Abstract
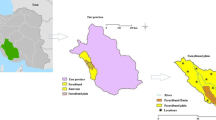
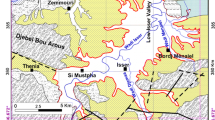
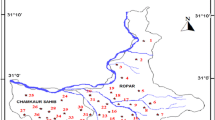
Groundwater in Wadi Fayd, western Saudi Arabia, is the main source of water for domestic and agricultural uses. Groundwater occurs under an unconfined condition in the Quaternary alluvial deposits filling the course of the wadi, where water table lies at shallow depths ranging from 1.08 to 27.40 m. Twenty representative groundwater samples were collected from the wadi. These samples were chemically analyzed for major cations and anions in order to understand the different geochemical processes affecting the groundwater quality. Groundwater has variable total dissolved solid (TDS) values ranging from fresh to saline water. Piper plot of the collected samples indicates that Ca and Mg exceed Na and K in most samples, whereas Cl and SO4 are the dominant anions. HCO3 is the predominant anion in few samples, indicating the effect of dissolution of carbonate minerals in the soil and silicates in the aquifer matrix. Groundwater quality assessment for drinking and irrigation purposes was carried out based on TDS, salinity, and sodium hazards. This indicated that groundwater can be used for drinking in some locations based on the WHO drinking water standard. On the other hand, most groundwater is suitable for irrigation purposes based on sodium and salinity hazards. However, soil type as well as proper selection of plants should be taken into consideration.






Similar content being viewed by others
References
Adams S, Titus R, Pietersen K, Tredoux G, Harris C (2001) Hydrochemical characteristics of aquifer near Sutherland in the Western Karoo, South Africa. J Hydrol 241:91–103
Al-Shaibani AM (2008) Hydrogeology and hydrochemistry of a shallow alluvial aquifer, western Saudi Arabia. Hydrogeol J 16:155–165
American Public Health Association (APHA) (1998) Standard methods for the examination of water and wastewater, 20th edn. APHA-AWWA-WET, Washington
Appelo CAJ, Postma D (2005) Geochemistry, groundwater and pollution, 2nd edn. Balkema, Rotterdam
Back W, Hanshaw BB (1965) Chemical geohydrology. Adv Hydro Sci 1:49–109
Bartarya SK (1993) Hydrochemistry and rock weathering in a subtropical lesser Himalayan river basin in Kumaun, India. J Hydrol 146:149–174
Bhatt KB, Salakani S (1996) Hydrogeochemistry of the upper Ganges River, India. J Geol Soc India 48:171–182
Burdon DJ, Mazloum S (1958) Some chemical types of groundwater from Syria. UNESCO Symposium, Tehran, pp 73–90
Domenico PA (1972) Concepts and models in groundwater hydrology. McGraw-Hill, New York
Domenico PA, Schwartz FW (1998) Physical and chemical hydrogeology, 2nd edn. Wiley, New York, p 506
Freeze RA, Cherry JA (1979) Groundwater. Prentice-Hall, Englewood Cliffs, p 553
Hem JD (1989) Study and interpretation of the chemical characteristics of natural water, 3rd edn. U.S. Geological Survey, Water Supply Paper 2254. US Govt. Printing Office, Washington, 263 pp
Jado AR, ZÖtl JG (1984) Quaternary period in Saudi Arabia, vol 2. Springer, Vienna
Jalali M (2009) Geochemistry characterization of groundwater in an agricultural area of Razan, Hamadan, Iran. Environ Geol 56:1479–1488
Karanth KB (1997) Groundwater assessment, development and management. McGraw-Hill, New Delhi
Kelly WR (1997) Heterogeneities in ground-water geochemistry in a sand aquifer beneath an irrigated field. J Hydrol 198:154–176
Kumar M, Ramanathan AL, Rao MS, Kumar B (2006) Identification and evaluation of hydrogeochemical processes in the groundwater environment of Delhi, India. J Environ Geol 50:1025–1039
Lloyd JW (1965) The hydrochemistry of the aquifer of north eastern Jordan. J Hydrol 3:319–330
Lloyd JW, Heathcote JA (1985) Natural inorganic hydrochemistry in relation to groundwater, an introduction. Clarendon, Oxford
Matthes G (1982) The properties of groundwater. Wiley, New York, 397 pp
Moore TA, Al-Rehalie MH (1989) Geologic map of the Makkah Quadrangle, Sheet 21D. Kingdom of Saudi Arabia, Ministry of Petroleum and Mineral Resources, Directorate General of Mineral Resources
Moral F, Croz-Sanjulia’n JJ, Olias M (2008) Geochemical evolution of groundwater in the carbonate aquifers of Sierra de Segura (Betic Cordillera, Southern Spin). J Hydrol 360:281–296
Ongley ED (1996) Control of water pollution from agriculture. FAO, Rome
Palmer CD, Cherry JA (1984) Geochemical evolution of groundwater in sequences of sedimentary rocks. J Hydrol 75:27–65
Piper AM (1944) A graphic procedure in the geochemical interpretation of water analysis. Am Geophys Union Trans 25:914–928
Plummer LN, Parkhourstm DL, Wigley TML (1979) Critical review of kinetics of calcite dissolution and precipitation. In: Jenne EA (ed) Chemical modeling in aqueous systems, speciation, sorption, solubility and kinetics. American Chemical Society Symposium Ser 93, pp 537–573
US Salinity Laboratory Staff (1954) Diagnosis and improvement of saline and alkali soils. US Department of Agriculture Handbook no. 60, Washington, DC, 160 pp
Schuh WM, Klinekebiel DL, Gardner JC, Meyar RF (1997) Tracer and nitrate movements to groundwater in the Norruem Great Plains. J Environ Qual 26:1335–1347
Siddiqui A, Naseem S, Jalil T (2005) Groundwater quality assessment in and around Kalu Khuhar, Super highway, Sindh, Pakistan. J Appl Sci 5(7):1260–1265
Srinivasamoorthy K, Chidambaram S, Prasanna MV, Vasanthavihar M, Peter J, Anandhan P (2008) Identification of major sources controlling groundwater chemistry from hard rock terrain—a case study from Mettur taluk, Salem district, Tamil Nadu, India. J Earth Syst Sci 117(1):49–58
Todd D (1980) Groundwater hydrology, 2nd edn. Wiley, New York, 535 pp
Wallick EI, Toth J (1976) Methods of regional groundwater flow analysis with suggestions for the use of environmental isotope and hydrochemical data in groundwater hydrology. IAEA, Vienna, pp 37–64
WHO (2004) Guidelines for drinking-water quality, 3rd edn, vol. 1. Recommendation. World Health Organization, Geneva
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Ahmadi, M.E. Groundwater quality assessment in Wadi Fayd, Western Saudi Arabia. Arab J Geosci 6, 247–258 (2013). https://doi.org/10.1007/s12517-011-0337-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12517-011-0337-0