Abstract
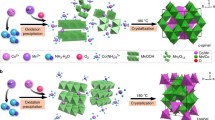
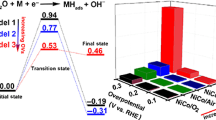
Spinel catalysts have been widely explored for the electrochemical oxygen reduction reaction (ORR) and oxygen evolution reaction (OER). To consolidate the understanding on electrocatalysis by spinel family, intermediate spinels should be deliberately examined because most spinel oxides are of intermediate structure. Here, we report an investigation on the ORR and OER performance of intermediate spinel MnFe2O4. The modulation of cation oxidation state and inversion degree of spinel MnFe2O4 were achieved by a simple annealing process. X-ray absorption spectroscopy analysis reveals that the Mn occupancy in octahedral sites varied from 0.25 ~ 0.41 and Mn cations were oxidized from 2+ to 3+ with increasing temperature treatment. Convinced by the leading role of octahedral-geometric, we further reveal the role of Mn oxidation state through normalizing the activity to active Mn[Oh] site number. Our findings clearly indicate that Mn3+ was more catalytically active than Mn2+ in catalyzing ORR and OER.

Both Mn occupancy in octahedral site and its oxidation state play dominant roles in determining the catalytic activities of spinel manganese ferrites toward oxygen electrocatalysis.





Similar content being viewed by others
References
J. Suntivich, H.A. Gasteiger, N. Yabuuchi, H. Nakanishi, J.B. Goodenough, Y. Shao-Horn, Nat. Chem. 3, 546 (2011)
Y. Zhu, W. Zhou, Y. Chen, J. Yu, M. Liu, Z. Shao, Adv. Mater. 27, 7150 (2015)
Y. Jiao, Y. Zheng, M. Jaroniec, S.Z. Qiao, J. Am. Chem. Soc. 136, 4394 (2014)
Y. Guo, Y. Tong, P. Chen, K. Xu, J. Zhao, Y. Lin, W. Chu, Z. Peng, C. Wu, Y. Xie, Adv. Mater. 27, 5989 (2015)
H. Zhu, S. Zhang, Y.-X. Huang, L. Wu, S. Sun, Nano Lett. 13, 2947 (2013)
C. Wei, Z. Feng, G.G. Scherer, J. Barber, Y. Shao-Horn, Z.J. Xu, Adv. Mater. (2017)
C. Wei, Z. Feng, M. Baisariyev, L. Yu, L. Zeng, T. Wu, H. Zhao, Y. Huang, M.J. Bedzyk, T. Sritharan, Z.J. Xu, Chem. Mater. 28, 4129 (2016)
D. Carta, M.F. Casula, A. Falqui, D. Loche, G. Mountjoy, C. Sangregorio, A. Corrias, J. Phys. Chem. C 113, 8606 (2009)
D. Levy, L. Pastero, A. Hoser, G. Viscovo, Solid State Commun. 201, 15 (2015)
J. Li, H. Yuan, G. Li, Y. Liu, J. Leng, J. Magn. Magn. Mater. 322, 3396 (2010)
X. Wang, Y. Liu, T. Zhang, Y. Luo, Z. Lan, K. Zhang, J. Zuo, L. Jiang, R. Wang, ACS Catal. (2017)
T.W. Kim, M.A. Woo, M. Regis, K.S. Choi, J. Phys. Chem. Lett. 5, 2370 (2014)
W. Guangping, W.D. Jun Wang, L.L. Yao Nie, X. Qi, S. Chen, Z. Wei, Angew. Chem. Int. Ed. 54, 1 (2015)
C.S. Zhichuan Xu, Y. Hou, H. Gao, S. Sun, Chem. Mater. 21, 1778 (2009)
D. Carta, M.F. Casula, G. Mountjoy, A. Corrias, Phys. Chem. Chem. Phys. 10, 3108 (2008)
H. Zhu, S. Zhang, Y.X. Huang, L. Wu, S. Sun, Nano Lett. 13, 2947 (2013)
C.M. Sorensen, K.J. Klabunde, A. Kostikas, Phys. Rev. B 54, 9288 (1996)
D. Carta, M.F. Casula, P. Floris, A. Falqui, G. Mountjoy, A. Boni, C. Sangregorio, A. Corrias, Phys. Chem. Chem. Phys. 12, 5074 (2010)
K. Vamvakidis, M. Katsikini, D. Sakellari, E.C. Paloura, O. Kalogirou, C. Dendrinou-Samara, Dalton Trans. 43, 12754 (2014)
H. Dau, P. Liebisch, M. Haumann, Anal. Bioanal. Chem. 376, 562 (2003)
H. Yamaguchi, A. Yamada, H. Uwe, Phys. Rev. B 58, 8 (1998)
M. Newville, Rev. Mineral. Geochem. 78, 33 (2014)
Y. Du, J.-O. Wang, L. Jiang, L.S. Borgna, Y. Wang, Y. Zheng, T. Hu, J. Synchrotron Radiat. 21, 756 (2014)
C. Wei, L. Yu, C. Cui, J. Lin, C. Wei, N. Mathews, F. Huo, T. Sritharan, Z. Xu, Chem. Commun. 50, 7885 (2014)
Y. Lee, J. Suntivich, K.J. May, E.E. Perry, Y. Shao-Horn, The Journal of Physical Chemistry Letters. 3, 399 (2012)
J. Suntivich, K.J.A. May, H. Gasteiger, J.B. Goodenough, Y. Shao-Horn, Science 334, 1383 (2011)
A. Grimaud, C.E. Carlton, M. Risch, W.T. Hong, K.J. May, Y. Shao-Horn, J. Phys. Chem. C 117, 25926 (2013)
E. Vago, E. Calvo, J. Electroanal. Chem. 339, 41 (1992)
K.A. Stoerzinger, M. Risch, B. Han, Y. Shao-Horn, ACS Catal. 5, 6021 (2015)
Y. Du, Y. Zhu, S. Xi, P. Yang, H.O. Moser, M.B.H. Breese, A. Borgna, J. Synchrotron Rad. 22, 839–843 (2015)
Acknowledgements
The authors thank the Facility for Analysis, Characterization, Testing and Simulation (FACTS) in Nanyang Technological University for materials characterizations. Authors appreciate the XAFCA beamline [30] of the Singapore Synchrotron Light Source for the XAFS characterization.
Funding
This work was supported by the Singapore Ministry of Education Tier 2 Grant (MOE2015-T2-1-020) and the Singapore National Research Foundation under its Campus for Research Excellence and Technological Enterprise (CREATE) program.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
ESM 1
(DOCX 361 kb)
Rights and permissions
About this article
Cite this article
Zhou, Y., Du, Y., Xi, S. et al. Spinel Manganese Ferrites for Oxygen Electrocatalysis: Effect of Mn Valency and Occupation Site. Electrocatalysis 9, 287–292 (2018). https://doi.org/10.1007/s12678-017-0429-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-017-0429-z