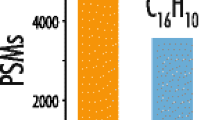
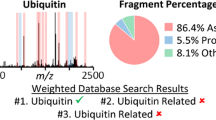
Abstract
Cysteine is unique among the proteinogenic amino acids due to its ability to form disulfide bonds. While this property is of vital importance for protein structures and biological processes, it causes difficulties for the mass spectrometric identification of cysteine-containing peptides. A common approach to overcome these problems in bottom-up proteomics is the reduction and covalent modification of sulfhydryl groups prior to enzymatic digestion. In this study, established alkylating agents and N-maleoyl amino acids with variable hydrophobicity were characterized with respect to a variety of relevant parameters and subsequently evaluated in a large-scale analysis using different ion sources. Depending on the compound, the ion source had a profound impact on the relative and absolute identification of cysteine-containing peptides. The best results were obtained by derivatization of the cysteine residues with 4-vinylpyridine and subsequent matrix-assisted laser desorption ionization (MALDI). Modification with 4-vinylpyridine increased the number of cysteine-containing peptides identified with any other compound using LC-MALDI/MS at least by a factor of 2. This experimental observation is mirrored by differences in the gas-phase basicities, which were computed for methyl thiolate derivatives of the compounds using density functional theory. With electrospray ionization (ESI), complementary use of reagents from three different compound classes, e.g., iodoacetamide, 4-vinylpyridine, and N-maleoyl beta-alanine, was beneficial compared to the application of a single reagent.

Cysteine-containing peptides are underrepresented in standard large scale proteomic experiments. However, deliberate matching of alkylating agent and ion source can help to increase identification rates, as outlined by characterization and validation of various reagents for cysteine-derivatization.






Similar content being viewed by others
References
Gundry RL, White MY, Murray CI, Kane LA, Fu Q, Stanley BA, Van Eyk JE (2009) Preparation of proteins and peptides for mass spectrometry analysis in a bottom-up proteomics workflow. Curr Protoc Mol Biol. doi:10.1002/0471142727.mb1025s88, Chapter 10: Unit 10.25
Shevchenko A, Wilm M, Vorm O, Mann M (1996) Mass spectrometric sequencing of proteins silver-stained polyacrylamide gels. Anal Chem 68(5):850–858
Nielsen ML, Vermeulen M, Bonaldi T, Cox J, Moroder L, Mann M (2008) Iodoacetamide-induced artifact mimics ubiquitination in mass spectrometry. Nat Methods 5(6):459–460. doi:10.1038/nmeth0608-459
Rehulkova H, Marchetti-Deschmann M, Pittenauer E, Allmaier G, Rehulka P (2009) Improved identification of hordeins by cysteine alkylation with 2-bromoethylamine, SDS-PAGE and subsequent in-gel tryptic digestion. J Mass Spectrom 44(11):1613–1621. doi:10.1002/jms.1675
Frahm JL, Bori ID, Comins DL, Hawkridge AM, Muddiman DC (2007) Achieving augmented limits of detection for peptides with hydrophobic alkyl tags. Anal Chem 79(11):3989–3995. doi:10.1021/ac070558q
Guan X, Hoffman B, Dwivedi C, Matthees DP (2003) A simultaneous liquid chromatography/mass spectrometric assay of glutathione, cysteine, homocysteine and their disulfides in biological samples. J Pharm Biomed Anal 31(2):251–261. doi:10.1016/S0731-7085(02)00594-0
Sechi S, Chait BT (1998) Modification of cysteine residues by alkylation. A tool in peptide mapping and protein identification. Anal Chem 70(24):5150–5158. doi:10.1021/ac9806005
Xu K, Zhang Y, Tang B, Laskin J, Roach PJ, Chen H (2010) Study of highly selective and efficient thiol derivatization using selenium reagents by mass spectrometry. Anal Chem 82(16):6926–6932. doi:10.1021/ac1011602
Guo Y, Chen L, Yang L, Wang Q (2008) Counting sulfhydryls and disulfide bonds in peptides and proteins using mercurial ions as an MS-tag. J Am Soc Mass Spectrom 19(8):1108–1113. doi:10.1016/j.jasms.2008.05.005
Wang Z, Zhang Y, Zhang H, Harrington PB, Chen H (2012) Fast and selective modification of thiol proteins/peptides by N-(phenylseleno)phthalimide. J Am Soc Mass Spectrom 23(3):520–529. doi:10.1007/s13361-011-0317-3
Lundblad RL (2014) Chemical reagents for protein modification, 4th edn. CRC, Taylor and Francis Group, Boca Raton
Boja ES, Fales HM (2001) Overalkylation of a protein digest with iodoacetamide. Anal Chem 73(15):3576–3582. doi:10.1021/ac0103423
Brewer CF, Riehm JP (1967) Evidence for possible nonspecific reactions between N-ethylmaleimide and proteins. Anal Biochem 18(2):248–255. doi:10.1016/0003-2697(67)90007-3
Gregory JD (1955) The stability of N-ethylmaleimide and its reaction with sulfhydryl groups. J Am Chem Soc 77(14):3922–3923. doi:10.1021/Ja01619a073
Rich DH, Gesellchen PD, Tong A, Cheung A, Buckner CK (1975) Alkylating derivatives of amino-acids and peptides—synthesis of N-maleoylamino acids, [1-(N-maleoylglycyl)cysteinyl]oxytocin, and [1-(N-maleoyl-11-aminoundecanoyl)cysteinyl]oxytocin—effects on vasopressin-stimulated water-loss from isolated toad bladder. J Med Chem 18(10):1004–1010. doi:10.1021/Jm00244a011
Hohenberg P, Kohn W (1964) Inhomogeneous electron gas. Phys Rev 136(3B):B864–B871
Allinger NL, Yuh YH, Lii JH (1989) Molecular mechanics. The MM3 force field for hydrocarbons. 1. J Am Chem Soc 111(23):8551–8566. doi:10.1021/Ja00205a001
Fifen JJ, Dhaouadi Z, Nsangou M (2014) Revision of the thermodynamics of the proton in gas phase. J Phys Chem A 118(46):11090–11097. doi:10.1021/Jp508968z
Bouchoux G (2012) Gas phase basicities of polyfunctional molecules. Part 3: amino acids. Mass Spectrom Rev 31(3):391–435. doi:10.1002/mas.20349
Friedman M, Krull LH, Cavins JF (1970) The chromatographic determination of cystine and cysteine residues in proteins as s-beta-(4-pyridylethyl)cysteine. J Biol Chem 245(15):3868–3871
Ren DY, Julka S, Inerowicz HD, Regnier FE (2004) Enrichment of cysteine-containing peptides from tryptic digests using a quaternary amine tag. Anal Chem 76(15):4522–4530. doi:10.1021/Ac0354645
Matsui S, Aida H (1978) Hydrolysis of some N-alkylmaleimides. J Chem Soc Perkin Trans 2(12):1277–1280. doi:10.1039/P29780001277
Smyth DG, Blumenfeld OO, Konigsberg W (1964) Reactions of N-ethylmaleimide with peptides and amino acids. Biochem J 91(3):589–595. doi:10.1042/bj0910589
Gorin G, Matic PA, Doughty G (1966) Kinetics of reaction of N-ethylmaleimide with cysteine and some congeners. Arch Biochem Biophys 115(3):593–597. doi:10.1016/0003-9861(66)90079-8
Lutolf MP, Tirelli N, Cerritelli S, Cavalli L, Hubbell JA (2001) Systematic modulation of Michael-type reactivity of thiols through the use of charged amino acids. Bioconjug Chem 12(6):1051–1056. doi:10.1021/bc015519e
Ueland PM, Skotland T, Doskeland SO, Flatmark T (1978) Adenosine 3′-5′-monophosphate-adenosine binding-protein from mouse-liver—some physicochemical properties. Biochim Biophys Acta 533(1):57–65. doi:10.1016/0005-2795(78)90547-0
Anslyn EV, Dougherty DA (2006) Modern physical organic chemistry. University Science, Sausalito
Rogers LK, Leinweber BL, Smith CV (2006) Detection of reversible protein thiol modifications in tissues. Anal Biochem 358(2):171–184. doi:10.1016/j.ab.2006.08.020
Nishikaze T, Takayama M (2006) Cooperative effect of factors governing molecular ion yields in desorption/ionization mass spectrometry. Rapid Commun Mass Spectrom 20(3):376–382. doi:10.1002/rcm.2316
Nishikaze T, Takayama M (2007) Study of factors governing negative molecular ion yields of amino acid and peptide in FAB, MALDI and ESI mass spectrometry. Int J Mass Spectrom 268(1):47–59. doi:10.1016/j.ijms.2007.08.004
Paulech J, Solis N, Cordwell SJ (2013) Characterization of reaction conditions providing rapid and specific cysteine alkylation for peptide-based mass spectrometry. Biochim Biophys Acta 1834(1):372–379. doi:10.1016/j.bbapap.2012.08.002
Kruger R, Hung CW, Edelson-Averbukh M, Lehmann WD (2005) Iodoacetamide-alkylated methionine can mimic neutral loss of phosphoric acid from phosphopeptides as exemplified by nano-electrospray ionization quadrupole time-of-flight parent ion scanning. Rapid Commun Mass Spectrom 19(12):1709–1716. doi:10.1002/rcm.1976
Miseta A, Csutora P (2000) Relationship between the occurrence of cysteine in proteins and the complexity of organisms. Mol Biol Evol 17(8):1232–1239. doi:10.1093/oxfordjournals.molbev.a026406
Karantza V (2011) Keratins in health and cancer: more than mere epithelial cell markers. Oncogene 30(2):127–138. doi:10.1038/onc.2010.456
Liao PC, Allison J (1995) Enhanced detection of peptides in matrix-assisted laser-desorption ionization mass-spectrometry through the use of charge-localized derivatives. J Mass Spectrom 30(3):511–512. doi:10.1002/jms.1190300318
Mirzaei H, Regnier F (2006) Enhancing electrospray ionization efficiency of peptides by derivatization. Anal Chem 78(12):4175–4183. doi:10.1021/ac0602266
Williams DK Jr, Comins DL, Whitten JL, Muddiman DC (2009) Evaluation of the ALiPHAT method for PC-IDMS and correlation of limits-of-detection with nonpolar surface area. J Am Soc Mass Spectrom 20(11):2006–2012. doi:10.1016/j.jasms.2009.07.019
Acknowledgments
The authors thank Friedrich Mandel and the team of the Agilent Demolab Waldbronn for their kind support during the Agilent Mass Spec Research Summer 2014. We are most thankful to Prof. Dr. Karl Gademann and to Prof. Dr. Bernd F. Straub for hosting us in their groups. Jaguar licenses were provided by the bwgrid. We gratefully acknowledge the financial support from the German Research Foundation (DFG grant BE 5492/1-1), the German Cancer Research Center, the Dietmar Hopp Stiftung, and the Boehringer Ingelheim Fonds.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 1252 kb)
Rights and permissions
About this article
Cite this article
Nadler, W., Berg, R., Walch, P. et al. Ion source-dependent performance of 4-vinylpyridine, iodoacetamide, and N-maleoyl derivatives for the detection of cysteine-containing peptides in complex proteomics. Anal Bioanal Chem 408, 2055–2067 (2016). https://doi.org/10.1007/s00216-015-9113-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-015-9113-4