Abstract
Purpose
Aim of this single center, retrospective study was to assess the efficacy and safety of linear accelerator-based hypofractionated stereotactic radiotherapy (HFSRT) to the resection cavity of brain metastases after surgical resection. Local control (LC), locoregional control (LRC = new brain metastases outside of the treatment volume), overall survival (OS) as well as acute and late toxicity were evaluated.
Patients and methods
46 patients with large (> 3 cm) or symptomatic brain metastases were treated with HFSRT. Median resection cavity volume was 14.16 cm3 (range 1.44–38.68 cm3) and median planning target volume (PTV) was 26.19 cm3 (range 3.45–63.97 cm3). Patients were treated with 35 Gy in 7 fractions prescribed to the 95–100 % isodose line in a stereotactic treatment setup. LC and LRC were assessed by follow-up magnetic resonance imaging.
Results
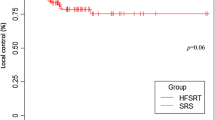
The 1-year LC rate was 88 % and LRC was 48 %; 57% of all patients showed cranial progression after HFSRT (4% local, 44% locoregional, 9% local and locoregional). The median follow-up was 19 months; median OS for the whole cohort was 25 months. Tumor histology and recursive partitioning analysis score were significant predictors for OS. HFSRT was tolerated well without any severe acute side effects > grade 2 according to CTCAE criteria.
Conclusion
HFSRT after surgical resection of brain metastases was tolerated well without any severe acute side effects and led to excellent LC and a favorable OS. Since more than half of the patients showed cranial progression after local irradiation of the resection cavity, close patient follow-up is warranted. A prospective evaluation in clinical trials is currently being performed.
Zusammenfassung
Ziel
Ziel dieser monozentrischen, retrospektiven Studie war es, die Effektivität und Sicherheit einer hypofraktionierter stereotaktischer Radiotherapie (HFSRT) nach chirurgischer Resektion von Hirnmetastasen zu untersuchen. Neben der lokalen Kontrolle (LC), der lokoregionären Kontrolle (LRC = neue Hirnmetastasen außerhalb des Behandlungsvolumens) und dem Gesamtüberleben (OS) wurden die akute und späte Toxizität analysiert.
Patienten und Methoden
46 Patienten wurden nach chirurgischer Resektion von großen (> 3 cm) oder symptomatischen Hirnmetastasen mit HFSRT behandelt. Das mediane Volumen der Resektionshöhle betrug 14,16 cm3 (Spanne 1,44–38,68 cm3), das mediane Planungszielvolumen (PTV) 26,19 cm3 (Spanne 3,45–63,97 cm3). Die Therapie erfolgte mit 35 Gy in 7 Fraktionen in einem stereotaktischem Setup. LC und LRC wurden mit Hilfe von Magnetresonanztomographie bewertet.
Ergebnisse
Nach einem Jahr betrug die LC 88% und die LRC 48%; 57% aller Patienten entwickelten eine intrakranielle Tumorprogression nach HFSRT (4% lokal, 44% lokoregionär, 9% lokal und lokoregionär). Das mediane Follow-up betrug 19 Monate; das mediane OS des gesamten Kollektivs lag bei 25 Monaten. Tumorhistologie und rekursiver Partitionierungsanalyse-Score waren signifikante Prädiktoren für das OS. Es traten keine höhergradigen akuten Nebenwirkungen (>Grad 2 gemäß der CTCAE-Kriterien) auf.
Schlussfolgerung
HFSRT nach chirurgischer Resektion von Hirnmetastasen führt zu einer exzellenten LC und positivem OS ohne höhergradige akute Nebenwirkungen. Da mehr als die Hälfte der Patienten einen intracraniellen Progress zeigten ist eine engmaschige Nachsorge zu empfehlen. Prospektive Evaluationen im Rahmen von klinischen Studien werden derzeit durchgeführt.




Similar content being viewed by others
References
Gavrilovic IT, Posner JB (2005) Brain metastases: epidemiology and pathophysiology. J Neurooncol 75:5–14
Tsao MN et al (2012) Whole brain radiotherapy for the treatment of newly diagnosed multiple brain metastases. Cochrane Database Syst Rev 4:CD003869
Spencer K, Hall A, Jain P (2014) Brain metastases. Clin Med 14:535–537
Kalkanis SN, Linskey ME (2010) Evidence-based clinical practice parameter guidelines for the treatment of patients with metastatic brain tumors: introduction. J Neurooncol 96:7–10
Schackert G et al (2013) Retrospective study of 127 surgically treated patients with multiple brain metastases: indication, prognostic factors, and outcome. Acta Neurochir (Wien) 155:379–387
Kocher M et al (2011) Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952–26001 study. J Clin Oncol 29:134–141
Berghoff AS, Preusser M (2013) Biology in prevention and treatment of brain metastases. Expert Rev Anticancer Ther 13:1339–1348
Chang EL et al (2009) Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. Lancet Oncol 10:1037–1044
Aoyama H et al (2006) Stereotactic radiosurgery plus whole-brain radiation therapy vs stereotactic radiosurgery alone for treatment of brain metastases: a randomized controlled trial. JAMA 295:2483–2491
Combs SE et al (2004) Treatment of cerebral metastases from breast cancer with stereotactic radiosurgery. Strahlenther Onkol 180:590–596
Patchell RA (2003) The management of brain metastases. Cancer Treat Rev 29:533–540
Soffietti R et al (2013) A European Organisation for Research and Treatment of Cancer phase III trial of adjuvant whole-brain radiotherapy versus observation in patients with one to three brain metastases from solid tumors after surgical resection or radiosurgery: quality-of-life results. J Clin Oncol 31:65–72
Hauswald H et al (2012) Efficacy and toxicity of whole brain radiotherapy in patients with multiple cerebral metastases from malignant melanoma. Radiat Oncol 7:130
Kocher M et al (2014) Stereotactic radiosurgery for treatment of brain metastases. A report of the DEGRO Working Group on Stereotactic Radiotherapy. Strahlenther Onkol 190:521–532
Roberge D, Parney I, Brown PD (2012) Radiosurgery to the postoperative surgical cavity: who needs evidence? Int J Radiat Oncol Biol Phys 83:486–493
Sperduto PW et al (2008) A new prognostic index and comparison to three other indices for patients with brain metastases: an analysis of 1,960 patients in the RTOG database. Int J Radiat Oncol Biol Phys 70:510–514
Gaspar L et al (1997) Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int J Radiat Oncol Biol Phys 37:745–751
Sperduto PW et al (2012) Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J Clin Oncol 30:419–425
Hartford AC et al (2013) Postoperative stereotactic radiosurgery without whole-brain radiation therapy for brain metastases: potential role of preoperative tumor size. Int J Radiat Oncol Biol Phys 85:650–655
Do L et al (2009) Resection followed by stereotactic radiosurgery to resection cavity for intracranial metastases. Int J Radiat Oncol Biol Phys 73:486–491
Jagannathan J et al (2009) Gamma Knife radiosurgery to the surgical cavity following resection of brain metastases. J Neurosurg 111:431–438
Kelly PJ et al (2012) Stereotactic irradiation of the postoperative resection cavity for brain metastasis: a frameless linear accelerator-based case series and review of the technique. Int J Radiat Oncol Biol Phys 82:95–101
Varlotto JM et al (2003) Analysis of tumor control and toxicity in patients who have survived at least one year after radiosurgery for brain metastases. Int J Radiat Oncol Biol Phys 57:452–464
Jarvis LA et al (2012) Tumor bed dynamics after surgical resection of brain metastases: implications for postoperative radiosurgery. Int J Radiat Oncol Biol Phys 84:943–948
Eaton BR et al (2015) Hypofractionated radiosurgery has a better safety profile than single fraction radiosurgery for large resected brain metastases. J Neurooncol 123:103–111
Minniti G et al (2013) Multidose stereotactic radiosurgery (9 Gy × 3) of the postoperative resection cavity for treatment of large brain metastases. Int J Radiat Oncol Biol Phys 86:623–629
Scharp M et al (2014) Re-irradiation in the treatment of patients with cerebral metastases of solid tumors: retrospective analysis. Radiat Oncol 9:4
Lawrence YR et al (2010) Radiation dose-volume effects in the brain. Int J Radiat Oncol Biol Phys 76:S20–S27
Maguire P, Selby P (1989) Assessing quality of life in cancer patients. Br J Cancer 60:437–440
Steinmann D et al (2012) Hypofractionated stereotactic radiotherapy (hfSRT) after tumour resection of a single brain metastasis: report of a single-centre individualized treatment approach. J Cancer Res Clin Oncol 138:1523–1529
Wang CC et al (2012) Cyberknife hypofractionated stereotactic radiosurgery (HSRS) of resection cavity after excision of large cerebral metastasis: efficacy and safety of an 800 cGy x 3 daily fractions regimen. J Neurooncol 106:601–610
Ahmed KA et al (2014) Fractionated stereotactic radiotherapy to the post-operative cavity for radioresistant and radiosensitive brain metastases. J Neurooncol 118:179–186
Ling DC et al (2015) Postoperative stereotactic radiosurgery to the resection cavity for large brain metastases: clinical outcomes, predictors of intracranial failure, and implications for optimal patient selection. Neurosurgery 76:150–156. (discussion 156–157; quiz 157)
Soltys SG et al (2008) Stereotactic radiosurgery of the postoperative resection cavity for brain metastases. Int J Radiat Oncol Biol Phys 70:187–193
Mathieu D et al (2008) Tumor bed radiosurgery after resection of cerebral metastases. Neurosurgery 62:817–823. (discussion 823–824)
Karlovits BJ et al (2009) Stereotactic radiosurgery boost to the resection bed for oligometastatic brain disease: challenging the tradition of adjuvant whole-brain radiotherapy. Neurosurg Focus 27:E7
Ogiwara H et al (2012) Intracranial relapse rates and patterns, and survival trends following post-resection cavity radiosurgery for patients with single intracranial metastases. J Neurooncol 108:141–146
Prabhu R et al (2012) Current dosing paradigm for stereotactic radiosurgery alone after surgical resection of brain metastases needs to be optimized for improved local control. Int J Radiat Oncol Biol Phys 83:e61–e66
Jensen CA et al (2011) Cavity-directed radiosurgery as adjuvant therapy after resection of a brain metastasis. J Neurosurg 114:1585–1591
Luther N et al (2013) Predicting tumor control after resection bed radiosurgery of brain metastases. Neurosurgery 73:1001–1006. (discussion 1006)
Brennan C et al (2014) A phase 2 trial of stereotactic radiosurgery boost after surgical resection for brain metastases. Int J Radiat Oncol Biol Phys 88:130–136
Ojerholm E et al (2014) Gamma Knife radiosurgery to four or more brain metastases in patients without prior intracranial radiation or surgery. Cancer Med 3:565–571
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H.M. Specht, K.A. Kessel, M. Oechsner, B. Meyer, C. Zimmer, and S.E. Combs state that there are no conflicts of interest.
All studies on humans described in the present manuscript were carried out with the approval of the responsible ethics committee and in accordance with national law and the Helsinki Declaration of 1975 (in its current, revised form). Informed consent was obtained from all patients included in studies.
Rights and permissions
About this article
Cite this article
Specht, H.M., Kessel, K.A., Oechsner, M. et al. HFSRT of the resection cavity in patients with brain metastases. Strahlenther Onkol 192, 368–376 (2016). https://doi.org/10.1007/s00066-016-0955-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-016-0955-2