Abstract
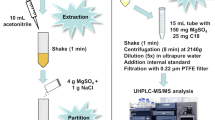
Three sample preparation procedures were compared for a multi-residue analysis of pesticides in hops: (a) modified Hengel’s method based on extraction with acetonitrile in combination with clean-up on a C18 SPE column, (b) miniaturized Biendl’s method based on acetone extraction and PSA SPE sample clean-up, and (c) modified Quick, Easy, Cheap, Effective, Rugged, and Safe (QuEChERS) method which utilizes a specific mixture of three sorbents (PSA, C18, and Z-Sep) for dispersive SPE sample clean-up. The performance of the methods was evaluated and validated for a mixture of 56 pesticides analyzed by the liquid chromatography–high resolution mass spectrometry in compliance with the analytical quality control criteria of the SANTE/11813/2017 guidelines. Strong matrix-dependent signal suppression caused by the co-eluting hop matrix was observed for all sample preparations involved in this method comparison study. The matrix effects in percentages (%ME) were used for the evaluation of the effectiveness of the individual sample clean-up procedures. The recovery experiments were performed by spiking pesticides at the concentration level 0.50 mg/kg into the blank matrix to evaluate the extraction efficiency of the compared methods. Recoveries obtained for the modified Hengel’s method were in the range of 70–120% with RSDs of less than 20% for all studied pesticides. The performances of the methods were tested on the set of 24 samples of hops harvested in the Czech Republic. The method comparison on the determined concentration levels of the pesticide residues clearly showed that the extraction efficiency of the QuEChERS method is significantly less effective for the extraction residues presented in a sample above the level 20 mg/kg. In terms of time consumption, labor, materials, and solvents consumptions, the methods were thoroughly compared, and these demands increase in the following order QuEChERS < modified Hengel’s < miniaturized Biendl’s method.




Similar content being viewed by others
References
Anastassiades M, Lehotay SJ, Stajnbaher D, Schenck FJ (2003) Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J AOAC Int 86(2):412–431
Anastassiades M, Kolberg DI, Eichhorn E, Benkenstein A, Lukačević S, Mack, D, Wildgrube C, Sigalov I, Dörk D, Barth A (2015) Quick method for the analysis of numerous highly polar pesticides in foods of plant origin via LC-MS/MS involving simultaneous extraction with methanol (QuPPe-Method). EU Reference Laboratory for pesticides requiring Single Residue methods (EURL-SRM). http://www.crl-pesticides.eu/library/docs/srm/meth_QuPPe.pdf. Accessed Dec 2018
Belitz HD, Grosch W, Schieberle P (2009) Alcoholic beverages in: food chemistry, 4th edn. Springer Science & Business Media, Berlin, pp 895–896
Biendl M, Jaeger G, Kittsteiner-Eberle R, Schmidt C (2014) Rapid and sensitive determination of pesticide residues in hops and hop products using HPLC-MS/MS and GC-MS/MS. BrSc 67:108–115
Čajka T, Sandy C, Bachanová V, Drábová L, Kalachová K, Pulkrabová J, Hajlšová J (2012) Streamlining sample preparation and gas chromatography–tandem mass spectrometry analysis of multiple pesticide residues in tea. Anal Chim Acta 743:51–60 https://doi.org/10.1016/j.aca.2012.06.051
Dušek M, Jandovská V, Olšovská J (2018a) Analysis of multiresidue pesticides in driedhops by LC–MS/MS using QuEChERS extraction together with dSPE clean-up. J I Brewing 124:222–229 https://doi.org/10.1002/jib.490
Dušek M, Jandovská V, Olšovská J (2018b) Pesticide residue analysis in hops: four different procedures of sample preparation are currently used. Is one of them more useful than the others? 55th North American Chemical Residue Workshop [Online], Naples, FL, USA, July 22–25, 2018. Research Institute of Brewing and Malting. https://beerresearch.cz/wp-content/uploads/2019/02/NACRW2018_Poster59_Dusek.pdf. Accessed Feb 2019
EURL-SRM (2016) Analytical observations report analysis of pymetrozine by the QuEChERS method - impact of pH on recovery rate. EU Reference Laboratory for pesticides requiring Single Residue methods (EURL-SRM). http://www.crl-pesticides.eu/userfiles/file/EurlSRM/EurlSrm_Observations_Pymetrozine.pdf. Accessed Dec 2018
Harris D (1998) Quantitative chemical analysis. In: Katz E, Eksteen R, Schoenmakers P, Miller N (eds) Handbook of HPLC, 9th edn. Marcel Dekker Inc., New York
Hengel MJ, Miller M (2008) Analysis of pesticides in dried hops by liquid chromatography-tandem mass spectrometry. J Agric Food Chem 56(16):6851–6856
Hengel MJ (2011) Expanded method development for the determination of pesticides in dried hops by liquid chromatography with tandem mass spectrometry. J Am Soc Brew Chem 69(3):121–126. https://doi.org/10.1094/ASBCJ-2011-0519-01
Kolberg DI, Prestes OD, Adaime MB, Zanella R (2011) Development of a fast multiresidue method for the determination of pesticides in dry samples (wheat grains, flour and bran) using QuEChERS based method and GC-MS. Food Chem 125(4):1436–1442. https://doi.org/10.1016/j.foodchem.2010.10.041
PPDB: Pesticide Properties DataBase (2007) Agriculture & Environment Research Unit (AERU) at the University of Hertfordshire. https://sitem.herts.ac.uk/aeru/ppdb/en/index.htm. Accessed Dec 2018
SANTE/11813 (2017) Guidance document on analytical quality control and method validation procedures for pesticide residues and analysis in food and feed (implemented 01/01/2018. European Commission, Health and Consumer Protection Directorate General. https://ec.europa.eu/food/sites/food/files/plant/docs/pesticides_mrl_guidelines_wrkdoc_2017-11813.pdf. Accessed Dec 2018
Schenck FJ, Lehotay SJ (2000) Does further clean-up reduce the matrix enhancement effect in gas chromatographic analysis of pesticide residues in food? J. Chromatogr A 868(1):51–61
Schenck FJ, Lehotay SJ, Vega V (2002) Comparison of solid-phase extraction sorbents for cleanup in pesticide residue analysis of fresh fruits and vegetables. J Sep Sci 25(14):883–890
Stone DC, Ellis J (2011) Stats tutorial - instrumental analysis and calibration. Department of Chemistry, University of Toronto. http://www.chem.utoronto.ca/coursenotes/analsci/stats/index.html. Accessed Dec 2018
Zhou W, Yang S, Wang PB (2017) Matrix effects and application of matrix effect factor. Bioanalysis 9(23):1839–1844. https://doi.org/10.4155/bio-2017-0214
Funding
The study was supported by the Ministry of Agriculture of the Czech Republic within Institutional Support MZE-RO1918 and by the Charles University within project SVV260440.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Martin Dušek declares that he has no conflict of interest. Vladimíra Jandovská declares that she has no conflict of interest. Kamila Kalachová declares that she has no conflict of interest. Jana Olšovská declares that she has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dušek, M., Jandovská, V., Kalachová, K. et al. Comparative Study of Three Sample Preparation Methods for Multi-residue Extraction of Pesticide Residues in Hop Samples. Food Anal. Methods 13, 503–515 (2020). https://doi.org/10.1007/s12161-019-01658-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-019-01658-6