Abstract
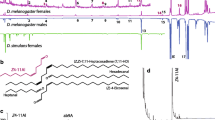
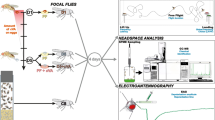
Organisms depend on visual, auditory, and olfactory cues to signal the presence of danger that could impact survival and reproduction. Drosophila melanogaster emits an olfactory alarm signal, termed the Drosophila stress odorant (dSO), in response to mechanical agitation or electric shock. While it has been shown that conspecifics avoid areas previously occupied by stressed individuals, the contextual underpinnings of the emission of, and response to dSO, have received little attention. Using a binary choice assay, we determined that neither age and sex of emitters, nor the time of the day, affected the emission or avoidance of dSO. However, both sex and mating status affected the response to dSO. We also demonstrated that while D. melanogaster, D. simulans, and D. suzukii, have different dSO profiles, its avoidance was not species-specific. Thus, dSO should not be considered a pheromone but a general alarm signal for Drosophila. However, the response levels to both intra- and inter-specific cues differed between Drosophila species and possible reasons for these differences are discussed.







Similar content being viewed by others
Data Availability
Upon request
References
Azzam ZS, Sharabi K, Guetta J, Bank EM, Gruenbaum Y (2010) The physiological and molecular effects of elevated CO2 levels. Cell Cycle 9:1528–1532. https://doi.org/10.4161/cc.9.8.11196
Bateson M, Desire S, Gartside SE, Wright GA (2011) Agitated honeybees exhibit pessimistic cognitive biases. Curr Biol 21:1070–1073. https://doi.org/10.1016/j.cub.2011.05.017
Billeter JC, Atallah J, Krupp JJ, Millar JG, Levine JD (2009) Specialized cells tag sexual and species identity in Drosophila melanogaster. Nature 461:987–991. https://doi.org/10.1038/nature08495
Brenman-Suttner DB, Long SQ, Kamisan V, de Belle JN, Yost RT, Kanippayoor RL, Simon AF (2018) Progeny of old parents have increased social space in Drosophila melanogaster. Sci Rep 8(1):3673. https://doi.org/10.1038/s41598-018-21731-0
Brenman-Suttner DB, Yost RT, Frame AK, Robinson JW, Moehring AJ, Simon AF (2019) Social behaviour and aging: a fly model. Genes Brain Behav 19:e12598. https://doi.org/10.1111/gbb.12598
Chao YC, Fleischer J, Yang RB (2018) Guanylyl cyclase-G is an alarm pheromone receptor in mice. EMBO J 37:39–49. https://doi.org/10.15252/embj.201797155
Chen N, Bai Y, Fan YL, Liu TX (2017) Solid-phase microextraction-based cuticular hydrocarbon profiling for intraspecific delimitation in Acyrthosiphon pisum. PLoS One 12(8):e0184243. https://doi.org/10.1371/journal.pone.0184243
Cook-Wiens E, Grotewiel MS (2002) Dissociation between functional senescence and oxidative stress resistance in Drosophila. Exp Gerontol 37:1345–1355. https://doi.org/10.1016/S0531-5565(02)00096-7
Dahanukar A, Ray A (2011) Courtship, aggression and avoidance: Pheromones, receptors and neurons for social behaviors in Drosophila. Fly 5:58–63. https://doi.org/10.4161/y.5.1.13794
Douglas T, Anderson R, Saltz JB (2020) Limits to male reproductive potential across mating bouts in Drosophila melanogaster. Anim Behav 160:25–33. https://doi.org/10.1016/j.anbehav.2019.11.009
Dubnau J, Varela N, Gaspar M, Dias S, Vasconcelos ML (2019) Avoidance response to CO2 in the lateral horn. PLoS Biol 17:e2006749. https://doi.org/10.1371/journal.pbio.2006749
Dubruille R, Emery P (2008) A plastic clock: how circadian rhythms respond to environmental cues in Drosophila. Mol Neurobiol 38:129–145. https://doi.org/10.1007/s12035-008-8035-y
Enjin A, Suh GS (2013) Neural mechanisms of alarm pheromone signaling. Mol Cells 35:177–181. https://doi.org/10.1007/s10059-013-0056-3
Farine JP, Ferveur JF, Everaerts C (2012) Volatile Drosophila cuticular pheromones are affected by social but not sexual experience. PLoS One 7:e40396. https://doi.org/10.1371/journal.pone.0040396
Faucher C, Forstreuter M, Hilker M, de Bruyne M (2006) Behavioral responses of Drosophila to biogenic levels of carbon dioxide depend on life-stage, sex and olfactory context. J Exp Biol 209:2739–2748. https://doi.org/10.1242/jeb.02297
Fernandez RW et al (2017) Modulation of social space by dopamine in Drosophila melanogaster, but no effect on the avoidance of the Drosophila stress odorant. Biol Let 13:20170369. https://doi.org/10.1098/rsbl.2017.0369
Fernandez RW, Akinleye AA, Nurilov M, Feliciano O, McDonald IS, Simon AF (2014) Straightforward assay for quantification of social avoidance in Drosophila melanogaster. J vis Exp 94:e52011. https://doi.org/10.3791/52011
Ferveur JF (2005) Cuticular hydrocarbons: their evolution and roles in Drosophila pheromonal communication. Behav Genet 35:279–295. https://doi.org/10.1007/s10519-005-3220-5
Hunt GJ (2007) Flight and fight: a comparative view of the neurophysiology and genetics of honey bee defensive behavior. J Insect Physiol 53:399–410. https://doi.org/10.1016/j.jinsphys.2007.01.010
Jakobs R, Ahmadi B, Houben S, Gariepy TD, Sinclair BJ (2017) Cold tolerance of third-instar Drosophila suzukii larvae. J Insect Physiol 96:45–52. https://doi.org/10.1016/j.jinsphys.2016.10.008
Krause Pham C, Ray A (2015) Conservation of olfactory avoidance in Drosophila species and identification of repellents for Drosophila suzukii. Sci Rep 5:11527. https://doi.org/10.1038/srep11527
Kumar Chaudhary M, Rizvi SI (2019) Invertebrate and vertebrate models in aging research. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub, A review. https://doi.org/10.5507/bp.2019.003
Kwon JY, Dahanukar A, Weiss LA, Carlson JR (2007) The molecular basis of CO2 reception in Drosophila. Proc Natl Acad Sci U S A 104:3574–3578. https://doi.org/10.1073/pnas.0700079104
Mathuru AS, Kibat C, Cheong WF, Shui G, Wenk MR, Friedrich RW, Jesuthasan S (2012) Chondroitin fragments are odorants that trigger fear behavior in fish. Curr Biol 22:538–544. https://doi.org/10.1016/j.cub.2012.01.061
Miller PB et al (2014) The song of the old mother: reproductive senescence in female Drosophila. Fly 8:127–139. https://doi.org/10.4161/19336934.2014.969144
Montgomery ME, Nault LR (1978) Effects of age and wing polymorphism on the sensitivity of Myzus persicae to alarm pheromone. Ann Entomol Soc Am 71:788–790. https://doi.org/10.1093/aesa/71.5.788
Mujica-Parodi LR et al (2009) Chemosensory cues to conspecific emotional stress activate amygdala in humans. PLoS ONE 4:e6415. https://doi.org/10.1371/journal.pone.0006415
Muria A, Musso PY, Durrieu M, Portugal FR, Ronsin B, Gordon MD, Jeanson R, Isabel G (2021) Social facilitation of long-lasting memory is mediated by CO2 in Drosophila. Curr Biol 31:2065–2074. https://doi.org/10.1016/j.cub.2021.02.044
Napper E, Pickett JA (2008) Alarm pheromones of insects. In: Capinera JL (ed) Encyclopedia of Entomology. Springer Netherlands, Dordrecht, pp 85–95. https://doi.org/10.1007/978-1-4020-6359-6_125
Nicolas G, Sillans D (1989) Immediate and latent effects of carbon dioxide on insects. Annu Rev Entomol 34:97–116. https://doi.org/10.1146/annurev.en.34.010189.000525
Rosewell J, Shorrocks B (1987) The implication of survival rates in natural populations of Drosophila: capture-recapture experiments on domestic species. Biol J Lin Soc 32:373–384. https://doi.org/10.1111/j.1095-8312.1987.tb00438.x
Royer L, McNeil JN (1993) Male investment in the European corn borer, Ostrinia nubilalis (Lepidoptera: Pyralidae): Impact on female longevity and reproductive performance. Funct Ecol 7:209–215. https://doi.org/10.2307/2389889
Ruhmann H, Koppik M, Wolfner MF, Fricke C (2018) The impact of ageing on male reproductive success in Drosophila melanogaster. Exp Gerontol 103:1–10. https://doi.org/10.1016/j.exger.2017.12.013
Sharabi K, Lecuona E, Helenius IT, Beitel GJ, Sznajder JI, Gruenbaum Y (2009) Sensing, physiological effects and molecular response to elevated CO2 levels in eukaryotes. J Cell Mol Med 13:4304–4318. https://doi.org/10.1111/j.1582-4934.2009.00952.x
Siju KP, Bracker LB, Grunwald Kadow IC (2014) Neural mechanisms of context-dependent processing of CO2 avoidance behavior in fruit flies. Fly (austin) 8:68–74. https://doi.org/10.4161/fly.28000
Simon AF, Liang DT, Krantz DE (2006) Differential decline in behavioral performance of Drosophila melanogaster with age. Mech Ageing Dev 127:647–651. https://doi.org/10.1016/j.mad.2006.02.006
Sokolowski MB (2010) Social interactions in “simple” model systems. Neuron 65:780–794. https://doi.org/10.1016/j.neuron.2010.03.007
Soto-Yeber L, Soto-Ortiz J, Godoy P, Godoy-Herrera R (2018) The behavior of adult Drosophila in the wild. PLoS ONE 13:e0209917. https://doi.org/10.1371/journal.pone.0209917
Suh GS et al (2004) A single population of olfactory sensory neurons mediates an innate avoidance behaviour in Drosophila. Nature 431:854–859. https://doi.org/10.1038/nature02980
Suh GS, Ben-Tabou de Leon S, Tanimoto H, Fiala A, Benzer S, Anderson DJ (2007) Light activation of an innate olfactory avoidance response in Drosophila. Curr Biol 17:905–908. https://doi.org/10.1016/j.cub.2007.04.046
Tatar M, Promislow DEL, Khazaeli AA, Curtsinger JW (1996) Age-specific patterns of genetic variance in Drosophila melanogaster. II. Fecundity and its genetic covariance with age-specific mortality. Genetics 143:849–858
Trannoy S, Chowdhury B, Kravitz EA (2015) A New Approach that Eliminates Handling for Studying Aggression and the “Loser” Effect in Drosophila melanogaster. J vis Exp 106:e53395. https://doi.org/10.3791/53395
Turner SL, Ray A (2009) Modification of CO2 avoidance behaviour in Drosophila by inhibitory odorants. Nature 461:277–281. https://doi.org/10.1038/nature08295
van Breugel F, Huda A, Dickinson MH (2018) Distinct activity-gated pathways mediate attraction and aversion to CO2 in Drosophila. Nature 564:420–424. https://doi.org/10.1038/s41586-018-0732-8
Vandermoten S, Mescher MC, Francis F, Haubruge E, Verheggen FJ (2012) Aphid alarm pheromone: an overview of current knowledge on biosynthesis and functions. Insect Biochem Mol Biol 42:155–163. https://doi.org/10.1016/j.ibmb.2011.11.008
Verheggen FJ, Haubruge E, Mescher MC (2010) Alarm pheromones—Chemical signaling in response to danger. In: Litwack G (ed) Vitamins & Hormones. Academic Press, vol 83, pp 215–239. https://doi.org/10.1016/S0083-6729(10)83009-2
Wasserstein RL, Lazar NA (2016) The ASA Statement on p-Values: Context, Process, and Purpose. Am Stat 70:129–133. https://doi.org/10.1080/00031305.2016.1154108
Yew JY, Chung H (2015) Insect pheromones: An overview of function, form, and discovery. Prog Lipid Res 59:88–105. https://doi.org/10.1016/j.plipres.2015.06.001
Yost RT, Scott AM, Walshe-Rousell B, Dukas R, Simon AF (2021) Recovery from social isolation requires dopamine in males, but not the autism-related gene nlg3 in either sex. Revisions requested: Frontiers in Neural Circuits. Manuscript ID: 734017
Zahiri N, Rau ME, Lewis DJ (1997) Starved larvae of Aedes aegypti (Diptera: Culicidae) render waters unattractive to ovipositing conspecific females. Environ Entomol 26:1087–1090. https://doi.org/10.1093/ee/26.5.1087
Zhou Y et al (2017) Potential nematode alarm pheromone induces acute avoidance in Caenorhabditis elegans. Genetics 206:1469–1478. https://doi.org/10.1534/genetics.116.197293
Ziegler AB, Berthelot-Grosjean M, Grosjean Y (2013) The smell of love in Drosophila. Front Physiol 4:72. https://doi.org/10.3389/fphys.2013.00072
Acknowledgements
We thank Natasha Bauer-Maison for her technical contribution, Yanira Jimenez Padilla, and the Sinclair laboratory for providing food for Drosophila suzukii during the experiment. Finally, we thank Justin B. Renaud for his expertise, constructive comments and oversight during dSO volatile analysis.
Funding
Graduate scholarship to RTY, Western Foundation internal grant, and NSERC Discovery Grants 04507–2015 to JNM and 04275–2015 to AFS.
Author information
Authors and Affiliations
Contributions
Data acquisition and analysis were carried out by RTY, EL, ISM, SC, ISM, TM, AFG and MS; Protocols elaborated by TM, AFS and JNM; the first draft was written by RTY, EL and AFS and the final draft edited by RTY, TM, AFS and JNM.
Corresponding author
Ethics declarations
Ethical Note
No approval is required from the Western’s Animal Care Committee or the Provincial and Federal regulatory bodies to study invertebrates. However, we provided appropriate rearing conditions and anesthetized flies using CO2 or cold anaesthesia when manipulating flies for colony maintenance. Stress treatments to produce dSO resulted in no mortality.
Preprint Finally, the behavioural portion of this manuscript can be found on bioRxiv—a preprint server for biology, operated by Cold Spring Harbor Laboratory: Yost et al. (2021) Is Drosophila melanogaster Stress Odorant (dSO) really an alarm pheromone? bioRxiv:534719 doi:10.1101/534719.
Conflicts of Interest
None
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ryley T. Yost, Emerald Liang, Megan P. Stewart, Selwyn Chui, Andrew F. Greco, and Shirley Q. Long these authors were undergraduates at the time of data collection
Rights and permissions
About this article
Cite this article
Yost, R.T., Liang, E., Stewart, M.P. et al. Drosophila melanogaster Stress Odorant (dSO) Displays the Characteristics of an Interspecific Alarm Cue. J Chem Ecol 47, 719–731 (2021). https://doi.org/10.1007/s10886-021-01300-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-021-01300-y