Abstract
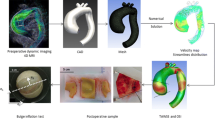
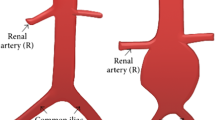
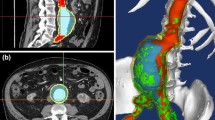
Current assessment and management of ascending thoracic aortic aneurysm (ATAA) rely heavily on the diameter of the ATAA and blood pressure rather than biomechanical and hemodynamic parameters such as arterial wall deformation or wall shear stress. The objective of the current study was to develop an accurate computational method for modeling the mechanical responses of the ATAA to provide additional information in patient evaluations. Fully coupled fluid structure interaction simulations were conducted using data from cases with ATAA with measured geometrical parameters in order to evaluate and analyze the change in biomechanical responses under normotensive and hypertensive conditions. Anisotropic hyperelastic material property estimates were applied to the ATAA data which represented three different geometrical configurations of ATAAs. The resulting analysis showed significant variations in maximum wall shear stress despite minimal differences in flow velocity between two blood pressure conditions. Additionally, the three different ATAA conditions identified different aortic expansions that were not uniform under pulsatile pressure. The elevated wall stress with hypertension was also geometry-dependent. The developed models suggest that ATTA cases have unique characteristic in biomechanical and hemodynamic evaluations that can be useful in risk management.













Similar content being viewed by others
Abbreviations
- ATAA:
-
ascending thoracic aortic aneurysm
- PWS:
-
peak wall stress
- WSS:
-
wall shear stress
- FSI:
-
fluid-structure interaction
References
Centers for Disease Control and Prevention, National Center for Health Statistics. Underlying cause of death 1999–2014 on CDC WONDER online database, released 2015. Data are from the Multiple Cause of Death Files, 1999–2014, as compiled from data provided. http://wonder.cdc.gov/ucd-icd10.html. Accessed 8 Nov 2016
Coady MA, Rizzo JA, Goldstein LJ, Elefteriades JA (1999) Natural history, pathogenesis, and etiology of thoracic aortic aneurysms and dissections. Cardiol Clin 17:615–635 vii
Olsson C, Thelin S, Ståhle E, Ekbom A, Granath F (2006) Thoracic aortic aneurysm and dissection: increasing prevalence and improved outcomes reported in a nationwide population-based study of more than 14,000 cases from 1987 to 2002. Circulation 114:2611–2618. doi:10.1161/CIRCULATIONAHA.106.630400
Davies RR, Goldstein LJ, Coady MA, Tittle SL, Rizzo JA, Kopf GS, Elefteriades JA (2002) Yearly rupture or dissection rates for thoracic aortic aneurysms: simple prediction based on size. Ann Thorac Surg 73:17–28. doi:10.1016/S0003-4975(01)03236-2
Pape LA, Tsai TT, Isselbacher EM, Oh JK, O’Gara PT, Evangelista A, Fattori R, Meinhardt G, Trimarchi S, Bossone E, Suzuki T, Cooper JV, Froehlich JB, Nienaber CA, Eagle KA (2007) Aortic diameter ≥5.5 cm is not a good predictor of type A aortic dissection: observations from the International Registry of Acute Aortic Dissection (IRAD). Circulation 116:1120–1127. doi:10.1161/CIRCULATIONAHA.107.702720
Norman PE, Powell JT (2010) Site specificity of aneurysmal disease. Circulation 121:560–568. doi:10.1161/CIRCULATIONAHA.109.880724
Rabkin SW (2015) Accentuating and opposing factors leading to development of thoracic aortic aneurysms not due to genetic or inherited conditions. Front Cardiovasc Med 2:21. doi:10.3389/fcvm.2015.00021
Emmott A, Garcia J, Chung J, Lachapelle K, El-Hamamsy I, Mongrain R, Cartier R, Leask RL (2016) Biomechanics of the ascending thoracic aorta: a clinical perspective on engineering data. Can J Cardiol 32:35–47. doi:10.1016/j.cjca.2015.10.015
Goldfinger JZ, Halperin JL, Marin ML, Stewart AS, Eagle KA, Fuster V (2014) Thoracic aortic aneurysm and dissection. J Am Coll Cardiol 64:1725–1739. doi:10.1016/j.jacc.2014.08.025
Hiratzka LF, Bakris GL, Beckman JA, Bersin RM, Carr VF, Casey DE Jr, Eagle KA, Hermann LK, Isselbacher EM, Kazerooni EA, Kouchoukos NT, Lytle BW, Milewicz DM, Reich DL, Sen S, Shinn JA, Svensson LG, Williams DM, American College of Cardiology F, American Heart Association Task Force on Practice G, American Association for Thoracic S, American College of R, American Stroke A, Society of Cardiovascular A, Society for Cardiovascular Angiography and I, Society of Interventional R, Society of Thoracic S, Society for Vascular M (2010) 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with thoracic aortic disease: executive summary. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Catheter Cardiovasc Interv 76:E43–E86
Poullis MP, Warwick R, Oo A, Poole RJ (2008) Ascending aortic curvature as an independent risk factor for type A dissection, and ascending aortic aneurysm formation: a mathematical model. Eur J Cardio-thoracic Surg 33:995–1001. doi:10.1016/j.ejcts.2008.02.029
Alberta HB, Secor JL, Smits TC, Farber MA, Jordan WD, Matsumura JS (2013) Differences in aortic arch radius of curvature, neck size, and taper in patients with traumatic and aortic disease. J Surg Res 184:613–618. doi:10.1016/j.jss.2013.05.098
Paini A, Boutouyrie P, Calvet D, Tropeano A-I, Laloux B, Laurent S (2006) Carotid and aortic stiffness determinants of discrepancies. Hypertension 47:371–376
Roccabianca S, Figueroa CA, Tellides G, Humphrey JD (2014) Quantification of regional differences in aortic stiffness in the aging human. J Mech Behav Biomed Mater 29:618–634. doi:10.1016/j.jmbbm.2013.01.026
Redheuil A, Yu WC, Wu CO, Mousseaux E, De Cesare A, Yan R, Kachenoura N, Bluemke D, Lima JAC (2010) Reduced ascending aortic strain and distensibility: earliest manifestations of vascular aging in humans. Hypertension 55:319–326. doi:10.1161/HYPERTENSIONAHA.109.141275
Krishnan K, Ge L, Haraldsson H, Hope MD, Saloner DA, Guccione JM, Tseng EE (2015) Ascending thoracic aortic aneurysm wall stress analysis using patient-specific finite element modeling of in vivo magnetic resonance imaging. Interact Cardiovasc Thorac Surg 21:471–480. doi:10.1093/icvts/ivv186
Beller CJ, Labrosse MR, Thubrikar MJ, Robicsek F (2008) Finite element modeling of the thoracic aorta: including aortic root motion to evaluate the risk of aortic dissection. J Med Eng Technol 32:167–170. doi:10.1080/03091900600687672
Shang EK, Nathan DP, Sprinkle SR, Vigmostad SC, Fairman RM, Bavaria JE, Gorman RC, Gorman JH, Chandran KB, Jackson BM (2013) Peak wall stress predicts expansion rate in descending thoracic aortic aneurysms. Ann Thorac Surg 95:593–598. doi:10.1016/j.athoracsur.2012.10.025
Bürk J, Blanke P, Stankovic Z, Barker A, Russe M, Geiger J, Frydrychowicz A, Langer M, Markl M (2012) Evaluation of 3D blood flow patterns and wall shear stress in the normal and dilated thoracic aorta using flow-sensitive 4D CMR. J Cardiovasc Magn Reson 14:84. doi:10.1186/1532-429x-14-84
Boyd AJ, Kuhn DCS, Lozowy RJ, Kulbisky GP (2016) Low wall shear stress predominates at sites of abdominal aortic aneurysm rupture. J Vasc Surg 63:1613–1619. doi:10.1016/j.jvs.2015.01.040
Lantz J, Renner J, Karlsson M (2011) Wall shear stress in a subject specific human aorta—influence of fluid-structure interaction. Int J Appl Mech 3:759–778. doi:10.1142/S1758825111001226
Grytsan A, Watton PN, Holzapfel GA (2015) A thick-walled fluid-solid-growth model of abdominal aortic aneurysm evolution: application to a patient-specific geometry. J Biomech Eng 137:31008. doi:10.1115/1.4029279
Chen Y, Canton G, Kerwin WS, Chiu B (2016) Modeling hemodynamic forces in carotid artery based on local geometric features. Med Biol Eng Comput 54:1437–1452. doi:10.1007/s11517-015-1417-1
García-Herrera CM, Celentano DJ, Herrera EA (2016) Modelling and numerical simulation of the in vivo mechanical response of the ascending aortic aneurysm in Marfan syndrome. Med Biol Eng Comput:1–10. doi: 10.1007/s11517-016-1524-7
García-Herrera CM, Celentano DJ, Cruchaga MA (2013) Bending and pressurisation test of the human aortic arch: experiments, modelling and simulation of a patient-specific case. Comput Methods Biomech Biomed Engin 16:830–839. doi:10.1080/10255842.2011.641123
Sokolis DP, Kritharis EP, Iliopoulos DC (2012) Effect of layer heterogeneity on the biomechanical properties of ascending thoracic aortic aneurysms. Med Biol Eng Comput 50:1227–1237. doi:10.1007/s11517-012-0949-x
García-Herrera CM, Atienza JM, Rojo FJ, Claes E, Guinea GV, Celentano DJ, García-Montero C, Burgos RL (2012) Mechanical behaviour and rupture of normal and pathological human ascending aortic wall. Med Biol Eng Comput 50:559–566. doi:10.1007/s11517-012-0876-x
Gasser TC, Ogden RW, Holzapfel GA (2006) Hyperelastic modelling of arterial layers with distributed collagen fibre orientations. J R Soc Interface 3:15–35. doi:10.1098/rsif.2005.0073
Tsamis A, Krawiec JT, Vorp DA (2013) Elastin and collagen fibre microstructure of the human aorta in ageing and disease: a review. J R Soc Interface 10:20121004. doi:10.1098/rsif.2012.1004
Crosetto P, Reymond P, Deparis S, Kontaxakis D, Stergiopulos N, Quarteroni A (2011) Fluid–structure interaction simulation of aortic blood flow. Comput Fluids 43:46–57. doi:10.1016/j.compfluid.2010.11.032
Reymond P, Crosetto P, Deparis S, Quarteroni A, Stergiopulos N (2013) Physiological simulation of blood flow in the aorta: comparison of hemodynamic indices as predicted by 3-D FSI, 3-D rigid wall and 1-D models. Med Eng Phys 35:784–791. doi:10.1016/j.medengphy.2012.08.009
Taelman L, Bols J, Degroote J, Muthurangu V, Panzer J, Vierendeels J, Segers P (2016) Differential impact of local stiffening and narrowing on hemodynamics in repaired aortic coarctation: an FSI study. Med Biol Eng Comput 54:497–510. doi:10.1007/s11517-015-1336-1
Malek AM, Alper SL, Izumo S (1999) Hemodynamic shear stress and its role in atherosclerosis. JAMA 282:2035–2042. doi:10.1001/jama.282.21.2035
Yeleswarapu KK, Antaki JF, Kameneva MV, Rajagopal KR (1995) A mathematical model for shear-induced hemolysis. Artif Organs 19:576–582. doi:10.1111/j.1525-1594.1995.tb02384.x
Meng H, Tutino VM, Xiang J, Siddiqui A (2014) High WSS or low WSS? Complex interactions of hemodynamics with intracranial aneurysm initiation, growth, and rupture: toward a unifying hypothesis. AJNR Am J Neuroradiol 35:1254–1262. doi:10.3174/ajnr.A3558
Vasava P, Jalali P, Dabagh M, Kolari PJ (2012) Finite element modelling of pulsatile blood flow in idealized model of human aortic arch: study of hypotension and hypertension. Comput Math Methods Med 2012:1–14. doi:10.1155/2012/861837
Weisbecker H, Pierce DM, Regitnig P, Holzapfel GA (2012) Layer-specific damage experiments and modeling of human thoracic and abdominal aortas with non-atherosclerotic intimal thickening. J Mech Behav Biomed Mater 12:93–106. doi:10.1016/j.jmbbm.2012.03.012
Holzapfel GA, Gasser TC, Ogden RW (2000) A new constitutive framework for arterial wall mechanics and a comparative study of material models. J Elast 61:1–48. doi:10.1023/a:1010835316564
Yeh HH, Grecov D, Karri S (2014) Computational modelling of bileaflet mechanical valves using fluid-structure interaction approach. J Med Biol Eng 34:482–486. doi:10.5405/jmbe.1699
Morrison TM, Choi G, Zarins CK, Taylor CA (2009) Circumferential and longitudinal cyclic strain of the human thoracic aorta: age-related changes. J Vasc Surg 49:1029–1036. doi:10.1016/j.jvs.2008.11.056
Acknowledgements
The financial support of the Natural Sciences and Engineering Research Council (NSERC) of Canada is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Yeh, H.H., Rabkin, S.W. & Grecov, D. Hemodynamic assessments of the ascending thoracic aortic aneurysm using fluid-structure interaction approach. Med Biol Eng Comput 56, 435–451 (2018). https://doi.org/10.1007/s11517-017-1693-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11517-017-1693-z