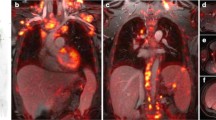
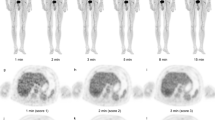
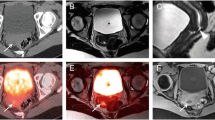
Abstract
Purpose
To provide clinically useful gadolinium-free whole-body cancer staging of children and young adults with integrated positron emission tomography/magnetic resonance (PET/MR) imaging in less than 1 h.
Procedures
In this prospective clinical trial, 20 children and young adults (11–30 years old, 6 male, 14 female) with solid tumors underwent 2-deoxy-2-[18F]fluoro-d-glucose ([18F]FDG) PET/MR on a 3T PET/MR scanner after intravenous injection of ferumoxytol (5 mg Fe/kg) and [18F]FDG (2–3 MBq/kg). Time needed for patient preparation, PET/MR image acquisition, and data processing was compared before (n = 5) and after (n = 15) time-saving interventions, using a Wilcoxon test. The ferumoxytol-enhanced PET/MR images were compared with clinical standard staging tests regarding radiation exposure and tumor staging results, using Fisher’s exact tests.
Results
Tailored workflows significantly reduced scan times from 36 to 24 min for head to mid thigh scans (p < 0.001). These streamlined PET/MR scans were obtained with significantly reduced radiation exposure (mean 3.4 mSv) compared to PET/CT with diagnostic CT (mean 13.1 mSv; p = 0.003). Using the iron supplement ferumoxytol “off label” as an MR contrast agent avoided gadolinium chelate administration. The ferumoxytol-enhanced PET/MR scans provided equal or superior tumor staging results compared to clinical standard tests in 17 out of 20 patients. Compared to PET/CT, PET/MR had comparable detection rates for pulmonary nodules with diameters of equal or greater than 5 mm (94 vs. 100 %), yet detected significantly fewer nodules with diameters of less than 5 mm (20 vs 100 %) (p = 0.03). [18F]FDG-avid nodules were detected with slightly higher sensitivity on the PET of the PET/MR compared to the PET of the PET/CT (59 vs 49 %).
Conclusion
Our streamlined ferumoxytol-enhanced PET/MR protocol provided cancer staging of children and young adults in less than 1 h with equivalent or superior clinical information compared to clinical standard staging tests. The detection of small pulmonary nodules with PET/MR needs to be improved.






Similar content being viewed by others
References
Federman N, Feig SA (2007) PET/CT in evaluating pediatric malignancies: a clinician’s perspective. J Nucl Med 48:1920–1922
Kleis M, Daldrup-Link H, Matthay K et al (2009) Diagnostic value of PET/CT for the staging and restaging of pediatric tumors. Eur J Nucl Med Mol Imaging 36:23–36
Tatsumi M, Miller JH, Wahl RL (2007) 18F-FDG PET/CT in evaluating non-CNS pediatric malignancies. J Nucl Med 48:1923–1931
London K, Cross S, Onikul E et al (2011) 18F-FDG PET/CT in paediatric lymphoma: comparison with conventional imaging. Eur J Nucl Med Mol Imaging 38:274–284
Cheng G, Servaes S, Zhuang H (2013) Value of 18F-fluoro-2-deoxy-D-glucose positron emission tomography/computed tomography scan versus diagnostic contrast computed tomography in initial staging of pediatric patients with lymphoma. Leukemia Lymphoma 54:737–742
London K, Stege C, Cross S et al (2012) 18F-FDG PET/CT compared to conventional imaging modalities in pediatric primary bone tumors. Pediatr Radiol 42:418–430
Walter F, Czernin J, Hall T et al (2012) Is there a need for dedicated bone imaging in addition to 18F-FDG PET/CT imaging in pediatric sarcoma patients? J Pediatr Hematol Oncol 34:131–136
Ponisio MR, McConathy J, Laforest R, Khanna G (2016) Evaluation of diagnostic performance of whole-body simultaneous PET/MRI in pediatric lymphoma. Pediatr Radiol 46:1258–1268
Miglioretti DL, Johnson E, Williams A et al (2013) The use of computed tomography in pediatrics and the associated radiation exposure and estimated cancer risk. J Am Med Assoc Pediatr 167:700–707
Weiser DA, Kaste SC, Siegel MJ, Adamson PC (2013) Imaging in childhood cancer: a Society for Pediatric Radiology and Children's oncology group joint task force report. Pediatr Blood Cancer 60:1253–1260
Grueneisen J, Nagarajah J, Buchbender C et al (2015) Positron emission tomography/magnetic resonance imaging for local tumor staging in patients with primary breast cancer: a comparison with positron emission tomography/computed tomography and magnetic resonance imaging. Investig Radiol 50:505–513
Souvatzoglou M, Eiber M, Takei T et al (2013) Comparison of integrated whole-body [11C]choline PET/MR with PET/CT in patients with prostate cancer. Eur J Nucl Med Mol Imaging 40:1486–1499
Uslu L, Donig J, Link M et al (2015) Value of 18F-FDG PET and PET/CT for evaluation of pediatric malignancies. J Nucl Med 56:274–286
Hirsch FW, Sattler B, Sorge I et al (2013) PET/MR in children. Initial clinical experience in paediatric oncology using an integrated PET/MR scanner. Pediatr Radiol 43:860–875
Huellner MW, Appenzeller P, Kuhn FP et al (2014) Whole-body nonenhanced PET/MR versus PET/CT in the staging and restaging of cancers: preliminary observations. Radiology 273:859–869
McDonald RJ, McDonald JS, Kallmes DF et al (2015) Intracranial gadolinium deposition after contrast-enhanced MR imaging. Radiology 275:772–782
Jiang W, Tao X, Fang F, Zhang S, Xu C (2013) Benign and malignant ovarian steroid cell tumors, not otherwise specified: case studies, comparison, and review of the literature. J Ovarian Res 6:53
Schafer JF, Gatidis S, Schmidt H et al (2014) Simultaneous whole-body PET/MR imaging in comparison to PET/CT in pediatric oncology: initial results. Radiology 273:220–231
Gatidis S, Schmidt H, Gucke B et al (2016) Comprehensive oncologic imaging in infants and preschool children with substantially reduced radiation exposure using combined simultaneous (1)(8)F-fluorodeoxyglucose positron emission tomography/magnetic resonance imaging: a direct comparison to (1)(8)F-fluorodeoxyglucose positron emission tomography/computed tomography. Investig Radiol 51:7–14
Sher AC, Seghers V, Paldino MJ et al (2016) Assessment of sequential PET/MRI in comparison with PET/CT of pediatric lymphoma: a prospective study. Am J Roentgenol 206:623–631
Ricard F, Cimarelli S, Deshayes E et al (2011) Additional benefit of F-18 FDG PET/CT in the staging and follow-up of pediatric rhabdomyosarcoma. Clin Nucl Med 36:672–677
Kneisl JS, Patt JC, Johnson JC, Zuger JH (2006) Is PET useful in detecting occult nonpulmonary metastases in pediatric bone sarcomas? Clin Orthop Relat Res 450:101–104
Klenk C, Gawande R, Uslu L et al (2014) Ionising radiation-free whole-body MRI versus 18F-fluorodeoxyglucose PET/CT scans for children and young adults with cancer: a prospective, non-randomised, single-centre study. Lancet Oncol 15:275–285
Mattsson S, Johansson L, Leide Svegborn S et al (2015) Radiation dose to patients from radiopharmaceuticals: a compendium of current information related to frequently used substances. Ann ICRP 44:7–321
Deak PD, Smal Y, Kalender WA (2010) Multisection CT protocols: sex- and age-specific conversion factors used to determine effective dose from dose-length product. Radiology 257:158–166
Cipriano C, Brockman L, Romancik J et al (2015) The clinical significance of initial pulmonary micronodules in young sarcoma patients. J Pediatr Hematol Oncol 37:548–553
Ciet P, Tiddens HA, Wielopolski PA et al (2015) Magnetic resonance imaging in children: common problems and possible solutions for lung and airways imaging. Pediatr Radiol 45:1901–1915
von Schulthess GK, Veit-Haibach P (2014) Workflow considerations in PET/MR imaging. J Nucl Med 55:19S–24S
Martinez-Moller A, Eiber M, Nekolla SG et al (2012) Workflow and scan protocol considerations for integrated whole-body PET/MRI in oncology. J Nucl Med 53:1415–1426
Vanderby SA, Babyn PS, Carter MW, Jewell SM, McKeever PD (2010) Effect of anesthesia and sedation on pediatric MR imaging patient flow. Radiology 256:229–237
Aghighi M, Pisani LJ, Sun Z et al (2016) Speeding up PET/MR for cancer staging of children and young adults. Eur Radiol 26:4239–4248
Eiber M, Martinez-Moller A, Souvatzoglou M et al (2011) Value of a Dixon-based MR/PET attenuation correction sequence for the localization and evaluation of PET-positive lesions. Eur J Nucl Med Mol Imaging 38:1691–1701
Klenk C, Gawande R, Tran VT et al (2016) Progressing toward a cohesive pediatric 18F-FDG PET/MR protocol: is Administration of Gadolinium Chelates Necessary? J Nucl Med 57:70–77
Daldrup-Link HE, Rummeny EJ, Ihssen B et al (2002) Iron-oxide-enhanced MR imaging of bone marrow in patients with non-Hodgkin’s lymphoma: differentiation between tumor infiltration and hypercellular bone marrow. Eur Radiol 12:1557–1566
Li YW, Chen ZG, Wang JC, Zhang ZM (2015) Superparamagnetic iron oxide-enhanced magnetic resonance imaging for focal hepatic lesions: systematic review and meta-analysis. World J Gastroenterol 21:4334–4344
Ferrucci JT, Stark DD (1990) Iron oxide-enhanced MR imaging of the liver and spleen: review of the first 5 years. AJR Am J Roentgenol 155:943–950
Perazella MA (2009) Current status of gadolinium toxicity in patients with kidney disease. Clin J Am Soc Nephrol 4:461–469
Varallyay CG, Nesbit E, Fu R et al (2013) High-resolution steady-state cerebral blood volume maps in patients with central nervous system neoplasms using ferumoxytol, a superparamagnetic iron oxide nanoparticle. J Cereb Blood Flow Metab 33:780–786
Lu M, Cohen MH, Rieves D, Pazdur R (2010) FDA report: ferumoxytol for intravenous iron therapy in adult patients with chronic kidney disease. Am J Hematol 85:315–319
Muehe AM, Feng D, von Eyben R et al (2016) Safety report of Ferumoxytol for magnetic resonance imaging in children and young adults. Investig Radiol 51:221–227
Fakhran S, Alhilali L, Kale H, Kanal E (2015) Assessment of rates of acute adverse reactions to gadobenate dimeglumine: review of more than 130,000 administrations in 7.5 years. Am J Roentgenol 204:703–706
Pearce MS, Salotti JA, Little MP et al (2012) Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: a retrospective cohort study. Lancet 380:499–505
Brenner DJ, Doll R, Goodhead DT et al (2003) Cancer risks attributable to low doses of ionizing radiation: assessing what we really know. Proc Nat Acad Scie (USA) 100:13761–13766
Minamimoto R, Levin C, Jamali M et al (2016) Improvements in PET image quality in time of flight (TOF) simultaneous PET/MRI. Mol Imaging Biol 18:776–781
Grant AM, Deller TW, Khalighi MM, Maramraju SH, Delso G, Levin CS (2016) NEMA NU 2-2012 performance studies for the SiPM-based ToF-PET component of the GE SIGNA PET/MR system. Med Phys 43:2334
Ward E, DeSantis C, Robbins A, Kohler B, Jemal A (2014) Childhood and adolescent cancer statistics, 2014. CA Cancer J Clin 64:83–103
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66:7–30
Borra RJ, Cho HS, Bowen SL et al (2015) Effects of ferumoxytol on quantitative PET measurements in simultaneous PET/MR whole-body imaging: a pilot study in a baboon model. Eur J Nucl Med Mol Imaging Phys 2:6. doi:10.1186/s40658-015-0109-0
Meignan M, Gallamini A, Meignan M et al (2009) Report on the first international workshop on interim-PET-scan in lymphoma. Leuk Lymph 50:1257–1260
Acknowledgements
This work was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, grant number R01 HD081123-01A1. We thank Praveen Gulaka, Dawn Holley, and Harsh Gandhi from the PET/MR Metabolic Service Centre for their assistance with the acquisition of PET/MR scans. We thank the members of Daldrup-Link lab for valuable input and discussions regarding this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 664 kb)
Rights and permissions
About this article
Cite this article
Muehe, A.M., Theruvath, A.J., Lai, L. et al. How to Provide Gadolinium-Free PET/MR Cancer Staging of Children and Young Adults in Less than 1 h: the Stanford Approach. Mol Imaging Biol 20, 324–335 (2018). https://doi.org/10.1007/s11307-017-1105-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-017-1105-7