Abstract
Background
Current diagnostic criteria for hypoxic–ischemic encephalopathy in the early hours lack objective measurement tools. Therefore, this systematic review aims to identify putative molecules that can be used in diagnosis in daily clinical practice (PROSPERO ID: CRD42021272610).
Data sources
Searches were performed in PubMed, Web of Science, and Science Direct databases until November 2020. English original papers analyzing samples from newborns > 36 weeks that met at least two American College of Obstetricians and Gynecologists diagnostic criteria and/or imaging evidence of cerebral damage were included. Bias was assessed by the Newcastle–Ottawa Scale. The search and data extraction were verified by two authors separately.
Results
From 373 papers, 30 met the inclusion criteria. Data from samples collected in the first 72 hours were extracted, and increased serum levels of neuron-specific enolase and S100-calcium-binding protein-B were associated with a worse prognosis in newborns that suffered an episode of perinatal asphyxia. In addition, the levels of glial fibrillary acidic protein, ubiquitin carboxyl terminal hydrolase isozyme-L1, glutamic pyruvic transaminase-2, lactate, and glucose were elevated in newborns diagnosed with hypoxic–ischemic encephalopathy. Moreover, pathway analysis revealed insulin-like growth factor signaling and alanine, aspartate and glutamate metabolism to be involved in the early molecular response to insult.
Conclusions
Neuron-specific enolase and S100-calcium-binding protein-B are potential biomarkers, since they are correlated with an unfavorable outcome of hypoxic–ischemic encephalopathy newborns. However, more studies are required to determine the sensitivity and specificity of this approach to be validated for clinical practice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Perinatal asphyxia (PA) can lead to severe brain injury and is the most frequent cause of hypoxic–ischemic encephalopathy (HIE), occurring in 1–8/1000 live births [1]. Several conditions might lead to an interruption of the blood flow to the brain, resulting in an insufficient supply of oxygen and nutrients to the brain required to maintain the high energy demands of this organ [2]. Briefly, after the initial insult, energy failure results in an impairment of active membrane transport and, consequently, membrane depolarization and glutamate release. Its accumulation in the synaptic cleft leads to increased excitotoxicity, culminating in cytotoxic edema, activation of inflammatory and apoptotic pathways, and finally neuronal death [3]. These events may lead to permanent sequelae in the neonatal brain, namely, epilepsy, cerebral palsy, mental disability, motor and sensorial impairment, or even death [4].
There are no accurate and objective tools with high sensitivity and specificity to diagnose newborns suffering from HIE. Some complementary blood tests have been proposed to evaluate liver and renal function and support the diagnosis, but they lack neuronal specificity [5]. According to the American College of Obstetricians and Gynecologists (ACOG) [6], if more than one of the following criteria is met, the newborn is more likely to suffer from a peripartum hypoxic–ischemic event: (1) appearance, pulse, grimace, activity, respiration (APGAR) score below 5 at 5 and 10 minutes; (2) fetal umbilical artery pH less than 7.0 and/or base deficit equal to or greater than 12 mmol/L; (3) neuroimaging evidence of acute brain injury seen on magnetic resonance imaging (MRI) or magnetic resonance spectroscopy (MRS), and (4) presence of multiorgan failure. However, imaging is usually performed in the first two weeks of life and is not suitable for a rapid diagnosis [5].
The therapeutic window to treat HIE is limited to the first 6 hours of life, before the beginning of inflammatory and apoptotic pathways [7]. Presently, therapeutic hypothermia (TH) is the treatment standard for moderate to severe cases of HIE, consisting of cooling either the newborn’s whole-body temperature (keeping it between 32 °C and 34 °C) or selectively the head for up to 72 hours. This approach aims to slow the metabolic rate and the accumulation of inflammatory cytokines, lowering the activation of intracellular pathways leading to programmed cell death. Furthermore, innovative treatments are emerging, including drugs, such as topiramate, erythropoietin, and stem cells, which are not yet used as standard guideline treatments [8]. The lack of a definitive test to diagnose HIE might lead to a misdiagnosis and a lack of proper treatment choices that can have an irretrievable impact on these neonates’ future.
Several studies have been published in recent decades proposing hypothetical biomarkers for HIE [5, 9]. Nevertheless, to our knowledge, there is no review on the literature that collects all these data. Therefore, this systematic review proposes to critically assess potential biomarkers for the diagnosis of term newborns who have been diagnosed with HIE in accordance with ACOG criteria and/or MRI brain injury evidence.
Methods
The study design was registered on PROSPERO on 1st October 2021 (ID: CRD42021272610) [10]. In addition, this review was written in accordance with PRISMA guidelines [11]. The search strategy, study eligibility, and quality assessment were performed by IC and MC, while IC and MR performed data extraction. The evaluation was performed independently, and disagreements were resolved by consensus.
Search strategy
An article search was conducted in three distinct databases until November 11, 2020: PubMed, Web of Science, Science Direct, and OpenGrey. Since HIE terminology is not consensual, four different terms were used: “neonatal brain injury”, “neonatal encephalopathy”, “hypoxic–ischemic encephalopathy” and “neonatal hypoxic–ischemic encephalopathy”. These terms were combined with the preposition and with the terms “biomarker*”, “proteomic*”, “metabolomic*”, restricting it to the title and abstract fields. The methods are described in detail in the supplementary material.
Study eligibility
Selected articles were subjected to abstract evaluation and three selection phases. The first approach aimed to categorize the results by document type, language, species of the samples studied, and biomarker type. Only original English papers in which the research was focused on biochemical HIE biomarkers in human samples were selected for method evaluation. The studies were then classified according to study type, sample size, sample type, association with other pathologies, gestational age (GA) or age, disease, sample collection time, therapy, outcome assessment, and association of the biomarkers with multiorgan failure. Only studies regarding term newborns collected in the first 72 hours of life were selected for a diagnostic criteria analysis. Moreover, studies analyzing cerebrospinal fluid (CSF) were excluded, since its collection from newborns may be considered unethical in many countries.
Since diagnosis criteria for HIE are not standardized in all studies, to analyze a homogenous population, the diagnostic criteria applied were assessed in each study: APGAR score, fetal acidemia, MRI and multiorgan failure. Studies that matched at least two ACOG diagnostic criteria or had neuroimaging evidence of brain injury were selected for quality assessment and data extraction.
Quality assessment
The quality of each study was evaluated by the Newcastle–Ottawa Scale (NOS) [12]. In accordance with the authors’ guidelines, different scales were applied depending on the study type (cohort or case–control). The scores for selection, comparability, and outcomes are presented separately.
Data extraction
Population characteristics were analyzed to infer the homogeneity of the populations being reviewed in this manuscript. Information about the study location, type of study, gestational age and/or birth weight, diagnosis criteria, HIE severity assessment, complementary diagnostic exams, therapeutic hypothermia, sample size, sample type, and the biomarker described in the study was extracted. In addition, information about the biomarker, namely, the biomarker type, the technique used to analyze the biomarker, the sample size of each group, collection time, and P value (when available), was also extracted. The extracted data is available in Tables 1-3 and in the supplementary material.
Data analysis
Venn diagrams were generated on InteractiVenn [13]. A UniProt accession number, Human Metabolome Database (HMBD) or GeneCard code was manually attributed to identified proteins, metabolites, or genes, respectively. Proteins and metabolites identified in serum and plasma samples were subjected to further analysis. Protein gene ontology (GO) analysis was performed on the DAVID Bioinformatic Database [14], and images were generated using the ggplot2 R package [15]. Metabolite pathway and mixomic analyses were performed on MetaboAnalyst [16].
Results
Literature search and study selection
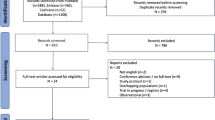
The PubMed search resulted in 279 hits and 227 unique records. The Web of Science search resulted in 233 hits and 121 unique records. The Science Direct search resulted in 145 hits and 93 unique records. After duplicate removal, a list of 373 unique records was obtained (Fig. 1a, b). From these, a total of 240 records were excluded considering nonoriginal records (n = 110), non-English records (n = 15), nonhuman studies (n = 73) and nonbiochemical, therapeutic response, genetic, pH, or associated with other disease biomarker studies (n = 140), resulting in 133 records for evaluation of the methods. During this selection phase, three articles were reclassified as review articles, one was reclassified as an imaging biomarker article, and four focused on therapeutic response biomarkers. Then, studies analyzing CSF samples were excluded (n = 7), because the collection of this fluid is not a common clinical practice in newborns. Works studying children, adults, and newborns < 36 week GA were also excluded (n = 31), since this review focuses on HIE biomarkers for term newborns. Finally, articles in which sample collection was not within the first 72 hours after birth (n = 15) were excluded, as this review focuses on identifying biomarkers of HIE to be used as an early diagnostic tool. These steps identified 81 records, of which diagnostic criteria were analyzed. It was not possible to obtain diagnosis criteria information in one of the reports; therefore, it was excluded. Studies that matched at least two ACOG diagnostic criteria or had neuroimaging evidence of acute brain injury by MRI or MRS (n = 29) were selected. Regarding the manual search, one study matched all eligibility criteria and was considered for further analysis. In conclusion, 30 studies were included in this systematic review [17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46] (Fig. 1a).
Database search results and summary of population characteristics. a PRISMA 2020 flow diagram. Only English manuscripts that analyzed human samples collected within 72 h (excluding CSF) and studied biochemical biomarkers associated with HIE were included. In addition, selected studies had to match at least two ACOG diagnosis criteria or present neuroimaging evidence of acute brain ischemia. b Common articles between PubMed, Web of Science, and Science Direct. Only 12 articles were common between the three databases. c and d Concerning population characteristics, the majority of selected studies were performed in Europe and America and published after 2014. f, g Almost all studies applied whole-body hypothermia and had MRI data available. h Studies lacked uniformity in comparison groups, but the severity was preferably assessed by the Sarnat scoring system. e, j Technique most prevalently used is ELISA, while plasma was the fluid with more identifications. k Summary of NOS scoring. CSF cerebrospinal fluid, ACOG American College of Obstetricians and Gynecologists, DBS dried blood spots, ELISA enzyme linked immunosorbent assay, miRNA microRNA, NIRS near-infrared spectroscopy, MRS magnetic resonance spectroscopy, FDNIRS–DCS frequency-domain near-infrared spectroscopy–diffuse correlation spectroscopy, MS mass spectrometer, GC–MS gas chromatography–mass spectrometer, ESI–MS electro spray ionization MS, ILMA immunoluminometric assay, SIMOA single molecular array, Epo erythropoietin, RT–PCR reverse transcription–polymerase chain reaction, MRI magnetic resonance imaging, EEG electroencephalogram, PET positron emission computed tomography, CRP C-reactive protein, NOS Newcastle–Ottawa scale
Quality assessment
Considering the NOS, no case–control study was considered to be at risk of bias (Supplementary Tables 1–2). Regarding cohort studies, one article was considered to be at high risk of bias (score ≤ 3), and four were at medium risk (score < 7). Five articles were identified as being potentially biased (Fig. 1k). These articles were not excluded from further analysis.
Studies and population characteristics
The population characteristics are summarized in Table 1, and the studies included were published between 2002 and 2020, despite the majority being released after 2014 (Fig. 1d). In addition, most studies took place in Europe and America (Fig. 1c) and are prospective studies. Regarding population characteristics, all studies included newborns older than 36 weeks of gestation that matched at least two ACOG diagnosis criteria or neuroimaging evidence of brain injury to allow studying a more homogeneous population. Moreover, almost all studies used whole-body hypothermia as a therapeutic approach and MRI as a complementary exam (Fig. 1f, g).
Potential biomarkers were identified in the cord blood, plasma, serum, whole blood, dried blood spots (DBS), and urine (Tables 2, 3 and Supplementary Tables 4–7). Interestingly, some studies used advanced cerebral imaging techniques to address the behavior of specific molecules during TH, while enzyme linked immunosorbent assay (ELISA) was the most commonly used technique (Fig. 1e). Although metabolites have a high number of identifications (due to the use of high throughput techniques) (Fig. 1j), most studies have focused their attention on proteins. Nevertheless, recent reports indicate that RNA and microRNA (miRNA) are emerging as possible diagnostic targets.
Advanced cerebral imaging techniques are emerging as a therapeutic response monitoring approach
A small number of studies have evaluated the therapeutic response of newborns to hypothermia by evaluating the redox state of cytochrome oxidase [26, 41] or hemoglobin oxygenation [26, 30, 41] using near-infrared spectroscopy (NIRS) (Supplementary Table 3). In addition, the lactate/N-acetylaspartate ratio (assessed by 1H MRS) was suggested as a promising severity predictor for HIE [26, 41]. However, additional studies are needed for more robust conclusions.
Several proteins as candidates for the diagnosis of hypoxic–ischemic encephalopathy are altered in various body fluids
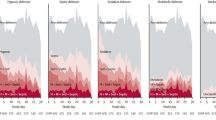
The proteins identified in more than one body fluid and/or cited in more than one study are summarized in Fig. 2a. While serum appears to be the most studied sample type, in which eleven different proteins were identified as potential biomarkers, only one protein was identified in urine—S100-calcium-binding protein-B (S100B). This protein was reported to be elevated in newborns' urine, presenting an unfavorable outcome in the first 48 hours of life [35] and severe cases in the first 24 hours of life [44]. Accordingly, higher S100B serum levels during hypothermia (at 72 hours) were associated with a worse prognosis [28, 36] and even death [28, 35, 36]. For glial fibrillary acidic protein (GFAP), data available from the umbilical cord are not consensual: one study reported no differences between control and HIE [17], while another describes that elevated GFAP levels are significantly different in infants who developed severe brain injury compared to mild HIE [18]. Although no differences were found between control and HIE on plasma samples during TH and on rewarming [29], GFAP serum levels were described to be significantly higher in infants with abnormal MRI during the first 24 hours [17, 25]. In severe cases of HIE, ubiquitin C-terminal hydrolase L1 (UCHL-1) levels were described to be increased in umbilical cord plasma, but no differences were found when comparing moderate and severe HIE groups with the mild HIE group [18]. It is also unclear if serum levels of this protein are associated with a worse prognosis, since no differences were found between the HIE and control groups [22], but the levels were described to be elevated at 6 hours and 72 hours in newborns with severe MRI brain injury when compared with those who did not develop or developed a mild brain injury [25]. Interestingly, newborns with HIE showed higher levels of plasma neuron-specific enolase (NSE) before TH than controls [29]. Increased serum levels of NSE in the first 72 hours were also found to be positively correlated with an unfavorable outcome [28, 35, 36], whereas one study did not find differences between newborns with different severity grades [39], and another did not have a control group [21].
Proteins and metabolites identified as potential biomarkers. a Representation of biomarkers that were present in more than one fluid and/or more than one study. Green, red and yellow triangles represent studies with evidence of significantly increased, decreased, or altered levels of the molecule in the HIE group. Blue circles represent molecules without significant differences, and on gray squares, no comparison was performed. Plasma and serum samples were then combined for pathway analysis. b, c Gene ontology analysis of proteins, namely, biological process and molecular function. Altered pathways were identified using a metabolite enrichment analysis (d) and a combined approach of protein and metabolite analysis (mixomics approach) (e). f Alterations in the alanine, aspartate and glutamate pathways were identified, where metabolites are highlighted in green and proteins in blue. Balls represent metabolites, and squares represent proteins. S100B S100-calcium-binding protein-B, DBS dried blood spots, GFAP glial fibrillary acidic protein, UCHL-1 ubiquitin C-terminal hydrolase L1, NSE neuron-specific enolase, IL interleukin, CRP C-reactive protein, VEGF vascular endothelial growth factor, ALT alanine aminotransferase, AST aspartate aminotransferase, TNF-α tumor necrosis factor α, GO Gene Ontology
Concerning the identified cytokines, no significant differences were found either in the umbilical cord serum [18] or the DBS [32]. Particularly for IL-6 serum levels, no control group was available in the study [33]. No comparisons were performed on erythropoietin levels in plasma or DBS [32]. However, a study reported that erythropoietin was increased in serum at 48 hours in newborns with moderate/severe HIE when compared to those who did not develop or developed mild HIE [38]. In addition, this study associated increased levels of erythropoietin at 72 hours with death [38]. C-reactive protein (CRP) serum levels and their correlation to an outcome are unclear, since one study did not find significant differences [34], and other studies did not have control groups to compare [33], or their levels were associated with microcirculatory issues [37]. Vascular endothelial growth factor (VEGF) serum levels were decreased in newborns with moderate/severe injury at 24 hours [38], a tendency also verified in plasma samples, where VEGF-C was decreased at 24 hours compared to controls [42]. Levels of VEGF in umbilical cord serum did not vary significantly [18].
Finally, alanine aminotransferase (ALT) serum levels were increased in HIE newborns with an abnormal electroencephalogram (EEG) in the first 6 hours of life [34] and continued to increase at 48 hours compared to the control group [40]. Aspartate aminotransferase (AST) levels were also reported to be elevated in newborns with HIE at 48 hours [40], but no difference was found in the first 6 hours of life [34]. However, it should be taken into consideration that they lack neurological specificity [47].
Together, these data suggest that additional studies are still required to corroborate the findings presented in this review regarding GFAP, UCHL-1, ALT, and VEGF. Concerning urinary S100B, receiver operating characteristic (ROC) curves show high sensitivity and specificity to predict death and short outcomes [35]. In addition, S100B and NSE serum levels also showed good predictive power for short- [28] and long-term outcomes [36]. Therefore, these two proteins should be validated to provide further support in the diagnosis of HIE.
Metabolites are promising candidates for the diagnosis of hypoxic–ischemic encephalopathy but require further studies. The metabolites identified and altered in more than one body fluid are summarized in Fig. 2a. Plasma is the sample type that reported more biomarkers, while umbilical cord blood metabolites were only studied by one research group. The most studied metabolite was lactate, which was reported to be increased in urine in the first 6 hours of life on HIE compared to controls [43]. In addition, HIE newborns with a severe phenotype or pathological MRI showed increased lactate levels in the cord blood [45] and plasma [24] at birth, respectively. However, the levels of this metabolite were reported not to change significantly in whole blood [27] or serum [34] in the first hours of life. Creatinine and urea are reported to be elevated in serum samples of the HIE group at 48 hours [40] but not in whole blood at birth [44]. In plasma, creatinine was reported to be significantly altered [24].
Interestingly, glucose levels were reported to be significantly increased in the serum of newborns in the first hours of life with severe injury or abnormal EEG [34, 46], but Bersani and his team did not find differences in plasma levels [44]. Last, glutamine, succinate, pyruvate and α-ketoglutarate urine levels were found to be decreased in HIE patients in the first 6 hours of life [43], whereas in plasma, glutamine and α-ketoglutarate were reported to be altered at 48 hours and 72 hours, and succinate was altered at 24 hours, 48 hours, and 72 hours [24]. Regarding pyruvate, its levels were elevated at 72 hours in newborns who presented a pathological MRI [24]. Furthermore, there are studies that propose ratios of metabolites as putative biomarkers. In urine, the lactate/creatinine ratio was described to be significantly elevated in the first 24 hours of life [23], but no differences were found in the lactate/pyruvate ratio [24] in plasma or the free/total carnitine ratio in DBS [21]. Inconsistencies and the reduced number of studies impaired the identification of any metabolite as a putative biomarker. Nevertheless, lactate is a candidate that should be explored in future studies along with new in-depth screenings, considering that it is a systemic severity marker but not neuronal-specific [48].
New approaches to identify other classes of biomarkers
MicroRNAs (miRNAs) were studied as potential biomarkers [31]. However, no differences were found between HIE patients with favorable and unfavorable outcomes in Let7b, miR-21, miR-29b, miR-124, and mir-155 levels in DBS. Alterations in the number of blood cells were also evaluated as an approach to identify biomarkers. Nucleated red blood cells in the cord blood were increased in newborns with moderate/severe HIE [45], but no differences were found in neutrophils or white blood cells in the whole blood [33, 40]. Platelet levels did not show significant differences when comparing newborns with high CRP levels [33] but were decreased in the HIE group compared to the control group [40]. Furthermore, electrolyte serum levels were also studied. One study showed that sodium, calcium, and selenium levels were decreased in the HIE group, while potassium levels were increased [40]. Finally, studies concerning alterations in the abundance of RNA for several inflammation markers were also reported. Proliferator-activated receptor gamma (PPARG), matrix metallopeptidase 9 (MMP-9), interleukin (IL)-8, heat shock protein family A (Hsp70) member 1A (HSPA1A), and toll-like receptor 8 (TLR8) were found to be increased in the whole blood of the HIE group, while C–C motif chemokine receptor 5 (CCR5) was decreased [20]. Since these approaches to identify biomarkers are recent, due to the lack of corroborating evidence from different authors, no objective conclusion can be drawn about the use of the aforementioned analytes as potential biomarkers.
Plasma and serum potential biomarkers showed altered pathways in hypoxic–ischemic encephalopathy
Pieces of evidence from plasma and serum were combined for further pathway analysis to highlight potential mechanisms. Gene Ontology (GO) of extracted proteins showed several biological processes and molecular functions associated with inflammatory responses, as well as insulin-growth factor pathways (Fig. 2b, c). Specifically, the majority of the proteins associated with these ontologies were decreased in HIE newborns [42]. Concerning metabolite pathways, arginine and proline metabolism, urea cycle and lysine degradation were the most significantly altered (Fig. 2d). Finally, integrating protein and metabolite data, two pathways were identified as being modified in HIE: alanine, aspartate and glutamate metabolism and arginine and proline metabolism (Fig. 2e, f).
Discussion
Presently, no accurate tools are available to diagnose HIE immediately after birth and quickly define the best therapeutic approach. In addition, the guidelines for diagnosing HIE are not standardized among all pediatric centers. While verifying the eligibility criteria of each study, we found huge discrepancies between the parameters applied to diagnose HIE, assessment of severity, and time of sample collection. Together, this reinforces the need to establish standard diagnostic criteria worldwide. In this review, to reduce bias, the population was homogenized by (1) matching at least two ACOG diagnosis criteria and (2) having neuroimaging evidence of brain injury, since these data are unequivocal proof of brain damage. In addition, potentially biased studies were not excluded, since they did not focus on the major findings of this review.
This systematic review summarized the potential biomarkers for HIE. Reported results lack high-throughput screenings, which hampers the identification of a larger number of putative biomarkers. Briefly, serum is the most cited fluid, and proteins are the major candidates with more consistent results among the different studies. In particular, NSE and S100B were identified as potential biomarkers for HIE. Interestingly, not only these proteins but also UCHL-1 and GFAP were described as potential biomarkers for traumatic brain injury in adults [49], since they are also involved in brain damage mechanisms. NSE is a brain and peripheral neuroendocrine-specific enolase that is highly expressed in neurons [50]. The postinsult collapse of the plasma membrane, as after a perinatal asphyxia event, could cause the release of this protein to peripheral fluids. In particular, after ischemic stroke, NSE protein levels were positively correlated with the extent of brain damage [51]. However, it should be considered that altered levels of this protein might also be associated with the diagnosis of small cell lung cancer, among others [52]. S100B is a calcium-binding protein expressed by glia, especially astrocytes [53]. This protein is associated with intracellular structures, but it is also secreted, playing a role in cell survival (in nanomolar concentrations), apoptosis, lipid peroxidation (in micromolar concentrations) and cytokine production [53, 54]. In addition, a highlighted pathway in GO analysis was the insulin-like growth factor signaling pathway (Fig. 2b, c). Although a single research group [42] studied the identified proteins, this is a promising target, since Insulin-like growth factor 1 (IGF-1) has already been tested as a therapeutic approach, exhibiting good outcomes in a rat HIE model [55].
Regarding the direct inflammatory response, no differences were found in the analysis of a panel of cytokines at the protein level [18, 32]. However, when analyzing the RNA levels of inflammatory markers, five were increased, and one was decreased [33]. GO analysis also identified alterations in several inflammatory pathways (Fig. 2b, c). However, some proteins have opposite tendencies, reinforcing the need to clarify the relevance of these molecules in the diagnosis of HIE. Nevertheless, it should be taken into consideration that the elevation of IL-6, for example, has been described to be associated with neonatal sepsis [56], which could lead to a misleading diagnosis.
The alanine, aspartate, and glutamate pathways were found to be altered in newborns with HIE. These metabolites were already described to be increased in brain tissue in a rat model of traumatic brain injury [57]. However, it should be taken into consideration that dysregulation of alanine transaminase was associated with liver dysfunction in HIE [58], enhancing the need to consider systemic biomarkers. Glutamate and alanine were also described to be elevated in the CSF in an HIE piglet model [58], while an excitotoxicity mouse model identified proline and arginine as players in response to injury [59]. Based on these pieces of evidence, future studies should focus on the characterization of these pathways in HIE.
Studies using advanced imaging techniques, such as NIRS and 1H MRS, are emerging as promising noninvasive approaches to monitor newborns’ response to TH [26, 27, 30, 37, 41]. However, they refer to a low number of publications, focusing on a low number of patients and without healthy controls, and were classified as potentially biased according to the NOS scale. Although these techniques can provide more information about the metabolic state of the newborns, access to these specialized techniques might be easier at reference centers to treat HIE but not in all maternities due to financial and logistical reasons.
One of the weak points of this review is the lack of studies with healthy newborn controls (Fig. 1i). Due to ethical reasons, it is not possible to obtain samples from healthy newborns at several timepoints. As an alternative, studies use non-neurological brain-injured newborns or newborns who have suffered an episode of perinatal asphyxia but did not develop/or developed a mild brain injury. In either case, using these populations as controls can bias the conclusions. Another limitation is the lack of uniformization of the groups between the studies (Fig. 1i), which makes it more difficult to compare studies. Likewise, the lack of uniformity of sample collection time, which might be influenced by hypothermia, hindered drawing conclusions. Unfortunately, some of the studies analyzed lack transparency on the methodologies and the availability of raw data, which compromised data extraction and further analysis of the published data.
It should be noted that only a small number of studies performed screenings, which limits the amount of information extracted from the samples and, therefore, reduces the chances of identifying a biomarker. In addition, the lack of raw data available (even after direct request) impaired a more detailed analysis to determine the sensitivity and specificity of the identified biomarkers and assess their predictive value to diagnose HIE and/or predict its severity. Therefore, future studies should present a higher consistency in the diagnosis criteria, establishment of groups, preferably using healthy controls, and sample collection time, so that data presented in this manuscript can be corroborated and finally get to a routine clinical application.
In conclusion, elevated serum levels of NSE and S100B correlated with a worse prognosis in newborns suffering from HIE. Nevertheless, future studies should focus on determining the sensitivity and specificity of these molecules before entering clinical practice. In addition, since other molecules were identified as potential biomarkers, such as GFAP, UCHL1, ALT, glutamate and lactate, we suggest that future studies focus on identifying a panel of biomarkers instead of a standalone biomarker.
Data Availability
All the data is provided on the tables of the manuscript, as well as in the supplementary data.
References
Kurinczuk JJ, White-Koning M, Badawi N. Epidemiology of neonatal encephalopathy and hypoxic-ischemic encephalopathy. Early Hum Dev. 2010;86:329–38.
Douglas-Escobar M, Weiss MD. Hypoxic-ischemic encephalopathy: a review for the clinician. JAMA Pediatr. 2015;169:397–403.
Gunn AJ, Thoresen M. Neonatal encephalopathy and hypoxic-ischemic encephalopathy. Handb Clin Neurol. 2019;162:217–37.
Shankaran S, Pappas A, McDonald SA, Vohr BR, Hintz SR, Yolton K, et al. Childhood outcomes after hypothermia for neonatal encephalopathy. NEJM. 2012;366:2085–92.
Rasineni GK, Panigrahy N, Rath SN, Chinnaboina M, Konanki R, Chirla DK, et al. Diagnostic and therapeutic roles of the “Omics” in hypoxic-ischemic encephalopathy in neonates. Bioengineering (Basel, Switzerland). 2022;9:498.
The Task Force onNeonatal Encephalopathy, TheAmerican College of Obstetricians and Gynecologists. Neonatal encephalopathy and neurologic outcome, second edition. Pediatrics. 2014;133:e1482–8.
Gunn AJ, Laptook AR, Robertson NJ, Barks JD, Thoresen M, Wassink G, et al. Therapeutic hypothermia translates from ancient history in to practice. Pediatr Res. 2017;81:202–9.
Nair J, Kumar VHS. Current and emerging therapies in the management of hypoxic ischemic encephalopathy in neonates. Children (Basel, Switzerland). 2018;5:99.
Douglas-Escobar M, Weiss M. Biomarkers of hypoxic-ischemic encephalopathy in newborns. Front Neurol. 2012;3:144.
Caramelo I, Coelho M, Rosado M, Cardoso C, Dinis A, Duarte CB, et al. Biomarkers for the diagnosis of hypoxic-ischemic encephalopathy: a systematic review: PROSPERO 2021 CRD42021272610. 2021. Available: https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=272610. Accessed 8 Nov 2021.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
Wells GA, Shea B, O'Connell D, Peterson J, Welch V, Losos M, Tugwell P. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses: Ottawa Hospital Research Institute. 2021. Available: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 3 Dec 2021.
Heberle H, Meirelles GV, da Silva FR, Telles GP, Minghim R. InteractiVenn: a web-based tool for the analysis of sets through Venn diagrams. BMC Bioinform. 2015;16:169.
da Huang W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009;4:44–57.
Bonnot T, Gillard MB, Nagel DH. A simple protocol for informative visualization of enriched Gene Ontology terms. Bioprotocol. 2019;9: e3429.
Pang Z, Chong J, Zhou G, de Lima Morais DA, Chang L, Barrette M, et al. MetaboAnalyst 5.0: narrowing the gap between raw spectra and functional insights. Nucleic Acids Res. 2021;49:W388–96.
Ennen CS, Huisman TA, Savage WJ, Northington FJ, Jennings JM, Everett AD, et al. Glial fibrillary acidic protein as a biomarker for neonatal hypoxic-ischemic encephalopathy treated with whole-body cooling. Am J Obstet Gynecol. 2011;205(251):e1-7.
Chalak LF, Sánchez PJ, Adams-Huet B, Laptook AR, Heyne RJ, Rosenfeld CR. Biomarkers for Severity of neonatal hypoxic-ischemic encephalopathy and outcomes in newborns receiving hypothermia therapy. J Pediatr. 2014;164:468-74.e1.
Akamatsu T, Sugiyama T, Aoki Y, Kawabata K, Shimizu M, Okazaki K, et al. A pilot study of soluble form of LOX-1 as a novel biomarker for neonatal hypoxic-ischemic encephalopathy. J Pediatr. 2019;206:49-55.e3.
Balada R, Tebe C, Leon M, Arca G, Alsina M, Castells AA, et al. Enquiring beneath the surface: can a gene expression assay shed light into the heterogeneity among newborns with neonatal encephalopathy? Pediatr Res. 2020;88:451–8.
Lopez-Suarez O, Concheiro-Guisan A, Sanchez-Pintos P, Cocho JA, Fernandez Lorenzo JR, Couce ML. Acylcarnitine profile in neonatal hypoxic-ischemic encephalopathy: the value of butyrylcarnitine as a prognostic marker. Medicine (Baltimore). 2019;98:e15221.
Douglas-Escobar M, Yang C, Bennett J, Shuster J, Theriaque D, Leibovici A, et al. A pilot study of novel biomarkers in neonates with hypoxic-ischemic encephalopathy. Pediatr Res. 2010;68:531–6.
Oh W, Perritt R, Shankaran S, Merritts M, Donovan EF, Ehrenkranz RA, et al. Association between urinary lactate to creatinine ratio and neurodevelopmental outcome in term infants with hypoxic-ischemic encephalopathy. J Pediatr. 2008;153:375–8.
Pineiro-Ramos JD, Nunez-Ramiro A, Llorens-Salvador R, Parra-Llorca A, Sanchez-Illana A, Quintas G, et al. Metabolic phenotypes of hypoxic-ischemic encephalopathy with normal vs. pathologic magnetic resonance imaging outcomes. Metabolites. 2020;10:109.
Massaro AN, Jeromin A, Kadom N, Vezina G, Hayes RL, Wang KK, et al. Serum biomarkers of MRI brain injury in neonatal hypoxic ischemic encephalopathy treated with whole-body hypothermia: a pilot study. Pediatr Crit Care Med. 2013;14:310–7.
Mitra S, Bale G, Meek J, Uria-Avellanal C, Robertson NJ, Tachtsidis I. Relationship between cerebral oxygenation and metabolism during rewarming in newborn infants after therapeutic hypothermia following hypoxic-ischemic brain injury. Adv Exp Med Biol. 2016;923:245–51.
Jain SV, Pagano L, Gillam-Krakauer M, Slaughter JC, Pruthi S, Engelhardt B. Cerebral regional oxygen saturation trends in infants with hypoxic-ischemic encephalopathy. Early Hum Dev. 2017;113:55–61.
Massaro AN, Chang T, Kadom N, Tsuchida T, Scafidi J, Glass P, et al. Biomarkers of brain injury in neonatal encephalopathy treated with hypothermia. J Pediatr. 2012;161:434–40.
Maggiotto LV, Sondhi M, Shin BC, Garg M, Devaskar SU. Circulating blood cellular glucose transporters - surrogate biomarkers for neonatal hypoxic-ischemic encephalopathy assessed by novel scoring systems. Mol Genet Metab. 2019;127:166–73.
Dehaes M, Aggarwal A, Lin PY, Rosa Fortuno C, Fenoglio A, Roche-Labarbe N, et al. Cerebral oxygen metabolism in neonatal hypoxic ischemic encephalopathy during and after therapeutic hypothermia. J Cereb Blood Flow Metab. 2014;34:87–94.
Ponnusamy V, Kapellou O, Yip E, Evanson J, Wong LF, Michael-Titus A, et al. A study of microRNAs from dried blood spots in newborns after perinatal asphyxia: a simple and feasible biosampling method. Pediatr Res. 2016;79:799–805.
Massaro AN, Wu YW, Bammler TK, MacDonald JW, Mathur A, Chang T, et al. Dried blood spot compared to plasma measurements of blood-based biomarkers of brain injury in neonatal encephalopathy. Pediatr Res. 2019;85:655–61.
Saito J, Shibasaki J, Shimokaze T, Kishigami M, Ohyama M, Hoshino R, et al. Temporal relationship between serum levels of interleukin-6 and c-reactive protein in therapeutic hypothermia for neonatal hypoxic-ischemic encephalopathy. Am J Perinatol. 2016;33:1401–6.
Jones R, Heep A, Odd D. Biochemical and clinical predictors of hypoxic-ischemic encephalopathy after perinatal asphyxia. J Matern Fetal Neonatal Med. 2018;31:791–6.
Alshweki A, Perez-Munuzuri A, Lopez-Suarez O, Bana A, Couce ML. Relevance of urinary S100B protein levels as a short-term prognostic biomarker in asphyxiated infants treated with hypothermia. Medicine (Baltimore). 2017;96:e8453.
Massaro AN, Chang T, Baumgart S, McCarter R, Nelson KB, Glass P. Biomarkers S100B and neuron-specific enolase predict outcome in hypothermia-treated encephalopathic newborns*. Pediatr Crit Care Med. 2014;15:615–22.
Fredly S, Nygaard CS, Skranes JH, Stiris T, Fugelseth D. Cooling effect on skin microcirculation in asphyxiated newborn infants with increased c-reactive protein. Neonatology. 2016;110:270–6.
Sweetman DU, Onwuneme C, Watson WR, Murphy JF, Molloy EJ. Perinatal asphyxia and erythropoietin and VEGF: serial serum and cerebrospinal fluid responses. Neonatology. 2017;111:253–9.
Ezgu FS, Atalay Y, Gucuyener K, Tunc S, Koc E, Ergenekon E, et al. Neuron-specific enolase levels and neuroimaging in asphyxiated term newborns. J Child Neurol. 2002;17:824–9.
El-Mazary AA, Abdel-Aziz RA, Mahmoud RA, El-Said MA, Mohammed NR. Correlations between maternal and neonatal serum selenium levels in full term neonates with hypoxic ischemic encephalopathy. Ital J Pediatr. 2015;41:83.
Bale G, Mitra S, Meek J, Robertson N, Tachtsidis I. A new broadband near-infrared spectroscopy system for in vivo measurements of cerebral cytochrome-c-oxidase changes in neonatal brain injury. Biomed Opt Express. 2014;5:3450–66.
Shaikh H, Boudes E, Khoja Z, Shevell M, Wintermark P. Angiogenesis dysregulation in term asphyxiated newborns treated with hypothermia. PLoS ONE. 2015;10: e0128028.
Locci E, Noto A, Puddu M, Pomero G, Demontis R, Dalmazzo C, et al. A longitudinal 1H-NMR metabolomics analysis of urine from newborns with hypoxic-ischemic encephalopathy undergoing hypothermia therapy. Clinical and medical legal insights. PLoS ONE. 2018;13:e0194267.
Bersani I, Ferrari F, Lugli L, Ivani G, Conio A, Moataza B, et al. Monitoring the effectiveness of hypothermia in perinatal asphyxia infants by urinary S100B levels. Clin Chem Lab Med. 2019;57:1017–25.
Haiju Z, Suyuan H, Xiufang F, Lu Y, Sun R. The combined detection of umbilical cord nucleated red blood cells and lactate: early prediction of neonatal hypoxic ischemic encephalopathy. J Perinat Med. 2008;36:240–7.
Chouthai NS, Sobczak H, Khan R, Subramanian D, Raman S, Rao R. Hyperglycemia is associated with poor outcome in newborn infants undergoing therapeutic hypothermia for hypoxic ischemic encephalopathy. J Neonatal Perinatal Med. 2015;8:125–31.
Choudhary M, Sharma D, Dabi D, Lamba M, Pandita A, Shastri S. Hepatic dysfunction in asphyxiated neonates: prospective case-controlled study. Clin Med Insights Pediatr. 2015;9:1–6.
Tu YF, Wu PM, Yu WH, Li CI, Wu CL, Kang L, et al. Lactate predicts neurological outcomes after perinatal asphyxia in post-hypothermia era: a prospective cohort study. Life (Basel). 2021;11:1193.
Amoo M, Henry J, O’Halloran PJ, Brennan P, Husien MB, Campbell M, et al. S100B, GFAP, UCH-L1 and NSE as predictors of abnormalities on CT imaging following mild traumatic brain injury: a systematic review and meta-analysis of diagnostic test accuracy. Neurosurg Rev. 2022;45:1171–93.
Marangos PJ, Schmechel DE. Neuron specific enolase, a clinically useful marker for neurons and neuroendocrine cells. Annu Rev Neurosci. 1987;10:269–95.
Oh SH, Lee JG, Na SJ, Park JH, Choi YC, Kim WJ. Prediction of early clinical severity and extent of neuronal damage in anterior-circulation infarction using the initial serum neuron-specific enolase level. Arch Neurol. 2003;60:37–41.
Isgro MA, Bottoni P, Scatena R. Neuron-specific enolase as a biomarker: biochemical and clinical aspects. Adv Exp Med Biol. 2015;867:125–43.
Donato R, Sorci G, Riuzzi F, Arcuri C, Bianchi R, Brozzi F, et al. S100B’s double life: intracellular regulator and extracellular signal. Biochim Biophys Acta. 2009;1793:1008–22.
Donato R. Intracellular and extracellular roles of S100 proteins. Microsc Res Tech. 2003;60:540–51.
Peeples ES, Dafferner A, Jiang J, Lyden E, Punsoni M, Agrawal DK. Combined treatment with insulin-like growth factor 1 and amd3100 improves motor outcome in a murine model of neonatal hypoxic-ischemic encephalopathy. Dev Neurosci. 2019;41:255–62.
Qiu X, Zhang L, Tong Y, Qu Y, Wang H, Mu D. Interleukin-6 for early diagnosis of neonatal sepsis with premature rupture of the membranes: a meta-analysis. Medicine (Baltimore). 2018;97:e13146.
Amorini AM, Lazzarino G, Di Pietro V, Signoretti S, Lazzarino G, Belli A, et al. Severity of experimental traumatic brain injury modulates changes in concentrations of cerebral free amino acids. J Cell Mol Med. 2017;21:530–42.
Kusaka T, Matsuura S, Fujikawa Y, Okubo K, Kawada K, Namba M, et al. Relationship between cerebral interstitial levels of amino acids and phosphorylation potential during secondary energy failure in hypoxic-ischemic newborn piglets. Pediatr Res. 2004;55:273–9.
Blaise BJ, Schwendimann L, Chhor V, Degos V, Hodson MP, Dallmann G, et al. Persistently altered metabolic phenotype following perinatal excitotoxic brain injury. Dev Neurosci. 2017;39:182–91.
Funding
Open access funding provided by FCT|FCCN (b-on). This work was financed by the European Regional Development Fund (ERDF), through the COMPETE 2020—Operational Programme for Competitiveness and Internationalization and Portuguese national funds via FCT – Fundação para a Ciência e a Tecnologia, under projects POCI-01–0145-FEDER-029311, POCI-01–0247-FEDER-045311, UIDB/04539/2020 and UIDP/04539/2020, and individual Ph.D. fellowships PD/BD/135178/2017 (Margarida Coelho), SFRH/BD/143442/2019 (Inês Caramelo), and 2020.07749.BD (Miguel Rosado).
Author information
Authors and Affiliations
Contributions
Inês Caramelo and Margarida Coelho contributed equally to this paper. Inês Caramelo wrote the manuscript and participated in the database search, articles selection, data extraction, quality assessment, and data analysis. Margarida Coelho participated in the database search, articles selection, quality assessment, data analysis and critically reviewed the article. Miguel Rosado participated in the data extraction of the final articles. Alexandra Dinis established clinically relevant criteria for articles selection and critically reviewed the manuscript. Carla Cardoso critically reviewed the manuscript. Carlos B. Duarte critically reviewed the manuscript. Mário Grãos critically reviewed the manuscript. Bruno Manadas supervised database search, article selection, data extraction and quality assessment, and critically reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose. No financial or non-financial benefits have been received or will be received from any party related directly or indirectly to the subject of this article."
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Caramelo, I., Coelho, M., Rosado, M. et al. Biomarkers of hypoxic–ischemic encephalopathy: a systematic review. World J Pediatr 19, 505–548 (2023). https://doi.org/10.1007/s12519-023-00698-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12519-023-00698-7