Abstract
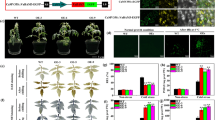
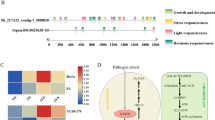
Ascorbate peroxidase (APX) is one of the most important antioxidant enzymes in the reactive oxygen metabolic pathway of plants. The role of APX under biotic and abiotic stress conditions has been explored, but the response pattern of APX under biotic stresses is relatively less known. In this study, seven CsAPXs gene family members were identified based on the sweet orange (Citrus sinensis) genome and subjected to evolutionary and structural analysis using bioinformatics software. The APX genes of lemon (ClAPXs) were cloned and showed a high conservation to CsAPXs by sequences alignment. In citrus yellow vein clearing virus (CYVCV)-infected Eureka lemons (C. limon) at 30th day post inoculation, APX activity and accumulation of hydrogen peroxide (H2O2) and malondialdehyde were measured to be 3.63, 2.29, and 1.73 times to that of the healthy control. The expression levels of 7 ClAPX genes in different periods of CYVCV-infected Eureka lemon were analyzed. Notably, ClAPX1, ClAPX5, and ClAPX7 showed higher expression levels compared to healthy plants, while ClAPX2, ClAPX3, and ClAPX4 showed lower expression levels. Functional identification of ClAPX1 in Nicotiana benthamiana showed that increasing the expression of ClAPX1 could significantly reduce the accumulation of H2O2, and it was verified that ClAPX1 is located in the plasma membrane of the cell. The present study provided information on the evolution and function of citrus APXs and revealed for the first time their response pattern to CYVCV infection.










Similar content being viewed by others
Data availability
Data available on request from the authors.
References
Ahlawat YS (1997) Viruses, greening bacterium and viroids associated with citrus (Citrus species) decline in India. Indian J Agric Sci 67:51–57
Asada K (1999) The water-water cycle in chloroplasts: scavenging of active oxygens and dissipation of excess photons. Annu Rev Plant Physiol Plant Mol Biol 50:601–639. https://doi.org/10.1146/annurev.arplant.50.1.601
Bailey TL, Williams N, Misleh C, Li WW (2006) MEME: discovering and analyzing DNA and protein sequence motifs. Nucleic Acids Res 34:W369–W373. https://doi.org/10.1093/nar/gkl198
Bethke PC, Jones RL (2001) Cell death of barley aleurone protoplasts is mediated by reactive oxygen species. Plant J 25:19–29. https://doi.org/10.1111/j.1365-313X.2001.00930.x
Biasini M, Bienert S, Waterhouse A, Arnold K, Studer G, Schmidt T, Kiefer F, Gallo Cassarino T, Bertoni M, Bordoli L, Schwede T (2014) SWISS-MODEL: modelling protein tertiary and quaternary structure using evolutionary information. Nucleic Acids Res 42:W252-258. https://doi.org/10.1093/nar/gku340
Catara A, Azzaro A, Davino M, Polizzi G (1992) Yellow vein clearing of lemon in Pakistan. In: Proceedings 12th conference IOCV, Riverside, pp 364–367
Chandrashekar S, Umesha S (2014) 2, 6-Dichloroisonicotinic acid enhances the expression of defense genes in tomato seedlings against Xanthomonas perforans. Physiol Mol Plant P 86:49–56. https://doi.org/10.1016/j.pmpp.2014.03.003
Chao J, Li Z, Sun Y, Aluko OO, Wu X, Wang Q, Liu G (2021) MG2C: a user-friendly online tool for drawing genetic maps. Mol Hortic 1:1–4. https://doi.org/10.1186/s43897-021-00020-x
Chen Z, Silva H, Klessig DF (1993) Active oxygen species in the induction of plant systemic acquired resistance by salicylic acid. Science 262:1883–1886. https://doi.org/10.1126/science.8266079
Chen CJ, Chen H, Zhang Y, Thomas HR, Frank MH, He YH, Xia R (2020) TBtools: An integrative toolkit developed for interactive analyses of big biological data. Mol Plant 13:1194–1202. https://doi.org/10.1016/j.molp.2020.06.009
Chew O, Whelan J, Millar AH (2003) Molecular definition of the ascorbate-glutathione cycle in Arabidopsis mitochondria reveals dual targeting of antioxidant defenses in plants. J Biol Chem 278:46869–46877. https://doi.org/10.1074/jbc.M307525200
Conrath U, Chen Z, Ricigliano JR, Klessig DF (1995) Two inducers of plant defense responses, 2,6-dichloroisonicotinec acid and salicylic acid, inhibit catalase activity in tobacco. Proc Natl Acad Sci 16:7143–7147. https://doi.org/10.1073/pnas.92.16.7143
Cui T, Bin Y, Yan J, Mei P, Li Z, Zhou C, Song Z (2018) Development of infectious cDNA clones of citrus yellow vein clearing virus using a novel and rapid strategy. Phytopathology 108:1212–1218. https://doi.org/10.1094/PHYTO-02-18-0029-R
Davletova S, Rizhsky L, Liang H, Shengqiang Z, Oliver DJ, Coutu J, Shulaev V, Schlauch K, Mittler R (2005) Cytosolic ascorbate peroxidase 1 is a central component of the reactive oxygen gene network of Arabidopsis. Plant Cell 17:268–281. https://doi.org/10.1105/tpc.104.026971
Deng Y Q (2017) Study on programmed cell death in Euneka lemon induced by Citrus yellow vein clearing virus. Dissertation, Southwest University
Faize M, Burgos L, Faize L, Petri C, Barba-Espin G, Díaz-Vivancos P, Clemente-Moreno MJ, Alburquerque N, Hernandez JA (2011) Modulation of tobacco bacterial disease resistance using cytosolic ascorbate peroxidase and Cu, Zn-superoxide dismutase. Plant Pathol 61:858–866. https://doi.org/10.1111/j.1365-3059.2011.02570.x
Felsenstein J (1981) Evolutionary trees from DNA-sequences—a maximum-likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/BF01734359
Fujiwara A, Togawa S, Hikawa T, Matsuura H, Masuta C, Inukai T (2016) Ascorbic acid accumulates as a defense response to Turnip mosaic virus in resistant Brassica rapa cultivars. J Exp Bot 67:4391–4402. https://doi.org/10.1093/jxb/erw223
Gasteiger E, Gattiker A, Hoogland C, Ivanyi I, Appel RD, Bairoch A (2003) ExPASy: the proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res 31:3784–3788. https://doi.org/10.1093/nar/gkg563
Geourjon C, Deleage G (1995) SOPMA: significant improvements in protein secondary structure prediction by consensus prediction from multiple alignments. Comput Appl Biosci 11:681–684. https://doi.org/10.1093/bioinformatics/11.6.681
Grimaldi V, Catara A (1996) Association of a filamentous virus with yellow vein clearing of lemon. In: International organization of citrus virologists conference proceedings (1957–2010)
Hammond-Kosack KE, Jones JDG (1996) Resistance gene-dependent plant defense responses. Plant Cell 8:1773–1791. https://doi.org/10.1105/tpc.8.10.1773
Horton P, Park KJ, Obayashi T, Fujita N, Harada H, Adams-Collier CJ, Nakai K (2007) WoLF PSORT: protein localization predictor. Nucleic Acids Res 35:W585–W587. https://doi.org/10.1093/nar/gkm259
Hu B, Jin J, Guo AY, Zhang H, Luo J, Gao G (2015) GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics 31:1296–1297. https://doi.org/10.1093/bioinformatics/btu817
Huang L, Jia J, Zhao X et al (2018) The ascorbate peroxidase APX1 is a direct target of a zinc finger transcription factor ZFP36 and a late embryogenesis abundant protein OsLE-A5 interacts with ZFP36 to co-regulate OsAPX1 in seed germination in rice. Biochem Biophys Res Commun 495:339–345. https://doi.org/10.1016/j.bbrc.2017.10.128
Huseynova IM, Mirzayeva SM, Sultanova NF, Aliyeva DR, Mustafayev NS, Aliyev JA (2017) Virus-induced changes in photosynthetic parameters and peroxidase isoenzyme contents in tomato (Solanum lycopersicum L.) plants. Photosynthetica 56:841–850. https://doi.org/10.1007/s11099-017-0737-9
Hyodo K, Hashimoto K, Kuchitsu K, Suzuki N, Okuno T (2017) Harnessing host ROS-generating machinery for the robust genome replication of a plant RNA virus. Proc Natl Acad Sci 114:E1282–E1290. https://doi.org/10.1073/pnas.1610212114
Jespersen HM, Kjærsgård IVH, Østergaard L, Welinder KG (1997) From sequence analysis of three novel ascorbate peroxidases from Arabidopsis thalina to structure, function and evolution of seven types of ascorbate peroxidase. Biochem J 326:305–310. https://doi.org/10.1042/bj3260305
Jiao Z, Tian Y, Cao Y, Wang J, Zhan B, Zhao Z, Sun B, Guo C, Ma W, Liao Z, Zhang H, Zhou T, Xia Y, Fan Z (2021) A novel pathogenicity determinant hijacks maize catalas-e 1 to enhance viral multiplication and infection. New Phytol 230:1126–1141. https://doi.org/10.1111/nph.17206
Kwon SY, Jeong YJ, Lee HS, Kim JS, Cho KY, Allen DR, Kwak SS (2002) Enhanced tolerances of transgenic tobacco plants expressing both superoxide dismutase and ascorbate peroxidase in chloroplasts against methyl viologen-mediated oxidative stress. Plant Cell Environ 25:873–882. https://doi.org/10.1046/j.1365-3040.2002.00870.x
Lescot M, Déhais P, Thijs G, Marchal K, Moreau Y, Van de Peer Y et al (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res 30:325–327. https://doi.org/10.1093/nar/30.1.325
Liu C, Liu H, Hurst J, Timko MP, Zhou C (2020) Recent advances on Citrus yellow vein clearing virus in Citrus. Hortic Plant J 6:216–222. https://doi.org/10.1016/j.hpj.2020.05.001
Loconsole G, Önelge N, Potere O, Giampetruzzi A, Bozan O, Satar S, De Stradis A, Savino V, Yokomi R, Saponari M (2012) Identification and characterization of Citrus yellow vein clearing virus, a putative new member of the genus Mandarivirus. Phytopathology 102:1168–1175. https://doi.org/10.1094/PHYTO-06-12-0140-R
Mittler R, Feng X, Cohen M (1998) Post-transcriptional suppression of cytosolic ascorbate peroxidase expression during pathogen-induced programmed cell death in tobacco. Plant Cell 10:461–473. https://doi.org/10.1105/tpc.10.3.461
Mittler R, Vanderauwera S, Gollery M, Breusegem FV (2004) Reactive oxygen gene network o-f plants. Trends Plant Sci 9:490–498. https://doi.org/10.1007/978-3-642-00390-5_10
Najami N, Janda T, Barriah W, Kayam G, Tal M, Guy M, Volokita M (2008) Ascorbate peroxidase gene family in tomato: its identification and characterization. Mol Genet Genomi-Cs 279:171–182. https://doi.org/10.1007/s00438-007-0305-2
Örvar BL, Ellis BE (1997) Transgenic tobacco plants expressing antisense RNA for cytosolic ascorbate peroxidase show increased susceptibility to ozone injury. Plant J 11:1297–1305. https://doi.org/10.1046/j.1365-313X.1997.11061297.x
Pérez-Clemente RM, Montoliu A, Vives V, López-Climent MF, Gómez-Cadenas A (2015) Photosynthetic and antioxidant responses of Mexican lime (Citrus aurantifolia) plants to Citrus tristeza virus infection. Plant Pathol 64:16–24. https://doi.org/10.1111/ppa.12241
Shang J, Zhang L, Jia Q, Tang Z, Yuan S, Yang H, Zhang M, Huang Y (2019) Early ROS accumulation in chloroplasts of Nicotiana glutinosa infected by Cucumber mosaic virus. Int J Agric Biol 21:149–154. https://doi.org/10.17957/IJAB/15.0875
Shigeoka S, Ishikawa T, Tamoi M, Miyagawa Y, Takeda T, Yabuta Y, Yoshimura K (2002) Regulation and function of ascorbate peroxidase isoenzymes. J Exp Bot 53:1305–1319. https://doi.org/10.1093/jexbot/53.372.1305
Swift ML (1997) GraphPad prism, data analysis, and scientific graphing. J Chem Inf Comp Sci 37:411–412. https://doi.org/10.1021/ci960402j
Tamura K, Stecher G, Kumar S (2021) MEGA11 molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
Teixeira FK, Menezes-Benavente L, Galvão VC, Margis R, Margis-Pinheiro M (2006) Rice ascorbate peroxidase gene family encodes functionally diverse isoforms localized in different subcellular compartments. Planta 224:300–314. https://doi.org/10.1007/s00425-005-0214-8
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680. https://doi.org/10.1093/nar/22.22.4673
Vandesompele J, Preter KD, Pattyn F, Poppe B, Roy NV, Paepe AD, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:1–12. https://doi.org/10.1186/gb-2002-3-7-research0034
Wang X, Jiang Z, Yue N et al (2021) Barley stripe mosaic virus γb protein disrupts chloroplast antioxidant defenses to optimize viral replication. EMBO J 40:e107660. https://doi.org/10.15252/embj.2021107660
Wu J, Su S, Fu L, Zhang Y, Chai L, Yi H (2014) Selection of reliable reference genes for gene expression studies using quantitative real-time PCR in navel orange fruit development and pummelo floral organs. Sci Hortic Amsterdam 176:180–188. https://doi.org/10.1016/j.scienta.2014.06.040
Yang M, Li Z, Zhang K, Zhang X, Zhang Y, Wang X, Han C, Yu J, Xu K, Li D (2017) Barley stripe mosaic virus γb interacts with glycolate oxidase and inhibits peroxisomal ROS production to facilitate virus infection. Mol Plant 11:338–341. https://doi.org/10.1016/j.molp.2017.10.007
Yu ZY, Ge CH, Wang XL, Xu CX, Wang QH, Cai XF (2018) Identifcation and expression analysis of ascorbate peroxidase gene family in spinach. J Shanghai Norm Univ 47:664–672. https://doi.org/10.3969/J.ISSN.1000-5137.2018.06.005
Zhou Y, Chen HM, Cao MJ, Wang XF, Jin X, Liu KH, Zhou CY (2017) Occurrence, distribution, and molecular characterization of Citrus yellow vein clearing virus in China. Plant Dis 101:137–143. https://doi.org/10.1094/PDIS-05-16-0679-RE
Acknowledgements
This work was supported by Natural Science Foundation of Chongqing (CSTB2022NSCQ-MSX0752), the National Key R&D Program of China (Grant no. 2021YFD1400800) and Innovation Research 2035 Pilot Plan of Southwest University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Q., Song, C., Cao, P. et al. Identification of citrus APX gene family and their response to CYVCV infection. J Plant Res 136, 371–382 (2023). https://doi.org/10.1007/s10265-023-01447-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-023-01447-7