Abstract
Objectives
The relative remission rates of tofacitinib, baricitinib, upadacitinib, and filgotinib compared with those of adalimumab were assessed in patients with rheumatoid arthritis (RA) who responded poorly to methotrexate (MTX).
Methods
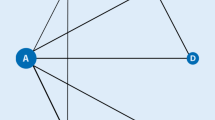
We performed a Bayesian network meta-analysis to combine direct and indirect evidence from randomized controlled trials (RCTs) to examine the Disease Activity Score in 28 joints with C‑reactive protein (DAS28-CRP), the Clinical Disease Activity Index (CDAI), the Simplified Disease Activity Index (SDAI), and the Boolean remission of tofacitinib, baricitinib, upadacitinib, filgotinib, and adalimumab in RA patients with inadequate responses to MTX.
Results
Four RCTs, comprising 3507 patients, met the inclusion criteria. The filgotinib 200 mg + MTX and upadacitinib 15 mg + MTX groups showed a significantly higher DAS28-CRP < 2.6 than adalimumab 40 mg + MTX. Upadacitinib 15 mg + MTX showed a significantly higher CDAI (≤ 2.8) than adalimumab 40 mg + MTX (odds ratio [OR]: 1.62; 95% credible interval [CrI]: 1.16–2.29). The ranking probability based on the surface under the cumulative ranking curve (SUCRA) indicated that upadacitinib 15 mg + MTX had the highest probability of being the best treatment as it achieved a CDAI ≤ 2.8, followed by filgotinib 200 mg + MTX, baricitinib 4 mg + MTX, tofacitinib 5 mg + MTX, and adalimumab 40 mg + MTX. The Boolean remission showed the same distribution pattern as that of the CDAI ≤ 2.8. Upadacitinib 15 mg + MTX showed a significantly higher SDAI ≤ 3.3 than adalimumab 40 mg + MTX (OR: 1.62; 95% CrI: 1.16–2.28). SUCRA ranking based on SDAI ≤ 3.3 indicated that upadacitinib 15 mg + MTX had the highest probability of being the best treatment for achieving an SDAI ≤ 3.3, followed by baricitinib 4 mg + MTX, filgotinib 200 mg + MTX, tofacitinib 5 mg + MTX, and adalimumab 40 mg + MTX.
Conclusions
In RA patients with an inadequate response to MTX, remission rates with JAK inhibitors were significantly higher; there is evidence for differences in efficacy regarding remission among the different JAK inhibitors.
Zusammenfassung
Ziel
Die relativen Remissionsraten von Tofacitinib, Baricitinib, Upadacitinib und Filgotinib im Vergleich zu denen von Adalimumab wurden bei Patienten mit rheumatoider Arthritis (RA) untersucht, die schlecht auf Methotrexat (MTX) ansprachen.
Methoden
Es wurde eine Bayes-Netzwerk-Metaanalyse durchgeführt, um direkte und indirekte Evidenz aus randomisierten kontrollierten Studien (RCT) zu kombinieren mit dem Ziel, den Disease Activity Score in 28 Gelenken mit C‑reaktivem Protein (DAS28-CRP), den Clinical Disease Activity Index (CDAI), den Simplified Disease Activity Index (SDAI) und die boolesche Remission von Tofacitinib, Baricitinib, Upadacitinib, Filgotinib und Adalimumab bei RA-Patienten mit unzureichendem Ansprechen auf MTX zu untersuchen.
Ergebnisse
Die Einschlusskriterien wurden von 4 RCT mit 3507 Patienten erfüllt. Die Gruppe mit Filgotinib 200 mg + MTX und die Gruppe mit Upadacitinib 15 mg + MTX wiesen einen signifikant höheren Anteil mit einem DAS28-CRP < 2,6 auf als Adalimumab 40 mg + MTX. Unter Upadacitinib 15 mg + MTX zeigte sich ein signifikant höherer Anteil mit einem CDAI ≤ 2,8 als unter Adalimumab 40 mg + MTX (Odds Ratio [OR]: 1,62; 95%-Kredibilitätsintervall [CrI]: 1,16–2,29). Die Ranglistenwahrscheinlichkeit, die auf der Oberfläche unter der kumulativen Rangkurve (SUCRA) basierte, zeigte, dass Upadacitinib 15 mg + MTX die höchste Wahrscheinlichkeit dafür aufwies, die beste Behandlung zu sein, da es einen CDAI ≤ 2,8 erzielte, es folgten Filgotinib 200 mg + MTX, Baricitinib 4 mg + MTX, Tofacitinib 5 mg + MTX und Adalimumab 40 mg + MTX. Die boolesche Remission wies das gleiche Verteilungsmuster auf wie bei einem CDAI ≤ 2,8. Unter Upadacitinib 15 mg + MTX zeigte sich ein signifikant höherer Anteil mit SDAI ≤ 3,3 als unter Adalimumab 40 mg + MTX (OR: 1,62; 95%-CrI: 1,16–2,28). Das SUCRA-Ranking auf der Basis eines SDAI ≤ 3,3 zeigte, dass Upadacitinib 15 mg + MTX die höchste Wahrscheinlichkeit dafür aufwies, die beste Behandlung zu sein, um einen SDAI ≤ 3,3 zu erzielen, es folgten Baricitinib 4 mg + MTX, Filgotinib 200 mg + MTX, Tofacitinib 5 mg + MTX und Adalimumab 40 mg + MTX.
Schlussfolgerung
Bei RA-Patienten mit unzureichendem Ansprechen auf MTX waren mit JAK-Inhibitoren signifikant höhere Remissionsraten zu erzielen; es gab Hinweise auf einen Unterschied hinsichtlich der Remissionswirksamkeit zwischen den verschiedenen JAK-Inhibitoren.




Similar content being viewed by others
References
Kim H, Sung Y‑K (2021) Epidemiology of rheumatoid arthritis in Korea. J Rheum Dis 28(2):60–67
Smolen JS, Aletaha D, Bijlsma JW, Breedveld FC, Boumpas D, Burmester G et al (2010) Treating rheumatoid arthritis to target: recommendations of an international task force. Ann Rheum Dis 69(4):631–637
Anderson J, Caplan L, Yazdany J, Robbins ML, Neogi T, Michaud K et al (2012) Rheumatoid arthritis disease activity measures: American college of rheumatology recommendations for use in clinical practice. Arthritis Care Res 64(5):640–647
Wells G, Becker J, Teng J, Dougados M, Schiff M, Smolen J et al (2009) Validation of the 28-joint disease activity score (DAS28) and European league against rheumatism response criteria based on C‑reactive protein against disease progression in patients with rheumatoid arthritis, and comparison with the DAS28 based on erythrocyte sedimentation rate. Ann Rheum Dis 68(6):954–960
Gwan Gyu S, Young Ho L (2016) Comparison of disease activity score 28 using C‑reactive protein and disease activity score 28 using erythrocyte sedimentation rate in assessing activity and treatment response in rheumatoid arthritis: a meta-analysisu. J Rheum Dis 23(4):241–249
Mack ME, Hsia E, Aletaha D (2017) Comparative assessment of the different American college of rheumatology/European league against rheumatism remission definitions for rheumatoid arthritis for their use as clinical trial end points. Arthritis Rheumatol 69(3):518–528
England BR, Tiong BK, Bergman MJ, Curtis JR, Kazi S, Mikuls TR et al (2019) 2019 update of the American college of rheumatology recommended rheumatoid arthritis disease activity measures. Arthritis Care Res 71(12):1540–1555
O’Dell JR, Mikuls TR, Taylor TH, Ahluwalia V, Brophy M, Warren SR et al (2013) Therapies for active rheumatoid arthritis after methotrexate failure. N Engl J Med 369(4):307–318
Choy E, Aletaha D, Behrens F, Finckh A, Gomez-Reino J, Gottenberg J‑E et al (2017) Monotherapy with biologic disease-modifying anti-rheumatic drugs in rheumatoid arthritis. Rheumatology 56(5):689–697
Ghoreschi K, Laurence A, O’Shea JJ (2009) Janus kinases in immune cell signaling. Immunol Rev 228(1):273–287
Changelian PS, Flanagan ME, Ball DJ, Kent CR, Magnuson KS, Martin WH et al (2003) Prevention of organ allograft rejection by a specific Janus kinase 3 inhibitor. Science 302(5646):875–878
Shi JG, Chen X, Lee F, Emm T, Scherle PA, Lo Y et al (2014) The pharmacokinetics, pharmacodynamics, and safety of baricitinib, an oral JAK 1/2 inhibitor, in healthy volunteers. J Clin Pharmacol 54(12):1354–1361
Ghoreschi K, Laurence A, O’Shea JJ (2009) Janus kinases in immune cell signaling. Immunol Rev 228(1):273–287
Nakase T, Wada H, Minamikawa K, Wakita Y, Shimura M, Hiyoyama K et al (1994) Increased activated protein C‑protein C inhibitor complex level in patients positive for lupus anticoagulant. Blood Coagul Fibrinolysis 5(2):173–177
Fleischmann R, Mysler E, Hall S, Kivitz AJ, Moots RJ, Luo Z et al (2017) Efficacy and safety of tofacitinib monotherapy, tofacitinib with methotrexate, and adalimumab with methotrexate in patients with rheumatoid arthritis (ORAL Strategy): a phase 3b/4, double-blind, head-to-head, randomised controlled trial. Lancet 390(10093):457–468
Taylor PC, Keystone EC, van der Heijde D, Weinblatt ME, Del Carmen Morales L, Reyes Gonzaga J et al (2017) Baricitinib versus placebo or adalimumab in rheumatoid arthritis. N Engl J Med 376(7):652–662
Fleischmann R, Pangan AL, Mysler E, Bessette L, Peterfy C, Durez P et al (eds) (2018) A phase 3, randomized, double-blind study comparing upadacitinib to placebo and to adalimumab, in patients with active rheumatoid arthritis with inadequate response to methotrexate. Wiley, Hoboken
Combe B, Kivitz A, Tanaka Y, van der Heijde D, Matzkies F, Bartok B et al (2019) Efficacy and safety of filgotinib for patients with rheumatoid arthritis with inadequate response to methotrexate: FINCH1 primary outcome results. BMJ Publishing Group Ltd, (LB0001)
Lee YH (2018) Association between the neutrophil-to-lymphocyte ratio, and platelet-to-lymphocyte ratio and rheumatoid arthritis and their correlations with the disease activity: a meta-analysis. J Rheum Dis 25(3):169–178
Lee YH (2015) Meta-analysis of genetic association studies. Ann Lab Med 35(3):283–287
Lee YH, Song GG (2017) Comparative efficacy and safety of secukinumab and adalimumab in patients with active ankylosing spondylitis: a Bayesian network meta-analysis of randomized controlled trials. J Rheum Dis 24(4):211–219
Hochberg MC, Chang RW, Dwosh I, Lindsey S, Pincus T, Wolfe F (1992) The American college of rheumatology 1991 revised criteria for the classification of global functional status in rheumatoid arthritis. Arthritis Rheum 35(5):498–502
Aletaha D, Landewe R, Karonitsch T, Bathon J, Boers M, Bombardier C et al (2008) Reporting disease activity in clinical trials of patients with rheumatoid arthritis: EULAR/ACR collaborative recommendations. Arthritis Care Res 59(10):1371–1377
Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJM, Gavaghan DJ et al (1996) Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 17(1):1–12
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(4):264–269
Brown S, Hutton B, Clifford T, Coyle D, Grima D, Wells G et al (2014) A Microsoft-Excel-based tool for running and critically appraising network meta-analyses—an overview and application of NetMetaXL. Syst Rev 3(1):110
Caldwell DM, Ades A, Higgins J (2005) Simultaneous comparison of multiple treatments: combining direct and indirect evidence. BMJ 331(7521):897
Salanti G, Ades A, Ioannidis JP (2011) Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol 64(2):163–171
Dias S, Welton NJ, Sutton AJ, Caldwell DM, Lu G, Ades A (2013) Evidence synthesis for decision making 4 inconsistency in networks of evidence based on randomized controlled trials. Med Decis Making 33(5):641–656
Higgins J, Jackson D, Barrett J, Lu G, Ades A, White I (2012) Consistency and inconsistency in network meta-analysis: concepts and models for multi-arm studies. Res Synth Methods 3(2):98–110
Valkenhoef G, Lu G, Brock B, Hillege H, Ades A, Welton NJ (2012) Automating network meta-analysis. Res Syn Meth 3(4):285–299
Lee YH (2018) An overview of meta-analysis for clinicians. Korean J Intern Med 33(2):277
Ajeganova S, Huizinga T (2017) Sustained remission in rheumatoid arthritis: latest evidence and clinical considerations. Ther Adv Musculoskelet Dis 9(10):249–262
Curtis JR, Chen L, Greenberg JD, Harrold L, Kilgore ML, Kremer JM et al (2017) The clinical status and economic savings associated with remission among patients with rheumatoid arthritis: leveraging linked registry and claims data for synergistic insights. Pharmacoepidemiol Drug Saf 26(3):310–319
Schett G, Tanaka Y, Isaacs JD (2021) Why remission is not enough: underlying disease mechanisms in RA that prevent cure. Nat Rev Rheumatol 17(3):135–144
Catalá-López F, Tobías A, Cameron C, Moher D, Hutton B (2014) Network meta-analysis for comparing treatment effects of multiple interventions: an introduction. Rheumatol Int 34(11):1489–1496
Song GG, Bae S‑C, Lee YH (2014) Association of the MTHFR C677T and A1298C polymorphisms with methotrexate toxicity in rheumatoid arthritis: a meta-analysis. Clin Rheumatol 33(12):1715–1724
Lee YH, Bae SC, Song GG (2011) The efficacy and safety of rituximab for the treatment of active rheumatoid arthritis: a systematic review and meta-analysis of randomized controlled trials. Rheumatol Int 31(11):1493–1499
Lee YH, Bae SC, Choi SJ, Ji JD, Song GG (2012) Associations between interleukin-10 polymorphisms and susceptibility to rheumatoid arthritis: a meta-analysis. Mol Biol Rep 39(1):81–87
Lee YH, Song GG (2020) Circulating interleukin-37 levels in rheumatoid arthritis and systemic lupus erythematosus and their correlations with disease activity: a meta-analysis. J Rheum Dis 27(3):152–158
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Y.H. Lee and G.G. Song declare that they have no financial or non-financial conflict of interest to declare.
For this article no studies with human participants or animals were performed by any of the authors. All studies performed were in accordance with the ethical standards indicated in each case.
Additional information
Redaktion
Ulf Müller-Ladner, Bad Nauheim
Uwe Lange, Bad Nauheim

Scan QR code & read article online
Rights and permissions
About this article
Cite this article
Lee, Y.H., Song, G.G. Relative remission rates of Janus kinase inhibitors in comparison with adalimumab in patients with active rheumatoid arthritis: a network meta-analysis. Z Rheumatol 83 (Suppl 1), 88–96 (2024). https://doi.org/10.1007/s00393-022-01165-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00393-022-01165-w