Abstract
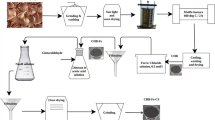
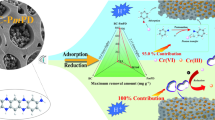
A novel approach was employed to craft innovative carbon-paste electrodes using cerium(IV) oxide (CeO2)-doped clay, aiming to modify graphite electrodes. The introduction of Ce involved the adsorption of Ce3+ ions onto an activated carbon (AC) clay mixture, showcasing an impressive adsorption capacity of 40.0 mg g−1, which surpasses that of clay alone. This step was undertaken to enhance the conductivity of raw clay and increase its electrochemical surface area. Sintering clay-CeO2 at 1000 °C resulted in the maximum electrochemical area when utilized as a 10 wt% dopant for graphite electrodes. Heavy metal ion analysis (HMI) involving Cd2+, Pb2+, Cu2+, and Hg2+ was conducted using five electrochemical techniques: Cyclic Voltammetry (CV), Square Wave Voltammetry (SWV), Normal Pulse Voltammetry (NPV), Differential Pulse Voltammetry (DPV), and Linear Sweep Voltammetry (LSV). SWV emerged as the optimal technique and was termed SWAdSV after the stripping process, displaying peak current responses for Cd2+ (74.7 µA), Pb2+ (42.4 µA), Cu2+ (36.0 µA), and Hg2+ (22.8 µA). Calibration curves were utilized to determine the Limit of Detection (LOD) and Limit of Quantification (LOQ) values for Cd2+, Pb2+, Cu2+, and Hg2+. The repeatability, expressed as %RSD, for the same electrode was found to be 1.70%, 2.04%, 1.14%, and 3.61% for Cd2+, Pb2+, Cu2+, and Hg2+. While the reproducibility, also expressed as %RSD, for different electrodes was determined to be 1.98%, 2.31%, 1.52%, and 1.89% for Cd2+, Pb2+, Cu2+, and Hg2+, respectively.
Graphical abstract











Similar content being viewed by others
Data availability
All relevant data is included in the paper.
References
Lahnafi A, Elgamouz A, Tijani N, Jaber L, Kawde AN (2022) Hydrothermal synthesis and electrochemical characterization of novel zeolite membranes supported on flat porous clay-based microfiltration system and its application of heavy metals removal of synthetic wastewaters. Microporous Mesoporous Mater 334:111778. https://doi.org/10.1016/j.micromeso.2022.111778
Jaber L, Elgamouz A, Kawde AN (2022) An insight to the filtration mechanism of Pb(II) at the surface of a clay ceramic membrane through its preconcentration at the surface of a graphite/clay composite working electrode. Arab J Chem 15(12):104303. https://doi.org/10.1016/j.arabjc.2022.104303
El-Kordy A (2022) Preparation of sodalite and faujasite clay composite membranes and their utilization in the decontamination of dye effluents. Membranes 12(1):1–18. https://doi.org/10.3390/membranes12010012
Mallik AK, Kabir SF, Bin Abdur Rahman F, Sakib MN, Efty SS, Rahman MM (2022) Cu(II) removal from wastewater using chitosan-based adsorbents: a review. J Environ Chem Eng 10(4):108048. https://doi.org/10.1016/j.jece.2022.108048
Jaber L, AbdelHamid AA, Elgamouz A, Kawde AN (2023) Prediction of a natural clay membrane selectivity towards heavy metal cations removal from wastewater: an electrochemical study. J Solid State Electrochem. https://doi.org/10.1007/s10008-023-05774-6
El-kordy A, Elgamouz A, Abdelhamid A, Kawde A (2024) Manufacturing of novel zeolite-clay composite membrane from natural clay and diatomite, an electrochemical study of the surface and application towards heavy metals removal. J Environ Chem Eng 12(2):112143. https://doi.org/10.1016/j.jece.2024.112143
Whitfield JB (2010) Genetic effects on toxic and essential elements in humans: arsenic, cadmium, copper, lead, mercury, selenium, and zinc in erythrocytes. Environ Health Perspect 118(6)776–782. https://doi.org/10.1289/ehp.0901541
Satarug S, Garrett SH, Sens MA, Sens DA (2010) Cadmium, environmental exposure, and health outcomes. Environ Health Perspect 118(2):182–190. https://doi.org/10.1289/ehp.0901234
Fisher MB (2021) Occurrence of lead and other toxic metals derived from drinking-water systems in three west african countries. Environ Health Perspect. https://doi.org/10.1289/EHP7804
Navas-Acien A, Guallar E, Silbergeld EK, Rothenberg SJ (2007) Lead exposure and cardiovascular disease—a systematic review. Environ Health Perspect 115(3):472–482. https://doi.org/10.1289/ehp.9785
Delgado CF, Ullery MA, Jordan M, Duclos C, Rajagopalan S, Scott K (2018) Lead exposure and developmental disabilities in preschool-aged children. J Public Health Manag Pract 24(2):e10–e17. https://doi.org/10.1097/PHH.0000000000000556
Ahamed M, Verma S, Kumar A, Siddiqui MKJ (2006) Delta-aminolevulinic acid dehydratase inhibition and oxidative stress in relation to blood lead among urban adolescents. Hum Exp Toxicol 25(9):547–553. https://doi.org/10.1191/0960327106het657oa
Martínez SA, Simonella L, Hansen C, Rivolta S, Cancela LM, Virgolini MB (2013) Blood lead levels and enzymatic biomarkers of environmental lead exposure in children in Córdoba, Argentina, after the ban of leaded gasoline. Hum Exp Toxicol 32(5):449–463. https://doi.org/10.1177/0960327112454893
Knollmann-Ritschel BEC, Markowitz M (2017) Educational case: lead poisoning. Acad Pathol 4(1):3. https://doi.org/10.1177/2374289517700160
Mishra S, Kannan S, Manager C, Statistics A, Comments R, Alert E (2019) Occupational and environmental exposure to lead and reproductive health impairment: an overview Indian. J Occup Int Med 23(1):8–13. https://doi.org/10.4103/ijoem.IJOEM
Kirk FT, Munk DE, Swenson ES, Schilsky ML, Ott P, Sandahl TD (2024) Effects of tetrathiomolybdate on copper metabolism in healthy volunteers and in patients with Wilson disease. J Hepatol 80(4):586–595. https://doi.org/10.1016/j.jhep.2023.11.023
Saylakcı R, Incebay H (2021) An electrochemical platform of tannic acid and carbon nanotubes for the sensitive determination of the antipsychotic medication clozapine in pharmaceutical and biological samples. J Electroanal Chem 898:115638. https://doi.org/10.1016/j.jelechem.2021.115638
El-kordy A (2023) Development and characterization of a Clay-HDTMABr composite for the removal of Cr (VI) from aqueous solutions with special emphasis on the electrochemical interface. Arab J Chem 6:105027. https://doi.org/10.1016/j.arabjc.2023.105027
Oliveira de JPJ, Hiranobe CT, Torres GB, Santos dos RJ, Paim LL (2022) Determination of Cr(VI) in leather residues using graphite/paraffin composite electrodes modified with reduced graphene oxide nanosheets. Sci Total Environ 834:155318. https://doi.org/10.1016/j.scitotenv.2022.155318
Pinto L, Lemos SG (2013) Multivariate optimization of the voltammetric determination of Cd, Cu, Pb and Zn at bismuth film. application to analysis of biodiesel. Microchem J 110:417–424. https://doi.org/10.1016/j.microc.2013.05.012
Durai L, Badhulika S (2020) Simultaneous sensing of copper, lead, cadmium and mercury traces in human blood serum using orthorhombic phase aluminium ferrite. Mater Sci Eng C 112:110865. https://doi.org/10.1016/j.msec.2020.110865
İncebay H, Yazıcıgil Z (2017) Effect of different copper salts on the electrochemical determination of Cu(II) by the application of the graphene oxide-modified glassy carbon electrode. Surf Interfaces 9:160–166. https://doi.org/10.1016/j.surfin.2017.09.004
Tan X, Li Z, Wang X, Xu M, Yang M, Zhao J (2022) Simultaneous determination of cadmium(ii) lead(ii) copper(ii) and mercury(ii) using an electrode modified by N/S co-doped graphene. New J Chem 46(22):10618–10627. https://doi.org/10.1039/D2NJ01060G
Elgamouz A, Tijani N (2018) Dataset in the production of composite clay-zeolite membranes made from naturally occurring clay minerals. Data Brief 19:2267–2278. https://doi.org/10.1016/j.dib.2018.06.117
El-kordy A, Dehmani Y, Douma M, Bouazizi A (2022) Experimental study of phenol removal from aqueous solution by adsorption onto synthesized Faujasite-type Y zeolite. Desalin Water Treat 277:144–154. https://doi.org/10.5004/dwt.2022.28958
Lynch JLV, Baykara H, Cornejo M, Soriano G, Ulloa NA (2018) Preparation, characterization, and determination of mechanical and thermal stability of natural zeolite-based foamed geopolymers. Constr Build Mater 172:448–456. https://doi.org/10.1016/j.conbuildmat.2018.03.253
Khalil AKA, Elgamouz A, Atieh MA, Shanableh A, Laoui T (2023) Processing and characterization of UAE clay ceramic membranes for water treatment applications. Adv Sci Technol 129:3–11. https://doi.org/10.4028/p-2RWstS
Tahiri A (2023) Synthesis of X-type zeolite and their influence on the filtration of dye effluents. Desalin Water Treat 284:229–239. https://doi.org/10.5004/dwt.2023.29223
Ma Z (2022) Sn auxiliary agent promoting dispersion of Pt atoms over CeO2 catalysts and DFT calculation. J Alloy Compd 929:167214. https://doi.org/10.1016/j.jallcom.2022.167214
Ray JR, Wu X, Neil CW, Jung H, Li Z, Jun YS (2019) Redox chemistry of CeO2 nanoparticles in aquatic systems containing Cr(VI)(aq) and Fe2+ ions. Environ Sci. https://doi.org/10.1039/C9EN00201D
Melchionna M, Bevilacqua M, Fornasiero P (2020) The electrifying effects of carbon-CeO2 interfaces in (electro) catalysis. Mater Today Adv 6:100050. https://doi.org/10.1016/j.mtadv.2019.100050
Lustemberg PG, Mao Z, Irigoyen B, Ver Ganduglia-pirovano M, Campbell CT (2021) Nature of the active sites on Ni/CeO2 catalysts for methane conversions. ACS Catal. https://doi.org/10.1021/acscatal.1c02154
Amanulla AM (2021) Selectivity, stability and reproducibility effect of CeM-CeO2 modified PIGE electrode for photoelectrochemical behaviour of energy application. Surf Interfaces 22:100835. https://doi.org/10.1016/j.surfin.2020.100835
Arif M (2015) High-performance porous carbon/CeO2 nanoparticles hybrid super-capacitors for energy storage. Smart Mater Nondestr Eval Energy Syst 9439:1–8. https://doi.org/10.1117/12.2084267
Kowsuki K, Nirmala R, Ra Y, Navamathavan R (2023) Results in Chemistry Recent advances in cerium oxide-based nanocomposites in synthesis, characterization, and energy storage applications: a comprehensive review. Results Chem 5:100877. https://doi.org/10.1016/j.rechem.2023.100877
Asanova T (2019) Cerium(III) nitrate derived CeO2 support stabilising PtOx active species for room temperature CO oxidation. Chemcatchem. https://doi.org/10.1002/cctc.201902146
Nguyen PKQ, Lunsford SK (2013) Square wave anodic stripping voltammetric analysis of lead and cadmium utilizing titanium dioxide/zirconium dioxide carbon paste composite electrode. J Electroanal Chem 711:45–52. https://doi.org/10.1016/j.jelechem.2013.10.021
Wang N, Huang S, Cai H, Lin X, Mei R, Wang N (2023) In-situ synthesis of bi-metallic metal organic framework/polyaniline nanocomposites as ultrasensitive and selective electrodes for electrochemical detection of heavy metal ions. Microchem J 193:109185. https://doi.org/10.1016/j.microc.2023.109185
Chailapakul O, Korsrisakul S, Siangproh W, Grudpan K (2008) Fast and simultaneous detection of heavy metals using a simple and reliable microchip-electrochemistry route: an alternative approach to food analysis. Talanta 74:683–689. https://doi.org/10.1016/j.talanta.2007.06.034
Muralikrishna S, Sureshkumar K, Varley TS, Nagaraju DH, Ramakrishnappa T (2014) Analytical Methods In situ reduction and functionalization of graphene oxide with L-cysteine for simultaneous electrochemical determination of cadmium (II). Anal Methods 6:8698–8705. https://doi.org/10.1039/C4AY01945H
Funding
This research was funded by the Research Institute of Science and Engineering (RISE), Targeted research grant: Ref. V.C.R.G./R. 447/2022, and College of Graduate Studies, Graduate students support provided to Student ID: U19103583, dated 16/12/2021, University of Sharjah, Sharjah, United Arab Emirates. University of Sharjah, Sharjah, United Arab Emirates.
Author information
Authors and Affiliations
Contributions
Authors contribution Abdelaziz Elgamouz, Taher Laoui and Abdel-Nasser Kawde, 1.Conceptualization 2.Data curation 3.Formal analysis 4.Funding acquisition 5.Investigation 6.Methodology 7.Project administration 8.Resources 9.Supervision 10.Validation 11.Visualization 12.Writing—original draft 13.Writing—review & editing Sondos Sadek, Abderrazek El-Kordy, Abdelrahman K. A. Khalil 1.Formal analysis 2.Investigation 3.Methodology 4.Validation 5.Visualization 6.Writing—original draft 7.Writing—review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sadek, S., El-Kordy, A., Khalil, A.K.A. et al. Exploring Ce3+ adsorption on clay minerals to propel the frontier of heavy metal sensing with clay-CeO2 modified carbon paste electrode. J Appl Electrochem (2024). https://doi.org/10.1007/s10800-024-02125-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10800-024-02125-9