median of 54 days (range 9–290 days) after surgery. Fourteen patients received immune checkpoint inhibitors (17%), 13 patients Her2-neu-targeted antibodies (16%), 17 patients cytotoxic chemotherapy (20%), nine patients targeted therapeutic agents (11%), four patients VEGF-directed antibodies (5%), and two patients anti-hormonal therapy as single-agent or combined therapy. In univariate analysis, systemic treatment, type of systemic treatment, histology, localization, size of metastasis, controlled systemic disease, and interval to start of radiotherapy had no impact on the development of recurrences in distant parts of the brain or leptomeningeal disease. The number of brain metastases showed a hazard ratio of 1.487 (95% CI 1.001–2.197; p = 0.04619) for distant brain failure in a Cox proportional hazards regression model. During their follow-up, 18 patients (21%) developed signs of radiotherapy-associated contrast-enhancing brain lesions (radionecrosis) on MRI scans, predominantly without any neurologic symptoms. Estimated 1‑year radionecrosis rate was 23% (95% CI 12.4–40.5%). Symptomatic radionecrosis occurred in five patients (6%), leading to estimation of a 1-year symptomatic radionecrosis rate of 3.6% (95% CI 0.5–22.8%). All symptomatic radionecrosis patients received a treatment of four infusions of bevacizumab (7.5 mg/kg every 2 weeks), which led to a significant improvement of their neurologic symptoms.
Discussion
The findings of our retrospective analysis showed that focal radiotherapy of the cavity after resection of brain metastases was associated with high local control rates. Furthermore, it could be shown that whole-brain radiotherapy could be avoided for three out of four of our patients in the complete further course of their disease, using radiosurgery to salvage observed distant brain relapse.
Our treatment strategy was associated with a high definitive neuro-control rate of 75% for all patients. This was in line with the outcome of the NCCTG N107C/CEC.3 trial showing no difference in overall survival of patients treated with focal radiotherapy versus patients treated with whole-brain radiotherapy in this situation [3]. The reported local control rate in this randomized study (61.8%) was remarkably lower than the one we found in our retrospective analysis. Possible explanations for this fact could be differences in target volume and dose concepts, as well as in the limitations of a monocentric retrospective analysis. Our observed local control rate was nevertheless in the range between 80 and 90% reported by other centers [5, 10,11,12].
As expected from published data [13,14,15], distant brain failure after focal radiotherapy of the resection cavity was found frequently in our analysis, stressing the importance of continuous follow-up including MRI. Leptomeningeal disease was found in 7.2% of the cases in our study. This is in line with published data [10, 16, 17]. Nearly half of the distant brain recurrences could be salvaged with radiosurgery. This contributed to the high estimated 1‑year WBI-free rate of 72.3% (95% CI 57.1–82.9%) and the simultaneously high estimated 1‑year neuro-control rate of 79.1% (95% CI 65.0–88.0%) of our retrospective analysis. In our dataset, no evidence for an additional benefit in terms of locoregional control in the brain could be shown for systemic treatments. This was supported by a subset analysis, which gave no evidence of an effect of type of systemic treatment, interval to start of systemic treatment after resection, or controlled systemic disease status of the patient. The heterogeneity and limited number of patients in our study might bias this.
Radiotherapy-associated contrast-enhancing brain lesions, i.e., radionecrosis [18, 19], have been inconsistently reported in publications so far. This is due to the fact that the differential diagnosis to local progression is challenging. In our series, those MRI findings were detected in 21% of all patients during follow-up. Most were diagnosed in asymptomatic patients and regressing or stable without any further treatment. Five of them were histologically proven by complete resection of the lesion due to suspected local recurrence on MRI ± FET-PET. The estimated 1‑year radionecrosis rate of 23% was in the range of other reported series [4, 5], while the 1‑year symptomatic radionecrosis rate of 3.6% was quite low in the light of these data.
The findings of this analysis have all the limitations of a retrospective study, which is liable to selection bias and incomplete follow-up data. Its limited sample size did not allow exploring of certain covariates like the influence of adjuvant treatment or specific genetic mutations on long-term neuro-control. For this purpose, larger multi-institutional register studies would be desirable.
Conclusion
In our study, focal radiotherapy of the tumor cavity, replacing whole-brain radiotherapy after resection of brain metastases, was associated with high local control. For three out of four patients, whole-brain radiotherapy could be avoided in the complete further course of their disease, using radiosurgery to salvage observed distant brain relapse without compromising neuro-control.
References
Vogelbaum MA, Brown PD, Messersmith H, Brastianos PK, Burri S, Cahill D, Dunn IF, Gaspar LE, Gatson NTN, Gondi V, Jordan JT, Lassman AB, Maues J, Mohile N, Redjal N, Stevens G, Sulman E, van den Bent M, Wallace HJ, Weinberg JS, Zadeh G, Schiff D (2022) Treatment for brain metastases: aSCO-SNO-ASTRO guideline. J Clin Oncol 40:492–516. https://doi.org/10.1200/JCO.21.02314
Mahajan A, Ahmed S, McAleer MF, Weinberg JS, Li J, Brown P, Settle S, Prabhu SS, Lang FF, Levine N, McGovern S, Sulman E, McCutcheon IE, Azeem S, Cahill D, Tatsui C, Heimberger AB, Ferguson S, Ghia A, Demonte F, Raza S, Guha-Thakurta N, Yang J, Sawaya R, Hess KR, Rao G (2017) Post-operative stereotactic radiosurgery versus observation for completely resected brain metastases: a single-centre, randomised, controlled, phase 3 trial. Lancet Oncol 18:1040–1048. https://doi.org/10.1016/S1470-2045(17)30414-X
Brown PD, Ballman KV, Cerhan JH, Anderson SK, Carrero XW, Whitton AC, Greenspoon J, Parney IF, Laack NNI, Ashman JB, Bahary J‑P, Hadjipanayis CG, Urbanic JJ, Barker FG, Farace E, Khuntia D, Giannini C, Buckner JC, Galanis E, Roberge D (2017) Postoperative stereotactic radiosurgery compared with whole brain radiotherapy for resected metastatic brain disease (NCCTG N107C/CEC·3): a multicentre, randomised, controlled, phase 3 trial. Lancet Oncol 18:1049–1060. https://doi.org/10.1016/S1470-2045(17)30441-2
Redmond KJ, De Salles AAF, Fariselli L, Levivier M, Ma L, Paddick I, Pollock BE, Regis J, Sheehan J, Suh J, Yomo S, Sahgal A (2021) Stereotactic Radiosurgery for postoperative metastatic surgical cavities: a critical review and international stereotactic radiosurgery society (ISRS) practice guidelines. Int J Radiat Oncol Biol Phys 111:68–80. https://doi.org/10.1016/j.ijrobp.2021.04.016
Minniti G, Niyazi M, Andratschke N, Guckenberger M, Palmer JD, Shih HA, Lo SS, Soltys S, Russo I, Brown PD, Belka C (2021) Current status and recent advances in resection cavity irradiation of brain metastases. Radiat Oncol 16:73. https://doi.org/10.1186/s13014-021-01802-9
Kamp MA, Rapp M, Bühner J, Slotty PJ, Reichelt D, Sadat H, Dibué-Adjei M, Steiger H‑J, Turowski B, Sabel M (2015) Early postoperative magnet resonance tomography after resection of cerebral metastases. Acta Neurochir 157:1573–1580. https://doi.org/10.1007/s00701-015-2479-4
Wen PY, Chang SM, Van den Bent MJ, Vogelbaum MA, Macdonald DR, Lee EQ (2017) Response assessment in neuro-oncology clinical trials. J Clin Oncol 35:2439–2449. https://doi.org/10.1200/JCO.2017.72.7511
Nichelli L, Casagranda S (2021) Current emerging MRI tools for radionecrosis and pseudoprogression diagnosis. Curr Opin Oncol 33:597–607. https://doi.org/10.1097/CCO.0000000000000793
Kanda Y (2013) Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 48:452–458. https://doi.org/10.1038/bmt.2012.244
Gutschenritter T, Venur VA, Combs SE, Vellayappan B, Patel AP, Foote M, Redmond KJ, Wang TJC, Sahgal A, Chao ST, Suh JH, Chang EL, Ellenbogen RG, Lo SS (2020) The judicious use of stereotactic radiosurgery and hypofractionated stereotactic radiotherapy in the management of large brain metastases. Cancers 13:70. https://doi.org/10.3390/cancers13010070
Kahl K‑H, Balagiannis N, Höck M, Schill S, Roushan Z, Shiban E, Müller H, Grossert U, Konietzko I, Sommer B, Maurer CJ, Berlis A, Heidecke V, Janzen T, Stüben G (2021) Intraoperative radiotherapy with low-energy x‑rays after neurosurgical resection of brain metastases—an Augsburg University Medical Center experience. Strahlenther Onkol. https://doi.org/10.1007/s00066-021-01831-z
Eitz KA, Lo SS, Soliman H, Sahgal A, Theriault A, Pinkham MB, Foote MC, Song AJ, Shi W, Redmond KJ, Gui C, Kumar AMS, Machtay M, Meyer B, Combs SE (2020) Multi-institutional analysis of prognostic factors and outcomes after hypofractionated stereotactic radiotherapy to the resection cavity in patients with brain metastases. JAMA Oncol. https://doi.org/10.1001/jamaoncol.2020.4630
Dincoglan F, Sager O, Uysal B, Demiral S, Gamsiz H, Gündem E, Elcim Y, Dirican B, Beyzadeoglu M (2019) Evaluatıon of hypofractıonated stereotactıc radıotherapy (HFSRT) to the resectıon cavıty after surgıcal resectıon of braın metastases: a single center experıence. Indian J Cancer 56:202. https://doi.org/10.4103/ijc.IJC_345_18
Specht HM, Kessel KA, Oechsner M, Meyer B, Zimmer C, Combs SE (2016) HFSRT of the resection cavity in patients with brain metastases. Strahlenther Onkol 192:368–376. https://doi.org/10.1007/s00066-016-0955-2
Combs SE, Bilger A, Diehl C, Bretzinger E, Lorenz H, Oehlke O, Specht HM, Kirstein A, Grosu A‑L (2018) Multicenter analysis of stereotactic radiotherapy of the resection cavity in patients with brain metastases. Cancer Med 7:2319–2327. https://doi.org/10.1002/cam4.1477
Minniti G, Lanzetta G, Capone L, Giraffa M, Russo I, Cicone F, Bozzao A, Alongi F, Nicosia L, Fineschi G, Marchetti L, Tufo T, Bianciardi F, Esposito V, Gentile P, Paolini S (2021) Leptomeningeal disease and brain control after postoperative stereotactic radiosurgery with or without immunotherapy for resected brain metastases. J Immunother Cancer 9:e3730. https://doi.org/10.1136/jitc-2021-003730
Nguyen TK, Sahgal A, Detsky J, Atenafu EG, Myrehaug S, Tseng C‑L, Husain Z, Heyn C, Maralani P, Ruschin M, Perry J, Soliman H (2020) Predictors of leptomeningeal disease following hypofractionated stereotactic radiotherapy for intact and resected brain metastases. Neuro-Oncology 22:84–93. https://doi.org/10.1093/neuonc/noz144
Le Rhun E, Dhermain F, Vogin G, Reyns N, Metellus P (2016) Radionecrosis after stereotactic radiotherapy for brain metastases. Expert Rev Neurother 16:903–914. https://doi.org/10.1080/14737175.2016.1184572
Kaufmann TJ, Smits M, Boxerman J, Huang R, Barboriak DP, Weller M, Chung C, Tsien C, Brown PD, Shankar L, Galanis E, Gerstner E, van den Bent MJ, Burns TC, Parney IF, Dunn G, Brastianos PK, Lin NU, Wen PY, Ellingson BM (2020) Consensus recommendations for a standardized brain tumor imaging protocol for clinical trials in brain metastases. Neuro-Oncology 22:757–772. https://doi.org/10.1093/neuonc/noaa030
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
K.-H. Kahl received speaker honoraria and travel grants from ELEKTA AB, Varian Medical Systems, Carl Zeiss Meditec AG, AstraZeneca, Bristol Myers Squibb, MSD, Merck, and Medical Intelligence Medizintechnik, and is member of the advisory board of Bristol Myers Squibb, MSD, and AstraZeneca. E. Shiban, S. Gutser, C.J. Maurer, B. Sommer, H. Müller, I. Konietzko, U. Grossert, A. Berlis, T. Janzen, and G. Stüben declare that they have no competing interests.
Ethical standards
The ethics committee of the Ludwig Maximilian University of Munich (PNR 22-0156) approved the treatment protocol. It consists of experts of all medical disciplines involved and conforms to the provisions of the Declaration of Helsinki in 1995 (as revised in Edinburgh in 2000). The statistical analysis was performed using anonymous clinical data.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kahl, KH., Shiban, E., Gutser, S. et al. Focal cavity radiotherapy after neurosurgical resection of brain metastases: sparing neurotoxicity without compromising locoregional control. Strahlenther Onkol 198, 1105–1111 (2022). https://doi.org/10.1007/s00066-022-02003-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-022-02003-3