Abstract
Background: Patient-specific cutting blocks in total knee arthroplasty have been promoted to improve mechanical alignment, reduce alignment outliers and improve patient outcomes. The aim of this study was to compare the efficacy of patient-specific instrumentation (PSI) and conventional instrumentation (CI) in achieving neutral alignment and accurate component positioning in total knee arthroplasty.
Methods: We conducted a double-blinded randomized controlled trial in which patients were randomly assigned to treatment with either PSI or CI.
Results: Fifty-four patients were included in the study. No relevant improvement in coronal alignment was found between the PSI and CI groups with post-hoc power of 0.91. Tibial slope was found to be more accurately reproduced to the preoperative target of 3° with PSI than with CI (3.8°± 3.1° v. 7.7°± 3.6°, respectively, p < 0.001). There were no differences found in patient-reported outcome measures, surgical time or length of hospital stay.
Conclusion: Given the added cost of the PSI technique, its use is difficult to justify given the small improvement in only a single alignment parameter.
Clinical trial registration Clinicaltrials.gov, no. NCT03416946.
Total knee arthroplasty (TKA) is the current gold standard treatment for advanced knee arthritis.1 The goal of TKA is to provide patients with an enhanced level of function and a decreased level of pain, leading to an overall improvement in quality of life.2,3 Although there are some conflicting reports, it is widely believed that achieving neutral mechanical alignment of the joint is critical for long-term joint function and patient satisfaction.4–8 Over time, an improperly aligned joint can lead to excessive wear as well as increased joint laxity and instability resulting in joint failure.2,9–11 As the demand for TKA continues to rise along with the cost of TKA and associated revisions, it is critical that steps are taken to ensure efficient ways to provide long-lasting joint replacements that are satisfactory to patients.1,12
Patient-specific instrumentation (PSI) is a modern TKA technique that attempts to improve upon the accuracy of conventional instrumentation (CI) in the implantation of knee prostheses. CI used for TKA relies on surgeons using external bony landmarks along with intramedullary guides for the positioning of bone cutting blocks on the distal femur and proximal tibia.12,13 This method can be imprecise, as even when it is performed by an experienced surgeon, it can result in a misaligned implant.14,15 PSI uses preoperative imaging to generate custom bone cutting guides that fit precisely on the unique articular surface of the bone of each patient to allow for more precise bony cuts.16,17 The proposed benefits of the PSI system include improved mechanical alignment of the limb, a decrease in surgical time, a reduction in blood loss and a decrease in the number of surgical trays used.12,13
A variety of PSI systems exist, each employing their own distinct imaging protocol to produce custom cutting guides. A few randomized controlled trials have compared PSI with CI systems, and several cohort studies have compated conventional TKA with the array of distinct PSI systems. 2,9,17–19 The findings of these studies have been inconsistent with regard to the proposed benefits of PSI and, as a result, debate continues regarding the role of PSI in TKA. The goal of this study was to further explore the efficacy of PSI versus CI in achieving neutral alignment and accurate component positioning. This study further aimed to clarify possible differences in patient function and satisfaction as measured by patient-reported outcome measures (PROMs).
Methods
Study design
This study was a double-blinded randomized controlled trial enrolling patients from September 2011 to May 2015. Participants were randomly assigned to receive TKA using either PSI or CI. Randomization was done in a block design stratified by 1 of the 4 participating surgeons to ensure equal distribution between the 2 surgical techniques by surgeon. Patients included in the study were those who were slated for primary knee replacement surgery for all diagnoses. Patients were excluded if they had contraindications for magnetic resonance imaging (MRI), if they had a deformity of the femur preventing the use of the intramedullary guide used in the CI set, if they required constrained implants, if they were undergoing revision surgery or if they were scheduled for bilateral knee surgery, either simultaneous or staged. Participants were blinded to group assignment and surgical technique. All participants underwent preoperative MRI assessment to ensure that blinding was maintained. All patients gave written informed consent and the study was approved by the University of Manitoba Bannatyne Research Ethics Board and the Concordia Hospital Research Ethics Board.
Surgical technique
The Smith & Nephew Legion primary total knee system was used in both groups. For the PSI group, the Visionaire system from Smith & Nephew was used. This system uses a short-sequence MRI scan coupled with a 3-foot standing radiograph to determine alignment, rotation and recommended sizing of the implant. These data are then used to create a personalized custom cutting guide for use during the surgical procedure. Implant sizing and alignment modelling for the PSI blocks were reviewed and approved by the treating surgeon before PSI production in all cases. The 4 surgeons participating in the study were fellowship trained and had a minimum of 5 years’ experience with the CI used for TKA in their high-volume practices before the study. Before the initiation of the study, the surgeons were instructed in the use of the Visionaire PSI system. Each surgeon performed a minimum of 2 PSI knee procedures before participating in the study. Anesthetic and surgical techniques were identical for both groups and differed only in the type of instrumentation used. A pneumatic tourniquet was used in all cases from skin incision until implant fixation. A midline skin incision was used, followed by a medial parapatellar arthrotomy. As per the surgical group’s normal routine, anteroposterior (AP) and lateral x-rays of the knee were performed in the recovery room to assess for any undetected intraoperative complications. Patient in-hospital care and rehabilitation were standardized using a total knee replacement care map.
Patients followed a routine follow-up schedule and were assessed by clinical and radiologic examination at 8 weeks, 6 months, 1 year and 2 years after their surgery.
Outcome measurements
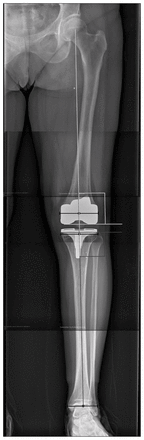
Limb alignment and component positioning were assessed using 3-foot standing anteroposterior (AP) and supine lateral radiographs (Fig. 1, Fig. 2). Full radiographic alignment was evaluated preoperatively and 6 months postoperatively. If patients’ images could not be assessed at 6 months, then images from subsequent visits were used. The mechanical axis of the lower limb, the femoral component alignment, and the tibial component alignment in the coronal plane were measured from the 3-foot standing AP radiograph. The mechanical axis of the femur was defined as the line drawn from the centre of the femoral head to the centre of the femoral condyles. The mechanical axis of the tibia was defined as the line drawn from the centre of the tibial plateau to the centre of the talus.20,21 The angle formed between the mechanical axes of the femur and tibia create the hip–knee–ankle (HKA) angle.20 The aim of TKA is to restore neutral mechanical alignment of the lower limb, as defined by an HKA angle of 180°.20,22 Alignment of the limb in a varus deviation was recorded as a negative value, while a valgus deviation was recorded as a positive value.
Coronal hip–knee–ankle (HKA) angle and coronal implant alignment. Black lines show the width and centre points of the hip, femoral condyles, tibial plateau and ankle. Dark grey lines demarcate the tibial mechanical axis and base-plate alignment. Light grey lines demarcate the femoral mechanical axis and femoral component alignment. The intersection of the femoral and tibial mechanical axes is the HKA angle.
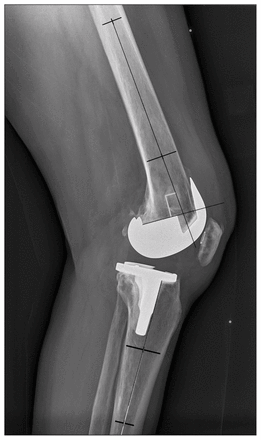
Sagittal femoral and tibial component alignment. The femoral component alignment is the angle subtended in light grey. The tibial component alignment is the intersection of the black and white lines.
Femoral component alignment in the coronal plane was measured as the angle formed by the mechanical axis of the femur and a line tangent to the distal femoral condyles as viewed on a 3-foot standing digital radiograph. Tibial component alignment in the coronal plane was measured as the angle formed by the mechanical axis of the tibia and a line tangent to the inferior surface of the base plate of the tibial component. 9,22,23 The desired femoral and tibial coronal alignments were 90°. A femoral or tibial coronal angle less than 90° indicated varus deviation relative to the mechanical axis while an angle greater than 90° was valgus deviation.23
Femoral and tibial component alignment was also assessed in the sagittal plane using a weight-bearing 43 cm × 43 cm digital detector image. The femoral sagittal alignment was measured as the angle formed by the anatomic axis of the femur (intramedullary canal bisecting the femur in half) and a line across the superior portion of the femoral implant component. The tibial sagittal alignment was measured as the angle formed from the anatomic axis of the tibia (intramedullary canal bisecting the tibia in half) and a line perpendicular to the inferior surface of the tibial base plate (parallel to the tibial post).23,24 The desired femoral sagittal alignment was 90° and the desired tibial sagittal alignment was a 3° posterior tibial slope as per the knee system design. A femoral sagittal angle less than 90° indicated femoral component flexion, while an angle greater than 90° was a flexion extension. A tibial sagittal angle greater than 0° indicated a posterior tibial slope while an angle less than 0° was an anterior tibial slope.23 All measurements were completed digitally using the IMPAX 6.6.1 picture archiving and communication system (Agfa HealthCare N.V.). All observations were made by an observer who was blinded to patient group assignment and surgical technique.
Clinical outcome scores were measured preoperatively and 1 year and 2 years postoperatively by the EuroQol 5-dimension 3-level (EQ-5D-3L) instrument with the EQ visual analogue scale (VAS),25 Oxford Knee Score (OKS),26 Pain Catastrophizing Scale (PCS),27 a VAS for knee pain, a VAS for satisfaction (Appendix 1, available at canjsurg.ca/018318-a1) and the University of California, Los Angeles (UCLA) Activity Score.28 Other measures, including surgical time, length of hospital stay and perioperative complications, were recorded.
Sample size
This study was powered to detect a difference in postoperative frontal limb alignment between the CI and PSI groups. The standard deviation of frontal plane alignment using CI for our surgical group was previously determined as 3.1°.29 Assuming p = 0.05 and power of 95%, a sample size of 35 patients per arm was selected to allow for 20% patient loss to follow-up.
Statistical analysis
Data were analyzed using SAS statistical analysis software (SAS Institute Inc.). Patient demographics and preoperative function scores were compared between the 2 groups to assess the adequacy of randomization. Data were assessed for normalcy using the Kolmogorov–Smirnov test. The Student t test was used for parametric comparisons between groups while the Mann–Whitney U test was used for nonparametric analysis. We used χ2 tests (for n ≥ 5) and Fisher exact tests (for n < 5) to compare proportions between study groups. Multiple linear regression was performed on the EQ VAS to control for preoperative scores. Significance was defined at p = 0.05.
Results
Baseline characteristics
Between September 2011 and May 2015, 55 participants were enrolled in the study and randomly assigned to a study arm. Given difficulties in acquiring MRI access in a timely fashion, we stopped enrolment and undertook analysis before we reached the target of 70 participants. One patient randomly assigned to the CI group did not receive surgery because of a clinical issue. The baseline characteristics of the study groups are presented in Table 1. All participants had a preoperative diagnosis of osteoarthritis. There were no significant differences in preoperative characteristics between the groups.
Baseline characteristics of the conventional and patient-specific instrumentation groups
Radiographic alignment
As shown in Table 2, there were no statistically significant differences in the mean postoperative HKA angle or in the number of mechanical alignment outliers (patients with an HKA angle more than ± 3° of the 180° target alignment) between the groups. Post-hoc power analysis for equivalence of the principal outcome metric of coronal alignment was 0.91, indicating that no clinically significant difference (± 3°) existed between the groups. As for component alignment, there was a significant difference in the tibial sagittal angle achieved as the PSI group was closer to the target of 3° than the CI group (3.8°± 3.1° v. 7.7°± 3.6°, respectively, p < 0.001). There were no other significant differences in component alignment between the groups.
Radiographic alignment in the conventional and patient-specific instrumentation groups
Function and satisfaction outcome questionnaires
Results of the functional and satisfaction outcome questionnaires are shown in Table 3. In preoperative PROMs, there was a significant difference between the CI and PSI groups for the EQ VAS. The remainder of the preoperative outcome measures were similar between the groups. For the postoperative results, at 1 year there was a statistically significant difference in the improvement in the EQ VAS, with a 20.2 ± 15.4 increase in score for the CI group compared with a 7.04 ± 22.4 increase for the PSI group. There were no other significant postoperative PROM findings. Multiple linear regression analysis revealed that preoperative EQ VAS scores were a confounding variable for postoperative EQ VAS improvement. After controlling for preoperative scores, there was no longer any difference in EQ VAS improvement between instrumentation groups at 1 year (p = 0.161).
Patient-reported outcome measures of the conventional and patient-specific instrumentation groups
Intraoperative and postoperative variables
There were no significant differences in surgical time, length of hospital stay or incidence of complications between the groups, as shown in Table 4. There were no cases in the PSI group in which the surgeon chose or required deviation from the PSI-directed plan. There was a trend toward increased surgical time in the PSI group, but this was not statistically significant. Six patients who participated in the study experienced complications following surgery. One participant in the CI group required lateral release with liner exchange 17 months after surgery because of extensor mechanism pain and weakness. The same patient ultimately underwent full revision for aseptic loosening of the patella and tibia 6 years after the initial surgery. One participant in each group was prescribed antibiotics for superficial wound infections with resolution and no further surgery required. One participant in the CI group underwent an isolated manipulation for arthrofibrosis. One participant in the PSI group underwent 2-stage revision beginning 15 months after the initial surgery and had an infection with Staphylococcus epidermidis. One participant in the PSI group underwent a manipulation in the early postoperative period and subsequently required staged revision for infection 22 months after the initial surgery and had an infection with Staphylococcus aureus. Although there were 2 revisions for infection in the PSI group and none in the CI group, the difference was not statistically significant (p = 0.23). One participant in the PSI group experienced a perioperative myocardial infarction.
Intra- and post-operative outcome metrics for the conventional and patient-specific instrumentation groups
Discussion
This study found no statistically significant differences in the accuracy of the mean HKA angle achieved or in the number of mechanical axis outliers between the CI and PSI groups. These results are both supported and refuted by similarly conducted randomized studies that compared CI with Visionaire PSI.2,9,17–19 A large multicentre randomized controlled trial by Abane and colleagues compared CI with the Visionaire PSI system and found no significant difference in the mean HKA angle or in the percentage of outliers.9 In another comparable study, Vide and colleagues also concluded that there was no significant difference in the mean HKA angle but did find a significant difference in the number of mechanical axis outliers. 2 As for component position, this study found that PSI participants achieved sagittal tibial alignment significantly closer to the desired posterior tibial slope of 3° than CI participants. A study by Vundelinckx and colleagues had a similar finding, as they also found improved tibial slope accuracy with Visionaire PSI.19 A study by Conteduca and colleagues, however, challenged this finding, as they found through computer navigation that extramedullary tibial guides were better than PSI in attaining the proper sagittal tibia component position.30 A recent meta-analysis of 44 studies, of which 13 assessed tibial sagittal alignment, concluded that PSI systems in general appear to produce more outliers in tibial sagittal alignment than CI.31 As there is heterogeneity in the data, it is unclear whether this finding encompasses all PSI systems or whether it is system dependent. The adjustable nature of extramedullary alignment of the tibial slope and surgeon variation in aligning the cutting block to native tibial slope may have increased the variation in the CI group in the present study. Overall, given the findings of this study and the conflicting results found in the literature, it is difficult to suggest that PSI offers any clear advantages to CI in achieving limb and component alignment in either plane.
Clinical outcome scores improved in both groups with no significant difference observed in the EQ-5D-3L, OKS, PCS, VAS for satisfaction, VAS for pain and UCLA Activity Score at 1 and 2 years postoperatively after we controlled for preoperative variation between groups. A study by Boonen and colleagues compared CI and PSI groups using a number of PROMs and also found no significant differences in scores between groups up to 2 years after surgery.32
A study by Hamilton and colleagues powered to determine differences in surgical times between CI and PSI found that a significant amount of time (4.4 min) was saved with CI.33 Although our study results were not significant, we found a trend to longer surgical times for the PSI group. Hamilton and colleagues proposed that the experience of the single surgeon participating in the study with CI combined with the novelty of the PSI contributed to the observed difference in surgical time in their study.33 A similar argument can be made for the surgeons in this study, as all were high-volume surgeons using CI, and given their level of experience, this may account for the differences in surgical time observed between groups. Chinnappa and colleagues also assessed the learning curve of another PSI system by comparing a single surgeon’s first 30 cases with the next 56 and found only a difference in surgical time from 85 to 78 minutes with no differences in alignment or other outcomes.34 Although these surgical times are significantly longer than the times in the present study, the difference is similar to what was observed between the CI and PSI groups. This further supports the conclusion that the trend to longer surgical times in the PSI group may have been part of the learning curve in the introduction of a new technology.
Length of stay in hospital did not differ significantly between groups. Results in the literature vary regarding length of hospital stay and PSI. A randomized controlled trial comparing CI and Visionaire PSI by Noble and colleagues found a significant decrease in the length of hospital stay with PSI,17 while a similarly conducted study by Vundelinckx and colleagues found no significant difference between groups.19 Length of hospital stay is influenced by multiple factors and therefore it is difficult to attribute surgical technique as the primary influence on the duration of the hospital stay.35
The potential cost savings associated with using PSI have been theorized as 1 of the main improvements over CI. Some of the financial benefits are believed to arise through a decrease in surgical time, a reduction in the number of instrument trays used and a decrease in the length of hospital stay.2,17 A study by Barrack and colleagues supported this hypothesis by showing that with PSI, a decrease in surgical time and instrument tray processing times led to modest cost savings.11 However, the savings obtained were far exceeded by the additional costs associated with PSI. The cost of the MRI, the custom instruments and the time it takes to review the preoperative template plan are additional expenses that must be considered when assessing the cost-utility of PSI.11,19 In Canada, the system cost for PSI blocks varies from approximately $500 to $1000. Additionally, the limited MRI imaging sequence without radiologist interpretation adds approximately $50 to $80 in additional health care system costs. As previously described, in our study we found that there were no significant differences in surgical times and length of hospital stay between PSI and CI (the number of instrument trays was not captured in this study). In addition, our study showed no significant differences in mechanical alignment or in PROMs between groups. Therefore, given the similarities in surgical efficiency and outcome measures between the groups in addition to the collective cost of PSI, it is unlikely that PSI is a cost-effective technology.
Strengths of this study include the randomized controlled design, blinding of the assessor, blinding of the patients and use of multiple PROMs to assess functional and health outcomes.
Limitations
There were some limitations to our study. Enrolment was hampered after initiation of the study by restricted access to preoperative MRI. We had to stop enrolling participants in the middle of the enrolment process for 14 months because of MRI wait-time reduction strategies before we were able to resume enrolment at a slowed pace. As a result, enrolment took far longer than originally anticipated and was stopped before we achieved the planned sample size. Despite this, the sample size that we obtained provided more than 90% power to detect a clinically significant difference in alignment. Owing to the added case-cost of using PSI, only 2 prestudy PSI cases were done by each surgeon. It is likely that this did not completely cover the learning curve of this technique; however, given the experience of the surgeons and the relatively small portion of the procedure represented by the difference between PSI and CI, the impact is believed to be small and restricted to the trend of increased surgical time in the PSI group. In addition, the x-ray measurements were done by only a single observer; however, the observer was blinded to the instrumentation used and performed a recheck of all measurements to ensure the methodology had not changed over the course of review.
Conclusion
Our study demonstrated that CI and PSI did not differ in achieving adequate coronal limb alignment. Visionaire PSI was significantly more accurate than CI in achieving the target tibial slope, but no other differences existed for component positioning and HKA alignment. There were also no significant differences between groups in terms of improvements in knee function, satisfaction and PROMs.
Acknowledgements
The authors acknowledge Sarah Tran and Trevor Gascoyne of the Orthopaedic Innovation Centre, Winnipeg, Man., for their assistance in executing this study. The authors also acknowledge Smith & Nephew for the in-kind donation of the Visionaire cutting blocks used in this study.
Footnotes
Competing interests: T. Turgeon and D. Hedden are consultants with Smith & Nephew. Smith & Nephew supplied the cutting blocks used in the study. No other competing interests were declared.
Contributors: T. Turgeon, C. Burnell, D. Hedden and E. Bohm designed the study. T. Turgeon, C. Burnell and D. Hedden acquired the data, which T. Turgeon and B. Cameron analyzed. T. Turgeon and B. Cameron wrote the article, which all authors reviewed and approved for publication. All authors agreed to be accountable for all aspects of the work.
- Accepted May 6, 2019.