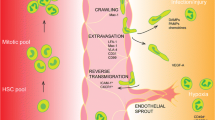
Abstract
Neurophils are a key component of the innate immune response and a major contributor to inflammation. Accordingly, their number in the blood is tightly controlled. Neutrophils are normally produced exclusively in the bone marrow, and their release into the blood is tightly regulated to maitain homeostatic levels in the blood. Herein, recent evidence is reviewed showing that stromal cell-derived factor-1 (SDF-1, CXCL12) plays a key role in regulating neutrophil trafficking in the bone marrow. Moreover, studies are discussed showing that granulocyte colony-stimulating factor, primarily through regulation of SDF-1 expression in the bone marrow, plays an essential role in maitaining neutrophil homeostasis in the blood.
Similar content being viewed by others
References
Yoder MC, Williams DA: Matrix molecule interactions with hematopoietic stem cells. Exp Hematol 1995;23: 961–967.
Campbell F: Ultrastructural studies of transmural migration of blood cells in the bone marrow of rats, mice and guinea pigs. Am J Anat 1972;135:521–536.
Inoue S, Osmond DG: Basement membrane of mouse bone marrow sinusoids shows distinctive structure and proteoglycan composition: a high resolution ultrastructural study. Anat Rec 2001;264:294–304.
Weiss L: Transmural cellular passage in vascular sinuses pf rat bone marrow. Blood 1970;36:189–208.
Fliedner T, Cronkite E, Killmann S, Bond V: Granulocytopoiesis II. Emergence and pattern of labeling of neutrophilic granulocytes in humans. Blood 1964;6:683–700.
Sémerad CL, Liu F, Gregory AD, Stumpf K, Link DC: G-CSF is an essential regulator of neutrophil trafficking from the bone marrow to the blood. Immunity 2002; 17:413–423.
Opdenakker G, Fibbe WE, Van Damme J: The molecular basis of leukocytosis. Immunol Today 1998;19: 182–189.
Anderlini P, Przepiorka D, Champlin R, Korbling M: Biologic and clinical effects of granulocyte colony-stimulating factor in normal individuals. Blood 1996;88:2819–2825.
Ulich TR, del Castillo J, Souza L: Kinetics and mechanisms of recombinant human granulocyte-colony stimulating factor-induced neutrophilia [erratum appears in Am J Pathol 1989;134(2):236] Am J Pathol 1988;133: 630–638.
Cohen AM, Zsebo KM, Inoue H, et al: In vivo stimulation of granulopoiesis by recombinant human granulocyte colony-stimulating factor. Proc Natl Acad Sci USA 1987;84:2484–2488.
Lieschke GJ, Grail D, Hodgson G, et al: Mice lacking granulocyte colony-stimulating factor have chronic neutropenia, granulocyte and macrophage progenitor cell deficiency, and impaired neutrophil mobilization. Blood 1994;84:1737–1746.
Liu F, Wu HY, Wesselschmidt R, Kornaga T, Link DC: Impaired production and increased apoptosis of neutrophils in granulocyte colony-stimulating factor receptor-deficient mice. Immunity 1996;5:491–501.
Richards MK, Liu F, Iwasaki H, Akashi H, Link DC: Pivotal role of granulocyte colony-stimulating factor in the development of progenitors in the common myeloid pathway. Blood 2003;102:3562–3568.
Semerad CL, Poursine-Laurent J, Liu F, Link DC: A role for G-CSF receptor signaling in the regulation of hematopoietic cell function but not lineage commitment or differentiation. Immunity 1999;11:153–161.
Basu S, Hodgson G, Zhang HH, Katz M, Quilici C, Dunn AR: “Emergency” granulopoiesis in G-CSF-deficient mice in response to Candida albicans infection. Blood 2000;95:3725–3733.
Forlow SB, Schurr JR, Kolls JK, Bagby GJ, Schwarzenberger PO, Ley K: Increased granulopoiesis through interleukin-17 and granulocyte colony-stimulating factor in leukocyte adhesion molecule-deficient mice. Blood 2001;98:3309–3314.
Kotto-Kome AC, Fox SE, Lu W, Yang BB, Christensen RD, Calhoun DA: Evidence that the granulocyte colony-stimulating factor (G-CSF) receptor plays a role in the pharmacokinetics of G-CSF and PegG-CSF using a G-CSF-R KO model. Pharmacol Res 2004;50:55–58.
Emmons RV, Reid DM, Cohen RL, et al: Human thrombopoietin levels are high when thrombocytopenia is due to megakaryocyte deficiency and low when due to increased platelet destruction. Blood 1996;87: 4068–4071.
Levesque JP, Hendy J, Takamatsu Y, Williams B, Winkler IG, Simmons PJ: Mobilization by either cyclophosphamide or granulocyte colony-stimulating factor transforms the bone marrow into a highly proteolytic environment. Exp Hematol 2002;30:440–449.
Petit I, Szyper-Kravitz M, Nagler A, et al: G-CSF induces stem cell mobilization by decreasing bone marrow SDF-1 and up-regulating CXCR4 [erratum appears in Nat Immunol 2002;3(8):787]. Nat Immunol 2002;3:687–694.
Levesque JP, Hendry J, Takamatsu Y, Simmons PJ, Bendall LJ: Disruption of the CXCR4/CXCL12 chemotactic interaction induced by GSCF or cyclophosphamide. J Clin Invest 2003;111:187–196.
Levesque JP, Takamatsu Y, Nilsson SK, Haylock DN, Simmons PJ: Vascular cell adhesion molecule-1 (CD106) is cleaved by neutrophil proteases in the bone marrow following hematopoietic progenitor cell mobilization by granulocyte colony-stimulating factor. Blood 2001;98:1289–1297.
Heissig B, Hattori K, Dias S, et al: Recruitment of stem and progenitor cells from the bone marrow niche requires MMP-9 mediated release of kit-ligand. Cell 2002;109:625–637.
Levesque JP, Liu F, Simmons PJ, et al: Characterization of hematopoietic progenitor mobilization in protease-deficient mice. Blood 2004;104:65–72.
Christopherson KW, Cooper S, Broxmeyer HE: Cell surface peptidase CD26/DPPIV mediates G-CSF mobilization of mouse progenitor cells. Blood 2003;101: 4680–4686.
Aiuti A, Webb IJ, Bleul C, Springer T, Gutierrez-Ramos JC: The chemokine SDF-1 is a chemoattractant for human CD34+ hematopoietic progenitor cells and provides a new mechanism to explain the mobilization of CD34+ progenitors to peripheral blood. J Exp Med 1997;185:111–120.
Bleul CC, Fuhlbrigge RC, Casasnovas JM, Aiuti A, Springer TA: A highly efficatious lymphocyte chemoattractant, stromal cell-derived factor 1 (SDF-1). J Exp Med 1996;184:1101–1109.
Chan JR, Hyduk SJ, Cybulsky MI: Chemoattractants induce a rapid and transient upregulation of monocyte alpha4 integrin affinity for vascular cell adhesion molecule 1 which mediates arrest: an early step in the process of emigration. J Exp Med 2001;193:1149–1158.
Nagase H, Miyamasu M, Yamaguchi M, et al: Cytokine-mediated regulation of CXCR4 expression in human neutrophils. J Leukoc Biol 2002;71:711–717.
Peled A, Kollet O, Ponomaryov T, et al: The chemokine SDF-1 activates the integrins LFA-1, VLA-4, and VLA-5 on immature human CD34(+) cells: role in transendothelial/stromal migration and engraftment of NOD/SCID mice. Blood 2000;95:3289–3296.
Lataillade JJ, Clay D, Bourin P, et al: Stromal cell-derived factor 1 regulates primitive hematopoiesis by suppressing apoptosis and by promoting G(0)/G(1) transition in CD34(+) cells: evidence for an autocrine/paracrine mechanism. Blood 2002;99:1117–1129.
Lataillade JJ, Clay D, Dupuy C, et al: Chemokine SDF-1 enhances circulating CD34(+) cell proliferation in synergy with cytokines: possible role in progenitor survival. Blood 2000;95:756–768.
Oberlin E, Amara A, Bachelerie F, et al: The CXC chemokine SDF-1 is the ligand for LESTR/fusin and prevents infection by T-cell-line-adapted HIV-1. Nature 1996;382:833–835.
Bleul CC, Farzan M, Choe H, et al: The lymphocyte chemoattractant SDF-1 is a ligand for LESTR/fusin, and blocks HIV-1 entry. Nature 1996;382:829–833.
Ma Q, Jones D, Borghesani PR, et al: Impaired B-lymphopoiesis, myelopoiesis, and derailed cerebellar neuron migration in CXCR4- and SDF-1-deficient mice. Proc Natl Acad Sci USA 1998;95:9448–9453.
Nagasawa T, Hirota S, Tachibana K, Takakura N, Nishikawa S, Kitamura Y, Yoshida N, Kikutani H, Kishimoto T: Defects of B-cell lymphopoiesis and bone-marrow myelopoiesis in mice lacking the CXC chemokine PBSF/SDF-1. Nature 1996;382:635–638.
Ma Q, Jones D, Springer TA: The chemokine receptor CXCR4 is required for the retention of B lineage and granulocytic precursors within the bone marrow microenvironment. Immunity 1999;10:463–471.
Broxmeyer HE, Hangoc G, Cooper S, et al: Interference of the SDF-1/CXCR4 axis in mice with AMD3100 induces rapid high level mobilization of hematopoietic progenitor cells, and AMD3100 acts synergistically with G-CSF and MIP-1α to mobilize progenitors. Blood 2001;98:811a.
Liles WC, Broxmeyer HE, Rodger E, et al: Mobilization of hematopoietic progenitor cells in healthy volunteers by AMD3100, a CXCR4 antagonist. Blood 2003; 102:2728–2730.
Martin C, Burdon PC, Bridger G, Gutierrez-Ramos JC, Williams TJ, Rankin SM: Chemokines acting via CXCR2 and CXCR4 control the release of neutrophils from the bone marrow and their return following sensescence. Immunity 2003;19:583–593.
Vroon A, Heijnen CJ, Raatgever R, et al: GRK6 deficiency is associated with enhanced CXCR4-mediated neutrophil chemotaxis in vitro and impaired responsiveness to G-CSF in vivo. J Leukoc Biol 2004;75:698–704.
Suratt BT, Petty JM, Young SK, et al: Role of the CXCR4/SDF-1 chemokine axis in circulating neutrophil homeostasis. Blood 2004;104:565–571.
Ponomaryov T, Peled A, Petit I, et al: Induction of the chemokine stromal-derived factor-1 following DNA damage improves human stem cell function. J Clin Invest 2000;106:1331–1339.
Ceradini DJ, Kulkarni AR, Callaghan MJ, et al: Progenitor cell trafficking is regulated by hypoxic gradients through HIF-1 induction of SDF-1. Nat Med 2004; 10:858–864.
Tokoyoda K, Egawa T, Sugiyama T, Choi BI, Nagasawa T: Cellular niches controlling B lymphocyte behavior within bone marrow during development. Immunity 2004;20:707–718.
Ara T, Tokoyoda K, Sugiyama T, Egawa T, Kawabata K, Nagasawa T: Long-term hematopoietic stem cells require stromal cell-derived factor-1 for colonizing bone marrow during ontogeny. Immunity 2003;19:257–267.
Liu F, Poursine-Laurent J, Link DC: Expression of the G-CSF receptor on hematopoietic progenitor cells is not required for their mobilization by G-CSF. Blood 2000;95:3025–3031.
Gorlin RJ, Gelb B, Diaz GA, Lofsness KG, Pittelkow MR, Fenyk JR Jr: WHIM syndrome, an, autosomal dominant disorder: clinical, hematological, and molecular studies. Am J Med Genet 2000;91:368–376.
Hord JD, Whitlock JA, Gay JC, Lukens JN: Clinical features of myelokathexis and treatment with hematopoietic cytokines: a case report of two patients and review of the literature. J Pediatr Hematol Oncol 1997;19:443–448.
Wetzler M, Talpaz M, Kellagher MJ, Gutterman JU, Kurzrock R: Myelokathexis: normalization of neutrophil counts and morphology by GM-CSF. JAMA 1992;267:2179, 2180.
Aprikyan AA, Liles WC, Park JR, Jonas M, Chi EY, Dale DC: Myelokathexis, a congenital disorder of severe neutropenia characterized by accelerated apoptosis and defective expression of bcl-x in neutrophil precursors. Blood 2000;95:320–327.
Taniuchi S, Yamamoto A, Fujiwara T, Hasui M, Tsuji S, Kobayashi Y: Dizygotic twin sisters with myelokathexis: mechanism of its neutropenia. Am J Hematol 1999;62:106–111.
Hernandez PA, Gorlin RJ, Lukens JN, et al: Mutations in the chemokine receptor gene CXCR4 are associated with WHIM syndrome, a combined immunodeficiency disease. Nat Genet 2003;34:70–74.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Link, D.C. Neutrophil homeostasis. Immunol Res 32, 169–178 (2005). https://doi.org/10.1385/IR:32:1-3:169
Issue Date:
DOI: https://doi.org/10.1385/IR:32:1-3:169