Abstract
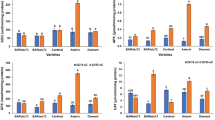
The effect of common intracellular signals (Ca2+ and cAMP) on the activity of protein phosphorylation in mitochondria was investigated in coleoptiles of maize (Zea mays L.). Treatment of isolated mitochondria with 2 mM CaCl2 brought about an increase in the level of phosphorylation of proteins with mol ws of 74, 60, and 33 kD but considerably reduced phosphorylation of the protein with a mol wt of 51.5 kD. In the presence of Ca2+, phosphorylation of polypeptides with mol wts of 59 and 66 kD was also detected. cAMP considerably reduced phosphorylation of essentially all the investigated proteins in isolated mitochondria, which could be explained by activation of their dephosphorylation. Phosphorylation of mitochondrial proteins involves a polypeptide of about 94 kD showing kinase activity, which may be proper protein kinase or one of the subunits of a compound structure. In maize mitochondria, PP1A phosphatases were found. A hypothesis was advanced that redox-dependent phosphorylation/dephosphorylation of mitochondrial proteins plays an important role in mitochondrial signaling in higher plants.
Similar content being viewed by others
Abbreviations
- BC:
-
buffer for chromatography
- HSP:
-
heat shock proteins
- MnSOD:
-
superoxide dismutase containing Mn
- MPT:
-
mitochondrial permeability transition
- PMSF:
-
phenylmethylsulfonyl fluoride
References
Pagliarini, D.J. and Dixon, J.E., Mitochondrial Modulation: Reversible Phosphorylation Takes Center Stage? Trends Biochem. Sci., 2006, vol. 31, pp. 26–34.
Pfannschmidt, T., Chloroplast Redox Signals: How Photosynthesis Controls Its Own Genes, Trends Plant Sci., 2003, vol. 8, pp. 33–41.
Subota, I.Yu., Arziev, A.Sh., and Konstantinov, Yu.M., The Involvement of Protein Phosphorylation/Dephosphorylation in the Redox Control of Translation in Cereal Mitochondria, Russ. J. Plant Physiol., 2004, vol. 51, pp. 784–789.
Juszczuk, I.M., Bykova, N.V., and Moller, I.M., Protein Phosphorylation in Plant Mitochondria, Physiol. Plant., 2007, vol. 129, pp. 90–103.
Wang, D., Harper, J.F., and Gribskov, M., Systematic Trans-Genomic Comparison of Protein Kinases between Arabidopsis and Saccharomyces cerevisiae, Plant Physiol., 2003, vol. 132, pp. 2152–2165.
Manning, G., Whyte, D.B., Martinez, R., Hunter, T., and Sudarsanam, S., The Protein Kinase Complement of the Human Genome, Science, 2002, vol. 298, pp. 1912–1934.
Bykova, N.V., Egsgaard, H., and Moller, I.M., Identification of 14 New Phosphoproteins Involved in Important Plant Mitochondrial Processes, FEBS Lett., 2003, vol. 540, pp. 141–146.
Lund, A.A., Rhoads, D.M., Lund, A.L., Cerny, R.L., and Elthon, T.E., In Vivo Modifications of the Maize Mitochondrial Small Heat Stress Protein, HSP22, J. Biol. Chem., 2001, vol. 276, pp. 924–929.
Moller, I.M., Plant Mitochondria and Oxidative Stress. Electron Transport, NADPH Turnover and Metabolism of Reactive Oxygen Species, Annu. Rev. Plant Physiol. Plant Mol. Biol., 2001, vol. 52, pp. 561–591.
Subota, I.Yu., Arziev, A.Sh., Senzhenko, L.P., Tarasenko, V.I., and Konstantinov, Yu.M., Inhibitor Analysis of Protein Phosphorylation/Dephosphorylation in Maize Mitochondria in Relation to Redox Conditions, Russ. J. Plant Physiol., 2007, vol. 54, pp. 343–349.
Papa, S., Sardanelli, A.M., Scacco, S., Petruzzella, V., Technikova-Dobrova, Z., Vergari, R., and Signorile, A., The NADH: Ubiquinone Oxidoreductase (Complex I) of the Mammalian Respiratory Chain and the cAMP Cascade, J. Bioenerg. Biomembr., 2002, vol. 34, pp. 1–10.
Livigni, A., Scorziello, A., Agnese, S., Andronetto, A., Carlucci, A., Garbi, C., Castaldo, I., Annunziato, L., Avvedimento, E.V., and Feliciello, A., Mitochondrial AKAP121 Links cAMP and Scr Signaling to Oxidative Metabolism, Mol. Biol. Cell, 2006, vol. 17, pp. 263–271.
Assmann, S.M., Cyclic AMP as Second Messenger in Higher Plants, Plant Physiol., 1995, vol. 108, pp. 885–889.
Konstantinov, Yu.M., Lutsenko, G.N., and Podsosonny, V.A., Inhibition of Adenine Nucleotide Translocation in Maize Seedling Mitochondria by Anionic Detergents, Physiol. Plant., 1988, vol. 72, pp. 403–406.
Geahlen, R.L., Austario, M., Low, P.S., and Harrison, M.L., Detection of Protein Kinase Activity in Sodium Dodecyl Sulfate Polyacrylamide Gels, Anal. Biochem., 1986, vol. 153, pp. 151–158.
Laemmli, U.K., Cleavage of Structural Proteins during the Assembly of Head of Bacteriophage T4, Nature, 1970, vol. 277, pp. 174–182.
Struglics, A., Fredlund, K.M., Konstantinov, Yu.M., Allen, J.F., and Moller, I.M., Protein Phosphorylation/Dephosphorylation in the Inner Membrane of Potato Tuber Mitochondria, FEBS Lett., 2000, vol. 475, pp. 213–217.
Lowry, O.H., Rosebrough, N.J., Farr, A.L., and Randall, R.J., Protein Measurements with the Folin Phenol Reagent, J. Biol. Chem., 1951, vol. 193, pp. 265–275.
Hulley, P. and Davison, A., Regulation of Tyrosine Phosphorylation Cascades by Phosphatases: What the Actions of Vanadium Teach Us, J. Trace Elem. Exp. Med., 2003, vol. 16, pp. 281–290.
Konstantinov, Yu.M., Subota, I.Yu., and Arziev, A.Sh., Effect of Glutathione Redox System on Maize Mitochondrial Translation Activity, Dokl. Akad. Nauk, 1999, vol. 365, pp. 126–128.
Feliciello, A., Gottesman, M.E., and Avvedimento, E.V., cAMP-PKA Signaling to the Mitochondria: Protein Scaffolds, mRNA and Phosphatases, Cell Signal., 2005, vol. 17, pp. 279–287.
Signorile, A., Sardanelli, A.M., Nuzzi, R., and Papa, S., Serine (Threonine) Phosphatase(s) Acting on cAMP-Dependent Phosphoproteins in Mammalian Mitochondria, FEBS Lett., 2002, vol. 512, pp. 91–92.
Hepler, P.K., Calcium: A Central Regulator of Plant Growth and Development, Plant Cell, 2005, vol. 17, pp. 2142–2155.
Virolainen, E., Blokhina, O., and Fagerstedt, K., Ca2+-Induced High Amplitude Swelling and Cytochrome c Release from Wheat (Triticum aestivum L.) Mitochondria under Anoxic Stress, Ann. Bot., 2005, vol. 90, pp. 509–516.
Bernardi, P., Scorrano, L., Collona, R., Peronilli, V., and di Lisa, F., Mitochondria and Cell Death. Mechanistic Aspects and Methodological Issues, Eur. J. Biochem., 1999, vol. 264, pp. 687–701.
Tiwari, B.S., Belenghi, B., and Levine, A., Oxidative Stress Increased Respiration and Generation of Reactive Oxygen Species, Resulting in ATP Depletion, Opening of Mitochondria Permeability Transition, and Programmed Cell Death, Plant Physiol., 2002, vol. 128, pp. 1271–1281.
Petrussa, E., Casolo, V., Braidot, E., Chiandussi, E., Macri, F., and Vianello, A., Cyclosporin A Induces the Opening of a Potassium-Selective Channel in Higher Plant Mitochondria, J. Bioenerg. Biomembr., 2001, vol. 33, pp. 107–117.
Feilner, T., Hultschig, C., Lee, J., Meyer, S., Immink, R.G.H., Koening, A., Possling, A., Seitz, H., Beveridge, A., Scheel, D., Cahill, D.J., Lehrach, H., Kreutzberger, J., and Kersten, B., High Throughput Identification of Potential Arabidopsis Mitogen-Activated Protein Kinases Substrates, Mol. Cell. Proteomics, 2005, vol. 4, pp. 1558–1568.
Hopper, R.K., Carroll, S., Aponte, A.M., Johnson, D.T., French, S., Shen, R.-F., Witzmann, F.A., Harris, R.A., and Balaban, R.S., Mitochondrial Matrix Phosphoproteome: Effect of Extra Mitochondrial Calcium, Biochemistry, 2006, vol. 45, pp. 2524–2536.
Azarashvilli, T.S., Tyynela, J., Odinokova, I.V., Grigorjev, P.A., Baumann, M., Evdodienko, Y.M., and Saris, N.E., Phosphorylation of Peptide Related to Subunit c of the F0F1-ATPase/ATP Synthase and Relationship to Permeability Transition Pore Opening in Mitochondria, J. Bioenerg. Biomembr., 2002, vol. 34, pp. 279–284.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.Yu. Subota, A.Sh. Arziev, L.P. Senzhenko, V.I. Tarasenko, G.A. Nevinskii, Yu.M. Konstantinov, 2010, published in Fiziologiya Rastenii, 2010, vol. 57, No. 1, pp. 42–49.
Rights and permissions
About this article
Cite this article
Subota, I.Y., Arziev, A.S., Senzhenko, L.P. et al. Effect of Ca2+ and cAMP on protein phosphorylation in mitochondria of maize seedlings. Russ J Plant Physiol 57, 37–44 (2010). https://doi.org/10.1134/S102144371001005X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S102144371001005X