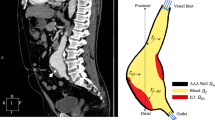
Abstract
Purpose
Abdominal Aortic Aneurysms (AAA) have extreme medical prevalence as an asymptomatic cause of death in developed countries. The probability of AAA rupture is promoted by the localized oxygen loss in the AAA wall which occurs in part because many AAAs contain a layer called intraluminal thrombus (ILT). Considering this strong clinical association, the purpose of this study is to investigate the key features that constitute to the oxygen diffusion, and therefore hypoxia in AAA.
Methods
A three-dimensional model of AAA containing ILT is created and numerical simulations are performed to simulate blood flow and oxygen distribution within the AAA. The model accounts for blood flow in the lumen and oxygen transport in the lumen, ILT, and arterial wall. The sub-model of the ILT is fully coupled with the wall sub-model as well as with the subdomain of the blood flow. The sensitivity of the oxygen flow with respect to the parameters of the problem is also analyzed.
Results
Model simulations are used to investigate the relation between AAA physical properties, hemodynamics, and oxygen concentration in different geometries of AAA. The results demonstrate that the diameter of the AAA bulge has little effect on the oxygen flow, but that the thickness of the ILT layer has a profound effect. Moreover, a significant sensitivity to the oxygen supply from vasa vasorum and its notable impact on oxygen transport within AAA are observed. The variability of the arterial wall oxygen concentration to the oxygen reaction rate remains however very low.
Conclusion
The presence of an ILT significantly impairs oxygen transport from the lumen to the wall. This study confirms that consideration of ILT size and anatomy may be important in considering the severity of a AAA, however, other parameters can also affect thrombus-mediated oxygen delivery within the aneurysmal wall.














Similar content being viewed by others
References
Adolph, R., D. A. Vorp, D. L. Steed, M. W. Webster, M. V. Kameneva, and S. C. Watkins. Cellular content and permeability of intraluminal thrombus in abdominal aortic aneurysm. J. Vasc. Surg. 25(5):916–926, 1997. https://doi.org/10.1016/S0741-5214(97)70223-4.
Ayyalasomayajula, A., J. P. Vande Geest, and B. R. Simon. Porohyperelastic finite element modeling of abdominal aortic aneurysms. J. Biomech. Eng. 132(10):104502, 2010. https://doi.org/10.1115/1.4002370.
Buerk, D. G., and T. K. Goldstick. Arterial wall oxygen consumption rate varies spatially. Am. J. Physiol. Heart Circ. Physiol. 243(6):H948–H958, 1982.
Buerk, D. G., and T. K. Goldstick. Oxygen tension changes in the outer vascular wall supplied by vasa vasorum following adenosine and epinephrine. J. Vasc. Res. 23(1):9–21, 1986.
Caputo, M., C. Chiastra, C. Cianciolo, E. Cutri, G. Dubini, J. Gunn, et al. Simulation of oxygen transfer in stented arteries and correlation with in-stent restenosis. Int. J. Numer. Method Biomed. Eng. 29(12):1373–1387, 2013. https://doi.org/10.1002/cnm.2588.
Chaikof, E. L., R. L. Dalman, M. K. Eskandari, B. M. Jackson, W. A. Lee, M. A. Mansour, et al. The Society for Vascular Surgery practice guidelines on the care of patients with an abdominal aortic aneurysm. J. Vasc. Surg. 67(1):2–77, 2018.
Fraser, K. H., S. Meagher, J. R. Blake, W. J. Easson, and P. R. Hoskins. Characterization of an abdominal aortic velocity waveform in patients with abdominal aortic aneurysm. Ultrasound Med. Biol. 34(1):73–80, 2008.
Grøndal, N., R. Søgaard, and J. S. Lindholt. Baseline prevalence of abdominal aortic aneurysm, peripheral arterial disease and hypertension in men aged 65–74 years from a population screening study (VIVA trial). Br. J. Surg. 102(8):902–906, 2015.
Haller, S. J., J. D. Crawford, K. M. Courchaine, C. J. Bohannan, G. J. Landry, G. L. Moneta, et al. Intraluminal thrombus is associated with early rupture of abdominal aortic aneurysm. J. Vasc. Surg. 67(4):1051–1058, 2018.
Hirsch, A. T., Z. J. Haskal, N. R. Hertzer, C. W. Bakal, M. A. Creager, J. L. Halperin, et al. ACC/AHA practice guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic). Circulation. 113(11):e463–e654, 2006.
Iannetti, L., G. D’Urso, G. Conoscenti, E. Cutri, R. S. Tuan, M. T. Raimondi, et al. Distributed and lumped parameter models for the characterization of high throughput bioreactors. PLoS ONE. 11(9):e0162774, 2016. https://doi.org/10.1371/journal.pone.0162774.
Kemmerling, E. M. C., and R. A. Peattie. Abdominal aortic aneurysm pathomechanics: current understanding and future directions. Adv. Exp. Med. Biol. 1097:157–179, 2018. https://doi.org/10.1007/978-3-319-96445-4_8.
Kolandavel, M. K., E. T. Fruend, S. Ringgaard, and P. G. Walker. The effects of time varying curvature on species transport in coronary arteries. Ann Biomed Eng. 34(12):1820–1832, 2006. https://doi.org/10.1007/s10439-006-9188-3.
Koole, D., H. J. Zandvoort, A. Schoneveld, A. Vink, J. A. Vos, L. L. van den Hoogen, et al. Intraluminal abdominal aortic aneurysm thrombus is associated with disruption of wall integrity. J. Vasc. Surg. 57(1):77–83, 2013.
Ku, D. N. Blood flow in arteries. Ann. Rev. Fluid Mech. 29(1):399–434, 1997.
Kuivaniemi, H., E. J. Ryer, J. R. Elmore, and G. Tromp. Understanding the pathogenesis of abdominal aortic aneurysms. Expert Rev. Cardiovasc. Therapy. 13(9):975–987, 2015. https://doi.org/10.1586/14779072.2015.1074861.
Liu, X., Y. Fan, X. Deng, and F. Zhan. Effect of non-Newtonian and pulsatile blood flow on mass transport in the human aorta. J. Biomech. 44(6):1123–1131, 2011. https://doi.org/10.1016/j.jbiomech.2011.01.024.
Ma, P., X. Li, and D. N. Ku. Heat and mass transfer in a separated flow region for high Prandtl and Schmidt numbers under pulsatile conditions. Int. J. Heat Mass Transf. 37(17):2723–2736, 1994.
Moll, F. L., J. T. Powell, G. Fraedrich, F. Verzini, S. Haulon, M. Waltham, et al. Management of abdominal aortic aneurysms clinical practice guidelines of the European society for vascular surgery. Eur. J. Vasc. Endovasc. Surg. 41:S1–S58, 2011.
Moore, J., and C. Ethier. Oxygen mass transfer calculations in large arteries. J. Biomech Eng. 119:469–475, 1997.
Oliver-Williams, C., M. Sweeting, G. Turton, D. Parkin, D. Cooper, C. Rodd, et al. Lessons learned about prevalence and growth rates of abdominal aortic aneurysms from a 25-year ultrasound population screening programme. Br. J. Surg. 105(1):68–74, 2018.
Olsen, P. S., T. Schroeder, K. Agerskov, O. Røder, S. Sørensen, M. Perko, et al. Surgery for abdominal aortic aneurysms: A survey of 656 patients. J. Cardiovasc. Surg. 32(5):636–642, 1991.
Polzer, S., and J. Bursa (eds.). Poroelastic model of intraluminal thrombus in FEA of aortic aneurysm: 6th World Congress of Biomechanics (WCB 2010). Singapore: Springer, 2010.
Polzer, S., T. C. Gasser, B. Markert, J. Bursa, and P. Skacel. Impact of poroelasticity of intraluminal thrombus on wall stress of abdominal aortic aneurysms. Biomed. Eng. Online. 11:62, 2012. https://doi.org/10.1186/1475-925X-11-62.
Polzer, S., T. C. Gasser, J. Swedenborg, and J. Bursa. The impact of intraluminal thrombus failure on the mechanical stress in the wall of abdominal aortic aneurysms. Eur.J. Vasc. Endovasc. Surg. 41(4):467–473, 2011.
Rappitsch, G., and K. Perktold. Computer simulation of convective diffusion processes in large arteries. J. Biomech. 29(2):207–215, 1996.
Raptis, A., M. Xenos, S. Dimas, A. Giannoukas, N. Labropoulos, D. Bluestein, et al. Effect of macroscale formation of intraluminal thrombus on blood flow in abdominal aortic aneurysms. Comput. Methods Biomech. Biomed. Eng. 19(1):84–92, 2016. https://doi.org/10.1080/10255842.2014.989389.
Riveros F, Martufi G, Gasser TC, Rodriguez JF, editors. Influence of intraluminal thrombus topology on AAA passive mechanics. Comput. Cardiol. 2013; 2013: IEEE.
Sakalihasan, N., J.-B. Michel, A. Katsargyris, H. Kuivaniemi, J.-O. Defraigne, A. Nchimi, et al. Abdominal aortic aneurysms. Nat. Rev. Dis. Prim. 4(1):1–22, 2018.
Salman, H. E., B. Ramazanli, M. M. Yavuz, and H. C. Yalcin. Biomechanical investigation of disturbed hemodynamics-induced tissue degeneration in abdominal aortic aneurysms using computational and experimental techniques. Front. Bioeng. Biotechnol. 7:111, 2019. https://doi.org/10.3389/fbioe.2019.00111.
Sonesson, B., T. Länne, F. Hansen, and T. Sandgren. Infrarenal aortic diameter in the healthy person. Eur.J. Vasc. Surg. 8(1):89–95, 1994.
Sun, N., J. H. Leung, N. B. Wood, A. D. Hughes, S. A. Thom, N. J. Cheshire, et al. Computational analysis of oxygen transport in a patient-specific model of abdominal aortic aneurysm with intraluminal thrombus. Br. J. Radiol. 821:S18–S23, 2009. https://doi.org/10.1259/bjr/89466318.
Swedenborg, J., M. I. Mäyränpää, and P. T. Kovanen. Mast cells: important players in the orchestrated pathogenesis of abdominal aortic aneurysms. Arterioscler. Thromb. Vasc. Biol. 31(4):734–740, 2011.
Takayama, T., and D. Yamanouchi. Aneurysmal disease: the abdominal aorta. Surg. Clin. 93(4):877–891, 2013.
Tanaka, H., N. Zaima, T. Sasaki, T. Hayasaka, N. Goto-Inoue, K. Onoue, et al. Adventitial vasa vasorum arteriosclerosis in abdominal aortic aneurysm. PLoS ONE. 8(2):e57398, 2013.
Tanaka, H., N. Zaima, T. Sasaki, M. Sano, N. Yamamoto, T. Saito, et al. Hypoperfusion of the adventitial vasa vasorum develops an abdominal aortic aneurysm. PLoS ONE 10(8):e0134386, 2015.
Virag, L., J. S. Wilson, J. D. Humphrey, and I. Karsaj. A computational model of biochemomechanical effects of intraluminal thrombus on the enlargement of abdominal aortic aneurysms. Ann. Biomed. Eng. 43(12):2852–2867, 2015. https://doi.org/10.1007/s10439-015-1354-z.
Vorp, D. A. Biomechanics of abdominal aortic aneurysm. J. Biomech. 40(9):1887–1902, 2007. https://doi.org/10.1016/j.jbiomech.2006.09.003.
Vorp, D. A., P. C. Lee, D. H. Wang, M. S. Makaroun, E. M. Nemoto, S. Ogawa, et al. Association of intraluminal thrombus in abdominal aortic aneurysm with local hypoxia and wall weakening. J. Vasc. Surg. 34(2):291–299, 2001. https://doi.org/10.1067/mva.2001.114813.
Vorp, D., W. Mandarino, M. Webster, and J. Gorcsan, III. Potential influence of intraluminal thrombus on abdominal aortic aneurysm as assessed by a new non-invasive method. Cardiovasc. Surg. 4(6):732–739, 1996.
Vorp, D., D. Wang, M. Webster, and W. Federspiel. Effect of intraluminal thrombus thickness and bulge diameter on the oxygen diffusion in abdominal aortic aneurysm. J. Biomech. Eng. 120(5):579–583, 1998.
Wang, D. H., M. S. Makaroun, M. W. Webster, and D. A. Vorp. Effect of intraluminal thrombus on wall stress in patient-specific models of abdominal aortic aneurysm. J. Vasc. Surg. 36(3):598–604, 2002.
Wolinsky, H., and S. Glagov. Comparison of abdominal and thoracic aortic medial structure in mammals. Circ. Res. 25(6):677–686, 1969.
Zakerzadeh, R., T. Cupac, and M. Durka. Oxygen transport in a permeable model of abdominal aortic aneurysm. Comput: Methods Biomech. Biomed. Eng, 2020. https://doi.org/10.1080/10255842.2020.1821193.
Zakerzadeh, R., and P. Zunino. A computational framework for fluid–porous structure interaction with large structural deformation. Meccanica. 54(1–2):101–121, 2019.
Acknowledgments
The Duquesne University departmental support and Faculty Development Fund for RZ is gratefully acknowledged. This research was supported in part by the University of Pittsburgh Center for Research Computing through the resources provided.
Author information
Authors and Affiliations
Contributions
RZ was responsible for project administration and designed the numerical simulations and methodology, wrote the first draft and revised the manuscript. TC, ND, and AG helped with data curation and visualization. All authors read and approved the submitted version of the manuscript.
Corresponding author
Additional information
Associate Editor Kerem Pekkan oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zakerzadeh, R., Cupac, T., Dorfner, N. et al. Coupled Hemodynamics and Oxygen Diffusion in Abdominal Aortic Aneurysm: A Computational Sensitivity Study. Cardiovasc Eng Tech 12, 166–182 (2021). https://doi.org/10.1007/s13239-020-00508-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-020-00508-5