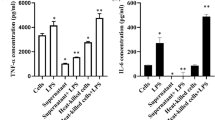
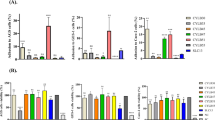
Abstract
Enterococcus bacteria are studied in various sectors including fermentation, food and dairy industries,as well as studied for their probiotic properties but have limited use due to their possible pathogenic behavior. The present report talks about the metabolites produced, by the previously isolated Enterococcus strain, E.villorum SB2 (accession number KX830968), from the vaginal source. The growth of the bacteria in three types of media (M17, MRS and LAPTg) was compared, where the M17 media gave better bacterial colonies, also maximum growth rate was observed in M17 media (Td = 1.6 h & k = 0.4 h−1), and thus was selected as the metabolite production media. Further, the studied bacteria did not show any hemolytic activity, making it safe for industrial applications. The HR-LCMS results showed the production of various amino acids, organic acids, peptides, and other metabolites like flavonoids (Quercetin 3-O-Manoglucoside), terpenoids (7’,8’,Dihydro-8’-hydroxycitraniaxanthin, O-Methylganoderic acid O, Thalicsessine, Austinol, Valdiate), indole derivatives produced by tryptophan metabolism (5-hydroxykynurenamine, 2S,4R)-4-(9H-Pyrido[3,4-b]indol-1-yl)-1,2,4-butanetriol, Indoleacrylic acid), antimicrobial compounds (Fortimicin A) and fatty acids (Stearic acid, Myristic acid), which were earlier unreported form Enterococcus species opening new scope for discovering new industrial applications of the strain. As the studied bacteria has been reported to be a potential probiotic, the detection of these industrially important metabolites can be studied further in future studies to reveal the potential industrial applications of the strain.



Similar content being viewed by others
References
Agus A, Planchais J, Sokol H (2018) Gut microbiota regulation of tryptophan metabolism in health and disease. Cell Host Microbe 23:716–724. https://doi.org/10.1016/j.chom.2018.05.003
Anjum J, Zaidi A, Barrett K, Tariq M (2022) A potentially probiotic strain of Enterococcus faecalis from human milk that is avirulent, antibiotic sensitive, and nonbreaching of the gut barrier. Arch Microbiol 204:158. https://doi.org/10.1007/s00203-022-02754-8
Arrieta M-C, Stiemsma LT, Dimitriu PA et al (2015) Early infancy microbial and metabolic alterations affect risk of childhood asthma. Sci Transl Med 7:30RAL752. https://doi.org/10.1126/scitranslmed.aab2271
Asahi S, Tsunemi Y, Doi M (1986) Method for production of cytidine and/or deoxycytidine.
Avalos M, Garbeva P, Vader L et al (2022) Biosynthesis, evolution and ecology of microbial terpenoids. Nat Prod Rep 39:249–272. https://doi.org/10.1039/D1NP00047K
Baldewijns S, Sillen M, Palmans I et al (2021) The role of fatty acid metabolites in vaginal health and disease: application to candidiasis. Front Microbiol. https://doi.org/10.3389/fmicb.2021.705779
Belviso S, Giordano M, Dolci P, Zeppa G (2011) Degradation and biosynthesis of terpenoids by lactic acid bacteria isolated from cheese: first evidence. Dairy Sci Technol 91:227–236. https://doi.org/10.1007/s13594-011-0003-z
Bhagwat A, Annapure US (2019a) In vitro assessment of physiological properties of enterococcus strains of human origin for possible probiotic use. Asian J Pharm Clin Res. https://doi.org/10.22159/ajpcr.2019a.v12i6.33294
Bhagwat A, Annapure US (2019b) Maternal-neonatal transmission of Enterococcus strains during delivery. Beni-Suef Univ J Basic Appl Sci 8:25. https://doi.org/10.1186/s43088-019-0029-5
Bhagwat A, Annapure US (2019c) In vitro assessment of metabolic profile of Enterococcus strains of human origin. J Genet Eng Biotechnol. https://doi.org/10.1186/s43141-019-0009-0
Carrau FM, Boido E, Dellacassa E (2008) Terpenoids in grapes and wines: origin and micrometabolism during the vinification Process. Nat Prod Commun 3:1934578X0800300419. https://doi.org/10.1177/1934578X0800300419
Carvalho AS, Silva J, Ho P et al (2003) Effect of various growth media upon survival during storage of freeze-dried Enterococcus faecalis and Enterococcus durans. J Appl Microbiol 94:947–952. https://doi.org/10.1046/j.1365-2672.2003.01853.x
Cotter PD, Ross RP, Hill C (2013) Bacteriocins — a viable alternative to antibiotics? Nat Rev Microbiol 11:95–105. https://doi.org/10.1038/nrmicro2937
Cryan J, Dinan T (2012) Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci 13:701–712. https://doi.org/10.1038/nrn3346
Cunha DV, Salazar SB, Lopes MM, Mira NP (2017) Mechanistic insights underlying tolerance to acetic acid stress in vaginal candida glabrata clinical isolates. Front Microbiol. https://doi.org/10.3389/fmicb.2017.00259
De Vuyst L, Foulquié Moreno MR, Revets H (2003) Screening for enterocins and detection of hemolysin and vancomycin resistance in enterococci of different origins. Int J Food Microbiol 84:299–318. https://doi.org/10.1016/s0168-1605(02)00425-7
Dehhaghi M, Kazemi Shariat Panahi H, Guillemin GJ (2019) Microorganisms, tryptophan metabolism, and kynurenine pathway: a complex interconnected loop influencing human health status. Int J Tryptophan Res IJTR 12:1178646919852996. https://doi.org/10.1177/1178646919852996
Dinçer E, Kıvanç M (2021) In vitro evaluation of probiotic potential of Enterococcus faecium strains isolated from Turkish pastırma. Arch Microbiol 203:2831–2841. https://doi.org/10.1007/s00203-021-02273-y
Dupont H, Montravers P, Mohler J, Carbon C (1998) Disparate findings on the role of virulence factors of Enterococcus faecalis in mouse and rat models of peritonitis. Infect Immun 66:2570–2575
Durack J, Kimes NE, Lin DL et al (2018) Delayed gut microbiota development in high-risk for asthma infants is temporarily modifiable by Lactobacillus supplementation. Nat Commun 9:707. https://doi.org/10.1038/s41467-018-03157-4
Eaton TJ, Gasson MJ (2001) Molecular screening of enterococcus virulence determinants and potential for genetic exchange between food and medical isolates. Appl Environ Microbiol 67:1628–1635. https://doi.org/10.1128/AEM.67.4.1628-1635.2001
El-Gendy AO, Brede DA, Essam TM et al (2021) Purification and characterization of bacteriocins-like inhibitory substances from food isolated Enterococcus faecalis OS13 with activity against nosocomial enterococci. Sci Rep 11:3795. https://doi.org/10.1038/s41598-021-83357-z
Giraffa G (1995) Enterococcal bacteriocins: their potential as anti-Listeria factors in dairy technology. Food Microbiol 12:291–299. https://doi.org/10.1016/S0740-0020(95)80109-X
Girolami RL, Stamm JM (1977) Fortimicins A and B, new aminoglycoside antibiotics. IV. In vitro study of fortimicin A compared with other aminoglycosides. J Antibiot (Tokyo) 30:564–70. https://doi.org/10.7164/antibiotics.30.564
Gross F, Lewis EA, Piraee M et al (2002) Isolation of 3’ -O-acetylchloramphenicol: a possible intermediate in chloramphenicol biosynthesis. Bioorg Med Chem Lett 12:283–286. https://doi.org/10.1016/s0960-894x(01)00739-9
Hadji-Sfaxi I, El-Ghaish S, Ahmadova A et al (2011) Antimicrobial activity and safety of use of Enterococcus faecium PC4.1 isolated from Mongol yogurt. Food Control 22:2020–2027. https://doi.org/10.1016/j.foodcont.2011.05.023
Hall BG, Acar H, Nandipati A, Barlow M (2014) Growth rates made easy. Mol Biol Evol 31:232–238. https://doi.org/10.1093/molbev/mst187
Hanchi H, Hammami R, Kourda R et al (2014) Bacteriocinogenic properties and in vitro probiotic potential of enterococci from Tunisian dairy products. Arch Microbiol 196:331–344. https://doi.org/10.1007/s00203-014-0978-y
Hayaloglu AA (2016) Cheese: Microbiology of cheese. Reference module in food science. Elsevier, Amsterdam
Huong NL, Hoang NH, Hong S-Y et al (2016) Characterization of fortimicin aminoglycoside profiles produced from Micromonospora olivasterospora DSM 43868 by high-performance liquid chromatography-electrospray ionization-ion trap-mass spectrometry. Anal Bioanal Chem 408:1667–1678. https://doi.org/10.1007/s00216-015-9281-2
Ike Y, Clewell DB, Segarra RA, Gilmore MS (1990) Genetic analysis of the pAD1 hemolysin/bacteriocin determinant in Enterococcus faecalis: Tn917 insertional mutagenesis and cloning. J Bacteriol 172:155–163. https://doi.org/10.1128/jb.172.1.155-163.1990
Jett BD, Jensen HG, Nordquist RE, Gilmore MS (1992) Contribution of the pAD1-encoded cytolysin to the severity of experimental Enterococcus faecalis endophthalmitis. Infect Immun 60:2445–2452. https://doi.org/10.1128/iai.60.6.2445-2452.1992
Kang BS, Seo J-G, Lee G-S et al (2009) Antimicrobial activity of enterocins from Enterococcus faecalis SL-5 against Propionibacterium acnes, the causative agent in acne vulgaris, and its therapeutic effect. J Microbiol 47:101–109. https://doi.org/10.1007/s12275-008-0179-y
Kim M, Kim N, Han J (2016) Deglycosylation of flavonoid O-glucosides by human intestinal bacteria Enterococcus sp. MRG-2 and Lactococcus sp. MRG-IF-4. Appl Biol Chem 59:443–449. https://doi.org/10.1007/s13765-016-0183-6
KumarRitika S (2020) A brief review of the biological potential of indole derivatives. Future J Pharm Sci 6:121. https://doi.org/10.1186/s43094-020-00141-y
Lee M-T, Le HH, Johnson EL (2021) Dietary sphinganine is selectively assimilated by members of the mammalian gut microbiome. J Lipid Res. https://doi.org/10.1194/jlr.RA120000950
Li Y, Wang Y, Wu P (2019) 5’-Methylthioadenosine and Cancer: old molecules, new understanding. J Cancer 10:927–936. https://doi.org/10.7150/jca.27160
Libertin CR, Dumitru R, Stein DS (1992) The hemolysin/bacteriocin produced by enterococci is a marker of pathogenicity. Diagn Microbiol Infect Dis 15:115–120. https://doi.org/10.1016/0732-8893(92)90033-p
Lo H-C, Entwistle R, Guo C-J et al (2012) Two separate gene clusters encode the biosynthetic pathway for the meroterpenoids austinol and dehydroaustinol in Aspergillus nidulans. J Am Chem Soc 134:4709–4720. https://doi.org/10.1021/ja209809t
Melander RJ, Minvielle MJ, Melander C (2014) Controlling bacterial behavior with indole-containing natural products and derivatives. Tetrahedron 70:6363–6372. https://doi.org/10.1016/j.tet.2014.05.089
Mokoena MP, Chelule PK, Gqaleni N (2005) Reduction of fumonisin B1 and zearalenone by lactic acid bacteria in fermented maize meal. J Food Prot 68:2095–2099. https://doi.org/10.4315/0362-028X-68.10.2095
Moore BS, Hertweck C, Hopke JN et al (2002) Plant-like biosynthetic pathways in bacteria: from benzoic acid to chalcone. J Nat Prod 65:1956–1962. https://doi.org/10.1021/np020230m
Moreno MF, Sarantinopoulos P, Tsakalidou E, De Vuyst L (2006) The role and application of enterococci in food and health. Int J Food Microbiol 106:1–24. https://doi.org/10.1016/j.ijfoodmicro.2005.06.026
Motarjemi Y, Adams M (2006) Emerging foodborne pathogens. CRC Press, Boca Raton
Nami Y, Abdullah N, Haghshenas B et al (2014) A newly isolated probiotic Enterococcus faecalis strain from vagina microbiota enhances apoptosis of human cancer cells. J Appl Microbiol 117:498–508. https://doi.org/10.1111/jam.12531
Nara T, Yamamoto M, Kawamoto I et al (1977) Fortimicins A and B, new aminoglycoside antibiotics. I. Producing organism, fermentation and biological properties of fortimicins. J Antibiot (tokyo) 30:533–540. https://doi.org/10.7164/antibiotics.30.533
네이버 학술정보. Available at https://academic.naver.com/article.naver?doc_id=302629212. Accessed 28 Mar 2022
Nguyen H, Nguyen Lan H, Kim B et al (2016) Istamycin aminoglycosides profiling and their characterization in Streptomyces tenjimariensis ATCC 31603 culture using high-performance liquid chromatography with tandem mass spectrometry. J Sep Sci. https://doi.org/10.1002/jssc.201600925
O’Hanlon DE, Moench TR, Cone RA (2011) In vaginal fluid, bacteria associated with bacterial vaginosis can be suppressed with lactic acid but not hydrogen peroxide. BMC Infect Dis 11:200. https://doi.org/10.1186/1471-2334-11-200
Oliver A, LaMere B, Weihe C et al (2020) Cervicovaginal microbiome composition is associated with metabolic profiles in healthy pregnancy. mBio 11:e 01851-20. https://doi.org/10.1128/mBio.01851-20
Olsen I, Jantzen E (2001) Sphingolipids in bacteria and fungi. Anaerobe 7:103–112. https://doi.org/10.1006/anae.2001.0376
Ossai J, Khatabi B, Nybo SE, Kharel MK (2022) Renewed interests in the discovery of bioactive actinomycete metabolites driven by emerging technologies. J Appl Microbiol 132:59–77. https://doi.org/10.1111/jam.15225
Petrova MI, Reid G, Vaneechoutte M, Lebeer S (2017) Lactobacillus iners: Friend or Foe? Trends Microbiol 25:182–191. https://doi.org/10.1016/j.tim.2016.11.007
Phumisantiphong U, Siripanichgon K, Reamtong O, Diraphat P (2017) A novel bacteriocin from Enterococcus faecalis 478 exhibits a potent activity against vancomycin-resistant enterococci. PLoS ONE 12:e0186415. https://doi.org/10.1371/journal.pone.0186415
Ravel J, Gajer P, Abdo Z et al (2011) Vaginal microbiome of reproductive-age women. Proc Natl Acad Sci U S A 108(Suppl 1):4680–4687. https://doi.org/10.1073/pnas.1002611107
Rohrhofer J, Zwirzitz B, Selberherr E, Untersmayr E (2021) The impact of dietary sphingolipids on intestinal microbiota and gastrointestinal immune homeostasis. Front Immunol 12:635704. https://doi.org/10.3389/fimmu.2021.635704
Russo P, López P, Capozzi V et al (2012) Beta-glucans improve growth, viability and colonization of probiotic microorganisms. Int J Mol Sci 13:6026–6039. https://doi.org/10.3390/ijms13056026
Russo D, Milella L (2020) Chapter 14 - Analysis of meroterpenoids. In: Sanches Silva A, Nabavi SF, Saeedi M, Nabavi SM (eds) Recent advances in natural products analysis. . Elsevier, Amsterdam, pp 477–501
Seshime Y, Juvvadi PR, Fujii I, Kitamoto K (2005) Genomic evidences for the existence of a phenylpropanoid metabolic pathway in Aspergillus oryzae. Biochem Biophys Res Commun 337:747–751. https://doi.org/10.1016/j.bbrc.2005.08.233
Shin NR, Moon JS, Shin S-Y et al (2016) Isolation and characterization of human intestinal Enterococcus avium EFEL009 converting rutin to quercetin. Lett Appl Microbiol 62:68–74. https://doi.org/10.1111/lam.12512
Smith MI, Yatsunenko T, Manary MJ et al (2013) Gut microbiomes of Malawian twin pairs discordant for kwashiorkor. Science 339:548–554. https://doi.org/10.1126/science.1229000
Tachedjian G, Aldunate M, Bradshaw CS, Cone RA (2017) The role of lactic acid production by probiotic Lactobacillus species in vaginal health. Res Microbiol 168:782–792. https://doi.org/10.1016/j.resmic.2017.04.001
Terzaghi BE, Sandine WE (1975) Improved medium for lactic streptococci and their bacteriophages. Appl Microbiol 29:807–813. https://doi.org/10.1128/am.29.6.807-813.1975
Veselá M, Drdák M, Simon P, Veselý M (2002) Utilization of Lactobacillus sp. for steroid glycoalkaloids degradation by lactic acid fermentation. Nahr 46:251–255. https://doi.org/10.1002/1521-3803(20020701)46:4%3c251::AID-FOOD251%3e3.0.CO;2-U
Villas-Bôas S, Bruheim P (2007) Cold glycerol-saline: the promising quenching solution for accurate intracellular metabolite analysis of microbial cells. Anal Biochem 370:87–97. https://doi.org/10.1016/j.ab.2007.06.028
Wlodarska M, Luo C, Kolde R et al (2017) Indoleacrylic acid produced by commensal peptostreptococcus species suppresses inflammation. Cell Host Microbe 22:25-37.e6. https://doi.org/10.1016/j.chom.2017.06.007
Yamada Y, Kuzuyama T, Komatsu M et al (2015) Terpene synthases are widely distributed in bacteria. Proc Natl Acad Sci 112:857–862. https://doi.org/10.1073/pnas.1422108112
Yamamoto M, Okachi R, Kawamoto I, Nara T (1977) Fortimicin a production by micromonospora olivoasterospora in a chemically defined medium. J Antibiot 30:1064–1072. https://doi.org/10.7164/antibiotics.30.1064
Zhao HY, Wang HJ, Lu Z, Xu SZ (2004) Intestinal microflora in patients with liver cirrhosis. Chin J Dig Dis 5:64–67. https://doi.org/10.1111/j.1443-9573.2004.00157.x
Acknowledgements
We want to express our special thanks to Bhagwat et al. (Bhagwat and Annapure 2019b) for providing us the isolated Enterococcus strains for the current study. We would like to express our gratitude towards DST and SAIF/CRNTS, IIT Bombay, for providing HR-LCMS analytical facility for the research work.
Funding
The authors declare that no funds, grants or other support was received during the preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, analysis and manuscript drafting was performed by SSG. The guidance, comments on the manuscript draft, and approval was provided by Prof. USA. Both the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors have no relevant financial or non-financial interests to disclose.
Consent to participate
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gaur, S.S., Annapure, U.S. Untargeted metabolite profiling of Enterococcus villorum SB2, isolated from the vagina of pregnant women, by HR-LCMS. World J Microbiol Biotechnol 38, 219 (2022). https://doi.org/10.1007/s11274-022-03404-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-022-03404-3