Abstract
Objective
This in situ study aimed to investigate the efficacy of CO2 laser at a 10.6-µm wavelength combined with 1.23% acidulated phosphate fluoride (APF) and fluoridated dentifrice with 1100 µg F/g (FD) to control enamel caries progression.
Materials and methods
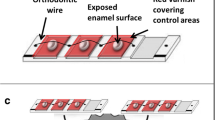
Sixteen volunteers wore palatal appliances containing eight demineralized enamel specimens for four 14-day phases under sucrose exposure. These specimens were submitted to CO2 laser irradiation and APF alone or combined with FD. Treatment groups were non-fluoridated dentifrice—NFD, NFD + CO2 laser, NFD + APF, NFD + CO2 laser + APF, FD, FD + CO2 laser, FD + APF, and FD + CO2 laser + APF. Mineral loss, calcium fluoride (CaF2), fluorapatite (FAp), and fluoride in the biofilm were analyzed by analysis of variance followed by the Student–Newman–Keuls test, p < 0.05.
Results
The highest mineral loss inhibition was noted when FD and CO2 laser irradiation were combined, which did not significantly differ from the FD + CO2 laser + APF group. The CaF2, FAp, and F in the biofilm were more pronounced when the FD and APF were combined. The CO2 laser irradiation promoted a slightly higher concentration of CaF2 in the enamel and F in the biofilm.
Conclusion
Although APF promotes the high formation of CaF2 and FAp, the combined use of FD with CO2 laser overcomes the APF effect in inhibiting the progression of artificial caries-like lesions in situ.
Clinical significance
Under the in situ design of this study, remineralization of white spot lesions was achieved through CO2 laser irradiation and daily use of fluoridated dentifrice. Future clinical trials are encouraged to substantiate this finding.




Similar content being viewed by others
References
Pitts NB, Zero DT, Marsh PD, Ekstrand K, Weintraub JA, Ramos-Gomez F, Tagami J, Twetman S, Tsakos G, Ismail A (2017) Dental caries. Nat Rev Dis Primers 3:17030. https://doi.org/10.1038/nrdp.2017.30
Soares RC, da Rosa SV, Moysés ST, Rocha JS, Bettega PVC, Werneck RI, Moysés SJ (2021) Methods for prevention of early childhood caries: overview of systematic reviews. Int J Paediatr Dent 31(3):394–421. https://doi.org/10.1111/ipd.12766
Jepsen S, Blanco J, Buchalla W, Carvalho JC, Dietrich T, Dörfer C, Eaton KA, Figuero E, Frencken JE, Graziani F, Higham SM, Kocher T, Maltz M, Ortiz-Vigon A, Schmoeckel J, Sculean A, Tenuta LM, van der Veen MH, Machiulskiene V (2017) Prevention and control of dental caries and periodontal diseases at individual and population level: consensus report of group 3 of joint EFP/ORCA workshop on the boundaries between caries and periodontal diseases. J Clin Periodontol 44(Suppl 18):S85–S93. https://doi.org/10.1111/jcpe.12687
Walsh T, Worthington HV, Glenny AM, Marinho VC, Jeroncic A (2019) Fluoride toothpastes of different concentrations for preventing dental caries. Cochrane Database Syst Rev 3(3):CD007868. https://doi.org/10.1002/14651858.CD007868.pub3.
Martignon S, Pitts NB, Goffin G, Mazevet M, Douglas GVA, Newton JT, Twetman S, Deery C, Doméjean S, Jablonski-Momeni A, Banerjee A, Kolker J, Ricketts D, Santamaria RM (2019) CariesCare practice guide: consensus on evidence into practice. Br Dent J 227(5):353–362. https://doi.org/10.1038/s41415-019-0678-8
Souza LFB, Fischer BV, Nora ÂD, Munareto BDS, Castro NC, Zenkner JEDA, Alves LS (2022) Efficacy of fluoride gel in arresting active non-cavitated caries lesions: a randomized clinical trial. Braz Oral Res 2(36):e062. https://doi.org/10.1590/1807-3107bor-2022.vol36.0062
Weyant RJ, Tracy SL, Anselmo TT, Beltrán-Aguilar ED, Donly KJ, Frese WA, Hujoel PP, Iafolla T, Kohn W, Kumar J, Levy SM, Tinanoff N, Wright JT, Zero D, Aravamudhan K, Frantsve-Hawley J, Meyer DM (2013) American dental association council on scientific affairs expert panel on topical fluoride caries preventive agents topical fluoride for caries prevention: executive summary of the updated clinical recommendations and supporting systematic review. J Am Dent Assoc 144(11):1279–1291. https://doi.org/10.14219/jada.archive.2013.0057
Marinho VC, Worthington HV, Walsh T, Chong LY (2015) Fluoride gels for preventing dental caries in children and adolescents. Cochrane Database Syst Rev. 15(6):CD002280. https://doi.org/10.1002/14651858.CD002280.pub2
Rošin-Grget K, Peroš K, Sutej I, Bašić K (2013) The cariostatic mechanisms of fluoride. Acta Med Acad 42(2):179–188. https://doi.org/10.5644/ama2006-124.85
Ogaard B (2001) CaF(2) formation: cariostatic properties and factors of enhancing the effect. Caries Res 35(Suppl 1):40–44. https://doi.org/10.1159/000049109
ten Cate JM, van Loveren C (1999) Fluoride mechanisms. Dent Clin North Am 43(4):713–742
Luk K, Zhao IS, Yu OY, Zhang J, Gutknecht N, Chu CH (2020) Effects of 10,600 nm carbon dioxide laser on remineralizing caries: a literature review. Photobiomodul Photomed Laser Surg 38(2):59–65. https://doi.org/10.1089/photob.2019.4690
Al-Maliky MA, Frentzen M, Meister J (2020) Laser-assisted prevention of enamel caries: a 10year review of the literature. Lasers Med Sci 35(1):13–30. https://doi.org/10.1007/s10103-019-02859-5
Rodrigues LK, Nobre dos Santos M, Pereira D, Assaf AV, Pardi V (2004) Carbon dioxide laser in dental caries prevention. J Dent 32(7):531–540. https://doi.org/10.1016/j.jdent.2004.04.004
Hsu CY, Jordan TH, Dederich DN, Wefel JS (2000) Effects of low-energy CO2 laser irradiation and the organic matrix on inhibition of enamel demineralization. J Dent Res 279(9):1725–1730. https://doi.org/10.1177/00220345000790091401
Poosti M, Ahrari F, Moosavi H, Najjaran H (2014) The effect of fractional CO2 laser irradiation on remineralization of enamel white spot lesions. Lasers Med Sci 29(4):1349–1355. https://doi.org/10.1007/s10103-013-1290-9
Souza-Gabriel AE, Turssi CP, Colucci V, Tenuta LM, Serra MC, Corona AS (2015) In situ study of the anticariogenic potential of fluoride varnish combined with CO2 laser on enamel. Arch Oral Biol 60(6):804–810. https://doi.org/10.1016/j.archoralbio.2015.01.016
Zancopé BR, Rodrigues LP, Parisotto TM, Steiner-Oliveira C, Rodrigues LK, Nobre-dos-Santos M (2016) CO2 laser irradiation enhances CaF2 formation and inhibits lesion progression on demineralized dental enamel-in vitro study. Lasers Med Sci 31(3):539–547. https://doi.org/10.1007/s10103016-1900-4
Loiola ABA, Aires CP, Curylofo-Zotti FA, Rodrigues Junior AL, Souza-Gabriel AE, Corona SAM (2019) The impact of CO2 laser treatment and acidulated phosphate fluoride on enamel demineralization and biofilm formation. J Lasers Med Sci 10(3):200–206. https://doi.org/10.15171/jlms.2019.32
Yu OY, Zhao IS, Mei ML, Lo EC, Chu CH (2017) A review of the common models used in mechanistic studies on demineralization-remineralization for cariology research. Dent J 5(2):20. https://doi.org/10.3390/dj5020020
Chow S, Wang H (2003) Sample size calculations in clinical research. Taylor & Francis, New York, pp 63–68
Rodrigues LK, Nobre Dos Santos M, Featherstone JD (2006) In situ mineral loss inhibition by CO2 laser and fluoride. J Dent Res 85(7):617–621. https://doi.org/10.1177/154405910608500707
Duggal MS, Toumba KJ, Amaechi BT, Kowash MB, Higham SM (2001) Enamel demineralization in situ with various frequencies of carbohydrate consumption with and without fluoride toothpaste. J Dent Res 80(8):1721–1724. https://doi.org/10.1177/00220345010800080801
PaesLeme AF, Tabchoury CP, Zero DT, Cury JA (2003) Effect of fluoridated dentifrice and acidulated phosphate fluoride application on early artificial carious lesions. Am J Dent 16(2):91–95
Zancopé BR, Dainezi VB, Nobre-Dos-Santos M, Duarte S Jr, Pardi V, Murata RM (2016) Effects of CO2 laser irradiation on matrix-rich biofilm development formation-an in vitro study. PeerJ 1(4):e2458. https://doi.org/10.7717/peerj.2458
Hara AT, Queiroz CS, PaesLeme AF, Serra MC, Cury JÁ (2003) Caries progression and inhibition in human and bovine root dentine in situ. Caries Res 37(5):339–344. https://doi.org/10.1159/000072165
Martínez-Mier EA, Cury JA, Heilman JR, Katz BP, Levy SM, Li Y, Maguire A, Margineda J, O’Mullane D, Phantumvanit P, Soto-Rojas AE, Stookey GK, Villa A, Wefel JS, Whelton H, Whitford GM, Zero DT, Zhang W, Zohouri V (2011) Development of gold standard ion-selective electrode-based methods for fluoride analysis. Caries Res 45(1):3–12. https://doi.org/10.1159/000321657
Lombardo G, Pagano S, Cianetti S, Capobianco B, Orso M, Negri P, Paglia M, Friuli S, Paglia L, Gatto R, Severino M (2019) Sub-ablative laser irradiation to prevent acid demineralisation of dental enamel. A systematic review of literature reporting in vitro studies. Eur J Paediatr Dent 20(4):295–301. https://doi.org/10.23804/ejpd.2019.20.04.07
Wu CC, Roan RT, Chen JH (2002) Sintering mechanism of the CaF2 on hydroxyapatite by a 10.6l microm CO2 laser. Lasers Surg Med 31(5):333–8. https://doi.org/10.1002/lsm.10124
Fowler BO, Kuroda S (1986) Changes in heated and in laser-irradiated human tooth enamel and their probable effects on solubility. Calcif Tissue Int 38(4):197–208. https://doi.org/10.1007/BF02556711
Newesely H (1977) High temperature behaviour of hydroxy- and fluorapatite. Crystalchemical implications of laser effects on dental enamel. J Oral Rehabil 4(1):97–104. https://doi.org/10.1111/j.13652842.1977.tb00971.x
Steiner-Oliveira C, Rodrigues LK, Soares LE, Martin AA, Zezell DM, Nobre-dos-Santos M (2006) Chemical, morphological and thermal effects of 10.6-microm CO2 laser on the inhibition of enamel demineralization. Dent Mater J 25(3):455–62. https://doi.org/10.4012/dmj.25.455
Ghadirian H, Geramy A, Shallal W, Heidari S, Noshiri N, Keshvad MA (2020) The Effect of remineralizing agents with/without CO2 laser irradiation on structural and mechanical properties of enamel and its shear bond strength to orthodontic brackets. J Lasers Med Sci 11(2):144-152. https://doi.org/10.34172/jlms.2020.25
Ahrari F, Mohammadipour HS, Hajimomenian L, Fallah-Rastegar A (2018) The effect of diode laser irradiation associated with photoabsorbing agents containing remineralizing materials on microhardness, morphology and chemical structure of early enamel caries. J Clin Exp Dent 10(10):e955–e962. https://doi.org/10.4317/jced.55059
Pai N, McIntyre J, Tadic N, Laparidis C (2007) Comparative uptake of fluoride ion into enamel from various topical fluorides in vitro. Aust Dent J 52(1):41–46. https://doi.org/10.1111/j.18347819.2007.tb00464.x
Dederich DN (1993) Laser/tissue interaction: what happens to laser light when it strikes tissue? J Am Dent Assoc 124(2):57–61. https://doi.org/10.14219/jada.archive.1993.0036
LeGeros RZ (1991) Calcium phosphates in oral biology and medicine. Monogr Oral Sci 15:108113
Chiang YC, Lee BS, Wang YL, Cheng YA, Chen YL, Shiau JS, Wang DM, Lin CP (2008) Microstructural changes of enamel, dentin-enamel junction, and dentin induced by irradiating outer enamel surfaces with CO2 laser. Lasers Med Sci 23(1):41–48. https://doi.org/10.1007/s10103-0070453-y
Tepper SA, Zehnder M, Pajarola GF, Schmidlin PR (2004) Increased fluoride uptake and acid resistance by CO2 laser-irradiation through topically applied fluoride on human enamel in vitro. J Dent 32(8):635–641. https://doi.org/10.1016/j.jdent.2004.06.010
Meurman JH, Hemmerlé J, Voegel JC, Rauhamaa-Mäkinen R, Luomanen M (1997) Transformation of hydroxyapatite to fluorapatite by irradiation with high-energy CO2 laser. Caries Res 31(5):397–400. https://doi.org/10.1159/000262425
Fox JL, Yu D, Otsuka M, Higuchi WI, Wong J, Powell GL (1992) Initial dissolution rate studies on dental enamel after CO2 laser irradiation. J Dent Res 71(7):1389–1398. https://doi.org/10.1177/00220345920710070701
Kang MK, Kim HE (2021) Remineralizing efficacy of fluoride in the presence of oral microcosm biofilms. J Dent 115:103848. https://doi.org/10.1016/j.jdent.2021.103848
de Sousa ET, Lima-Holanda AT, Nobre-Dos-Santos M (2020) Changes in the salivary electrolytic dynamic after sucrose exposure in children with early childhood caries. Sci Rep 10(1):4146. https://doi.org/10.1038/s41598-020-61128-6
Crescente CL, de Sousa ET, Lima-Holanda AT, Steiner-Oliveira C, Nobre-Dos-Santos M (2022) Biofilm accumulation and sucrose rinse modulate calcium and fluoride bioavailability in the saliva of children with early childhood caries. Sci Rep 12(1):10283. https://doi.org/10.1038/s41598-022-14583-2
Lippert F, Lynch RJ (2014) Comparison of Knoop and Vickers surface microhardness and transverse microradiography for the study of early caries lesion formation in human and bovine enamel. Arch Oral Biol 59(7):704–710. https://doi.org/10.1016/j.archoralbio.2014.04.005
Nobbs AH, Jenkinson HF, Jakubovics NS (2011) Stick to your gums: mechanisms of oral microbial adherence. J Dent Res 90(11):1271–1278. https://doi.org/10.1177/0022034511399096
Theuns HM, Driessens FCM, van Dijk JWE, van Groeneveld A (1986) Experimental evidence for a gradient in the solubility and in the rate of dissolution of human enamel. Caries Res 20:24–31. https://doi.org/10.1159/000260916
Funding
The São Paulo Research Foundation supported this work, grant number 2012/02885–7.
Author information
Authors and Affiliations
Contributions
Bruna Raquel Zancopé performed data collection and analysis and wrote the original draft of the manuscript; Lívia Pagotto Rodrigues and Lenita Marangoni Lopes conducted laboratory experiments, contributed to data interpretation, and provided critical revisions to the manuscript; Emerson Tavares de Sousa provided expertise in statistical analysis, contributed to data analysis and interpretation, and reviewed and edited the manuscript; Carolina Steiner-Oliveira1 and Lidiany Karla Azevedo-Rodrigues provided guidance on the conceptual framework, contributed to data interpretation, and critically revised the manuscript; Marinês Nobre-dos-Santos contributed to acquiring funding and study design, supervised the study, and substantially revised the manuscript. All authors have read and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Ethical approval
The Research Ethics Committee of Piracicaba Dental School—University of Campinas approved this study under Protocol No. 013/2012.
Consent to participate
Teeth donors and volunteers of the in situ phase signed a written informed consent to participate.
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zancopé, B., Rodrigues, L.P., Lopes, L.M. et al. CO2 laser irradiation combined with fluoridated dentifrice improved its protective effect on caries lesion progression regardless of the acidulated phosphate fluoride gel application: An in situ study. Clin Oral Invest 27, 7753–7763 (2023). https://doi.org/10.1007/s00784-023-05365-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-023-05365-9