Abstract
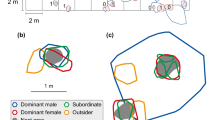
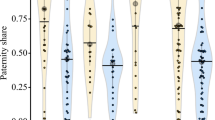
Cooperative breeding has been studied intensively in many species of birds and mammals but remain less well studied in fish. We report a remarkable new example of a cooperatively breeding cichlid from Lake Tanganyika, Neolamprologus obscurus. Using field observations and microsatellite DNA analyses, we studied group structure, helping behavior, relatedness, and dispersal of this species. We present four major observations. First, large territorial breeding males mated with one to eight breeding females, each of which was territorial and unrelated to another. Second, one to ten smaller fish (“subordinates”) of both sexes were allowed to stay inside the breeding females’ territories. Subordinates were often highly related to both the respective breeding male and female and performed territory defense and shelter maintenance, which is regarded as helping behaviors. Third, one to three subordinate males, similar in size to breeding females, were allowed to stay inside a breeding male’s territory but were not tolerated in the breeding females’ territories. Pairwise relatedness suggests these individuals are usually sons of the respective breeding male. Fourth, pairwise relatedness estimates suggest that juveniles delay dispersal and assist their mothers in raising offspring. As female subordinates grow up, they leave the father’s territory and disperse into other groups. In contrast, male subordinates leave their mother’s territory but remain within the territory of their father. The described social system makes N. obscurus a promising new model species to study the evolution of cooperative breeding.



Similar content being viewed by others
References
Awata S, Kohda M (2004) Parental roles and the amount of care in a bi-parental substrate brooding cichlid: the effect of size differences within pairs. Behaviour 141:1135–1149
Awata S, Munehara H, Kohda M (2005) Social system and reproduction of helpers in the cooperatively breeding cichlid fish Julidochromis ornatus in Lake Tanganyika: field observations and parentage analyses. Behav Ecol Sociobiol 58:506–516
Awata S, Heg D, Kohda M, Munehara H, Kohda M (2006) Testis size depends on social status and the presence of male helpers in the cooperatively breeding cichlid Julidochromis ornatus. Behav Ecol 17:372–379
Balshine-Earn S, Neat F, Reid H, Taborsky M (1998) Paying to stay or paying to breed? Field evidence for direct benefits of helping behavior in a cooperatively breeding fish. Behav Ecol 9:432–438
Bates D, Maechler M, Bolker B (2011) lme4: linear mixed-effects models using S4 classes. R package version 0.999375-39. http://CRAN.R-project.org/package=lme4
Bergmüller R, Heg D, Taborsky M (2005) Helpers in a cooperatively breeding cichlid stay and pay or disperse and breed, depending on ecological constraints. Proc R Soc Lond B 272:325–331
Bolker BM, Brooks ME, Clark CI, Geange SW, Poulsen JR, Stevens MH, White JS (2009) Generalized linear mixed models: a practical guide for ecology and evolution. Trend Ecol Evol 24:127–135
Brouwer L, Heg D, Taborsky M (2005) Experimental evidence for helper effects in a cooperatively breeding cichlid. Behav Ecol 16:667–673
Clarke A, Saether B, Roskaft E (1997) Sex biases in avian dispersal: a reappraisal. Oikos 79:429–438
Clutton-Brock TH, Lukas D (2012) The evolution of social philopatry and dispersal in female mammals. Mol Ecol 21:472–492
Cockburn A (2006) Prevalence of different modes of parental care in birds. Proc R Soc Lond B 273:1375–1383
Creel SR (1994) Inclusive fitness and reproductive strategies in dwarf mongooses. Behav Ecol 5:339–348
Desjardins JK, Fitzpatrick JL, Stiver KA, van der Kraak GJ, Balshine S (2008) Costs and benefits of polygyny in the cichlid Neolamprologus pulcher. Anim Behav 75:1771–1779
Dierkes P, Heg D, Taborsky M, Skubic E, Achmann R (2005) Genetic relatedness in groups is sex-specific and declines with age of helpers in a cooperatively breeding cichlid. Ecol Lett 8:968–975
Duftner N, Sefc K, Koblmüller S, Salzburger W, Taborsky M, Sturmbauer C (2007) Parallel evolution of facial stripe patterns in the Neolamprologus brichardi/pulcher species complex endemic to Lake Tanganyika. Mol Phylogenet Evol 45:706–715
Eikenaar C, Richardson DS, Brouwer L, Komdeur J (2007) Parent presence, delayed dispersal, and territory acquisition in the Seychelles warbler. Behav Ecol 18:874–879
Eikenaar C, Richardson DS, Brouwer L, Komdeur J (2008) Sex biased natal dispersal in a closed, saturated population of Seychelles warblers Acrocephalus sechellensis. J Avian Biol 39:73–80
Ekman J, Bylin A, Tegelström H (1999) Parental nepotism enhances survival of retained offspring in the Siberian jay. Behav Ecol 11:416–420
Feeney W, Medina I, Somveille M, Heinsohn R, Hall ML, Mulder RA, Stain JA, Kilner RM, Langmore NE (2013) Brood parasitism and the evolution of cooperative breeding in birds. Science 342:1506–1508
Fitzpatrick JL, Desjardins JK, Stiver KA, Montgomerie R, Balshine S (2005) Male reproductive suppression in the cooperatively breeding fish Neolamprologus pulcher. Behav Ecol 17:25–33
Fitzpatrick JL, Desjardins JK, Milligan N, Stiver KA, Montgomerie R, Balshine S (2008) Female-mediated causes and consequences of social status change in a social fish. Proc R Soc Lond B 275:929–936
Goodnight KF, Queller DC (1999) Computer software for performing likelihood tests of pedigree relationship using genetic markers. Mol Ecol 8:1231–1234
Grantner A, Taborsky M (1998) The metabolic rates associated with resting, and with the performance of agonistic, submissive and digging behaviours in the cichlid fish Neolamprologus pulcher (Pisces: Cichlidae). J Comp Physiol B 168:427–433
Greenwood PJ (1980) Mating systems, philopatry and dispersal in birds and mammals. Anim Behav 28:1140–1162
Greenwood PJ (1983) Mating systems and the evolutionary consequences of dispersal. In: Swingland IR, Greenwood PJ (eds) The ecology of animal movement. Oxford University Press, Oxford, pp 116–131
Greenwood PJ, Harvey PH (1982) The natal and breeding dispersal of birds. Annu Rev Ecol Syst 13:1–21
Griffin AS, West SA (2003) Kin discrimination and the benefit of helping in cooperatively breeding vertebrates. Science 302:634–636
Heg D, Bachar Z (2006) Cooperative breeding in the Lake Tanganyika cichlid Julidochromis ornatus. Environ Biol Fish 76:265–281
Heg D, Bachar Z, Brouwer L, Taborsky M (2004) Predation risk is an ecological constraint for helper dispersal in a cooperatively breeding cichlid. Proc R Soc Lond B 271:2367–2374
Heg D, Bachar Z, Taborsky M (2005) Cooperative breeding and group structure in the Lake Tanganyika cichlids Neolamprologus savory. Ethology 111:1017–1043
Heg D, Rothenberger S, Schürch R (2011) Habitat saturation, benefits of philopatry, relatedness, and the extent of co-operative breeding in a cichlid. Behav Ecol 22:82–92
Koenig WD, Dickinson JL (2004) Ecology and evolution of cooperative breeding in birds. Cambridge University Press, Cambridge
Kohler U (1998) Zur Struktur und Evolution des Sozialsystems von Neolamprologus multifasciatus (Cichlidae, Pisces), dem kleinsten Schneckenbuntbarsch des Tanganjikasees. Shaker Verlag, Aachen
Kokko H, Johnstone RA, Clutton-Brock TH (2001) The evolution of cooperative breeding through group augmentation. Proc R Soc Lond B 268:187–196
Komdeur J, Edelaar P (2001) Male Seychelles warblers use territory budding to maximize lifetime fitness in a saturated environment. Behav Ecol 12:706–715
Konings A (1998) Tanganyika cichlids in their natural habitat. Cichlid Press, El Paso
Konovalov DA, Heg D (2008) TECHNICAL ADVANCES: a maximum-likelihood relatedness estimator allowing for negative relatedness values. Mol Ecol Resour 8:256–263
Konovalov DA, Manning C, Henshaw MT (2004) KINGROUP: a program for pedigree relationship reconstruction and kin group assignments using genetic markers. Mol Ecol Notes 4:779–782
Lukas D, Clutton-Brock T (2012) Cooperative breeding and monogamy in mammalian societies. Proc R Soc Lond B 279:2151–2156
Lukas D, Clutton-Brock TH (2013) The evolution of social monogamy in mammals. Science 341:526–530
Munehara H, Awata S, Katoh R, Kohda M, Sunobe T (2001) Primer sequences and cross-species amplification for parentage discrimination of Tanganyikan cichlid fishes. Bull Fac Fish, Hokkaido Univ 52:131–133
Parker A, Kornfield I (1996) Polygynandry in Pseudotropheus zebra, a cichlid fish from Lake Malawi. Environ Biol Fish 47:345–352
Poll M (1978) A new cichlid fish genus-Lamprologus Schth. Description of four new species from Lake Tanganyika [in Central Africa]. B Cl Sci Ac Roy Belg 64:725–758
Pusey AE (1987) Sex-biased dispersal and inbreeding avoidance in birds and mammals. Trend Ecol Evol 2:295–299
Pusey AE, Wolf M (1996) Inbreeding avoidance in animals. Trend Ecol Evol 11:201–206
R development core team (2011) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rossiter A (1993) Studies on the biology of Neolamprologus multifasciatus. In: Nagoshi M, Yanagisawa Y, Kawanabe H (eds) Ecological and limnological study on Lake Tanganyika and its adjacent regions VIII. Kyoto University Press, Kyoto, pp 32
Sato T, Gashagaza MM (1997) Shell-brooding cichlid fishes of Lake Tanganyika: their habitats and mating systems. In: Kawanabe H, Hori M, Nagoshi M (eds) Fish communities in Lake Tanganyika. Kyoto University Press, Kyoto, pp 221–240
Schliewen U, Rassmann K, Markmann M, Markert J, Kocker T, Tautz D (2001) Genetic and ecological divergence of a monophyletic cichlid species pair under fully sympatric conditions in the Lake Ejagham, Cameroon. Mol Ecol 10:1471–1488
Schradin C, Lamprecht J (2000) Female-biased immigration and male peace-keeping in groups of the shelldwelling cichlid fish Neolamprologus multifasciatus. Behav Ecol Sociobiol 48:236–242
Schradin C, Lamprecht J (2002) Causes of female emigration in the group-living cichlid fish Neolamprologus multifasciatus. Ethology 108:237–248
Stiver KA, Dierkes P, Taborsky M, Balshine S (2004) Dispersal patterns and status change in a co-operatively breeding cichlids Neolamprologus pulcher: evidence from microsatellite analyses and behavioural observations. J Fish Biol 65:91–105
Stiver KA, Dierkes P, Taborsky M, Gibbs HL, Balshine S (2005) Relatedness and helping in fish: examining the theoretical predictions. Proc R Soc Lond B 272:1593–1599
Stiver KA, Desjardins JK, Fitzpatrick JL, Neff B, Quinn JS, Balshine S (2007) Evidence for size and sex-specific dispersal in a cooperatively breeding cichlid fish. Mol Ecol 16:2974–2984
Sturmbauer C, Salzburger W, Duftner N, Schelly R, Koblmuller S (2010) Evolutionary history of the Lake Tanganyika cichlid tribe Lamprologini (Teleostei: Perciformes) derived from mitochondrial and nuclear DNA data. Mol Phylogenet Evol 57:266–284
Sunobe T, Munehara H (2003) Mating system and kin relationship between adults and young in the shell-brooding cichlid fish Neolamprologus meeli in Lake Tanganyika. J Ethol 21:87–92
Taborsky M (1984) Broodcare helpers in the cichlid fish Lamprologus brichardi: their costs and benefits. Anim Behav 32:1236–1252
Taborsky M (1985) Breeder-helper conflict in a cichlid fish with broodcare helpers: an experimental analysis. Behaviour 95:45–75
Taborsky M (1994) Sneakers, satellites, and helpers: parasitic and cooperative behavior in fish reproduction. Adv Stud Behav 23:1–100
Taborsky M (2001) The evolution of parasitic and cooperative reproductive behaviors in fishes. J Hered 92:100–110
Taborsky M, Limberger D (1981) Helpers in fish. Behav Ecol Sociobiol 8:143–145
Tommkins JL, Simmons LW (2002) Measuring relative investment: a case study of testes investment in species with alternative male reproductive tactics. Anim Behav 63:1009–1016
van Oppen MJH, Rico C, Deutsch JC, Turner GF, Hewitt GM (1997) Isolation and characterization of microsatellite loci in the cichlid fish Pseudotropheus zebra. Mol Ecol 6:387–388
West SA, Pen I, Griffin AS (2002) Cooperation and competition between relatives. Science 296:72–75
Wisenden BD (1999) Alloparental care in fishes. Rev Fish Biol Fish 9:45–70
Wong M, Balshine S (2010) The evolution of cooperative breeding in the African cichlid fish, Neolamprologus pulcher. Biol Rev 86:511–530
Woolfenden GE, Fitzpatrick JW (1990) Florida Scrub Jays: a synopsis after 18 years of study. In: Stacey PB, Koenig WD (eds) Cooperative breeding in birds: long-term studies of ecology and behavior. Cambridge University Press, New York, pp 241–266
Yamagishi S, Kohda M (1996) Is the cichlid fish Julidochromis marlieri polyandrous? Ichthyol Res 43:469–471
Zardoya R, Vollmer DM, Craddock C, Streelman JT, Karl S, Meyer A (1996) Evolutionary conservation of microsatellite flanking regions and their use in resolving the phylogeny of cichlid fishes (Pisces: Perciformis). Proc R Soc Lond B 263:1589–1598
Acknowledgments
We thank Tetsumi Takahashi, Kazutaka Ota, Haruki Ochi, Hiroki Hata, Michio Hori, and the staff of the Lake Tanganyika Research Unit, Mpulungu, Zambia, especially Harris Phiri, Danny Syniynza, Ruben Shapola, and Henry Simpembwa for supporting our studies at the field. We are grateful to Satoshi Nanami and Sho Furuichi for statistical discussions and Joachim Frommen and three anonymous referees for helpful comments on early versions of this manuscript. This work was financially supported by Grants-in-Aid from the Ministry of Education, Culture, Sports, Science and Technology, Japan, to MK.
Ethical standards
The research presented here was conducted with permission from the Zambian Ministry of Agriculture, Food and Fisheries and complies with current Zambian law. We treated fish in compliance with the guidelines of the Animal Care and Use Committee of Osaka City University, the Japan Ethological Society, and the Ichthyological Society of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. Frommen
Rights and permissions
About this article
Cite this article
Tanaka, H., Heg, D., Takeshima, H. et al. Group composition, relatedness, and dispersal in the cooperatively breeding cichlid Neolamprologus obscurus . Behav Ecol Sociobiol 69, 169–181 (2015). https://doi.org/10.1007/s00265-014-1830-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-014-1830-8