Abstract
The present work reported the synthesis of pH-responsive hybrid polymer network (HPN) of chitosan and gelatine by using glutaraldehyde as a cross-linker. Different reaction parameters like backbone ratio, amount of solvent, pH, the time, the temperature of reaction mixture and concentration of cross-linker were optimized with response surface methodology in order to maximize the percentage swelling. The maximum percentage swelling was obtained 530.48% under most optimized conditions of the ratio (1:1), pH (7) and cross-linker (0.241 mol/l). Poor loading of drug in traditional drug delivery is improved by incorporating preformed inclusion complex of chlorphenamine maleate (CPM) with β-cyclodextrin (1:1) under microwave conditions directly into the HPN matrix. Further, HPN matrix was used to investigate the in situ controlled release of CPM under different pH conditions at 37 °C. CPM release showed the best fit to the Peppas–Sahlin equation under all pH conditions at 37 °C. Thus, HPN prepared by using RSM design is a good device to deliver the CPM in a controlled manner.
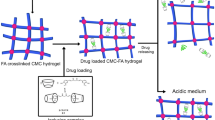
Graphical abstract








Similar content being viewed by others
Abbreviations
- CH:
-
Chitosan
- GEL:
-
Gelatin
- HPN:
-
Hybrid polymer network
- GLA:
-
Glutaraldehyde
- IC:
-
Inclusion complex
- β-CD:
-
Beta-cyclodextrin
- CPM:
-
Chlorphenamine maleate
- IPN:
-
Interpenetrating network
- IA:
-
In air
References
Sun Y, Huang J, Lin F, Lai J (1997) Composite poly(2-hydroxyethyl methacrylate) membranes as rate-controlling barriers for transdermal applications. Biomaterials 18:527–533
Kashyap N, Kumar N, Kumar MNVR (2005) Hydrogels for pharmaceutical and biomedical applications. Crit Rev Ther Drug Carrier Syst 22:107–149
Jagur-Grodzinski J (2010) Polymeric gels and hydrogels for biomedical and pharmaceutical applications. Polym Adv Technol 21:27–47. https://doi.org/10.1002/pat.1504
Peppas NA, Bures P, Leobandung W, Ichikawa H (2000) Hydrogels in pharmaceutical formulations. Eur J Pharm Biopharm 50:27–46
Bajpai AK, Shukla SK, Bhanu S, Kankane S (2008) Responsive polymers in controlled drug delivery. Prog Polym Sci 33:1088–1118. https://doi.org/10.1016/j.progpolymsci.2008.07.005
Martens P, Anseth KS (2000) Characterization of hydrogels formed from acrylate modified poly(vinyl alcohol) macromers. Polym (Guildf) 41:7715–7722. https://doi.org/10.1016/S0032-3861(00)00123-3
Xue W, Champ S, Huglin MB, Jones TGJ (2004) Rapid swelling and deswelling in cryogels of crosslinked poly(N-isopropylacrylamide-co-acrylic). Eur Polym J 40:703–712. https://doi.org/10.1016/j.eurpolymj.2003.10.021
Sukriti BS, Kaith R, Jindal M, Kumari M (2017) Kaur, Biodegradable-stimuli sensitive xanthan gum based hydrogel: evaluation of antibacterial activity and controlled agro-chemical release. React Funct Polym 120:1–13. https://doi.org/10.1016/j.reactfunctpolym.2017.08.012
Kaith BS, Jindal R, Sharma R (2015) Synthesis of a Gum rosin alcohol-poly(acrylamide) based adsorbent and its application in removal of malachite green dye from waste water. RSC Adv 5:43092–43104. https://doi.org/10.1039/C5RA04256A
Sionkowska A, Wisniewski M, Skopinska J, Poggi GF, Marsano E, Maxwell CA, Wess TJ (2006) Thermal and mechanical properties of UV irradiated collagen/chitosan thin films. Polym Degrad Stab 91:3026–3032. https://doi.org/10.1016/j.polymdegradstab.2006.08.009
Stockwell AF, Davis SS (1986) In vitro evaluation of alginate gel systems as sustained release drug delivery systems. J Control Release 3:167–175. https://doi.org/10.1016/0168-3659(86)90077-5
Kurita K (2006) Chitin and chitosan: functional biopolymers from marine crustaceans. Mar Biotechnol 8:203–226. https://doi.org/10.1007/s10126-005-0097-5
Blaney LM, Cinar S, SenGupta AK (2007) Hybrid anion exchanger for trace phosphate removal from water and wastewater. Water Res 41:1603–1613. https://doi.org/10.1016/j.watres.2007.01.008
Mao S, Sun W, Kissel T (2010) A drug delivery reviews: chitosan-based formulations for delivery of DNA and siRNA. Elsevier, Amsterdam
Fu X, Liu H, Liu Y, Liu Y (2013) Application of chitosan and its derivatives in analytical chemistry: a mini-review. J Carbohydr Chem 32:463–474. https://doi.org/10.1080/07328303.2013.863318
Das S, Subuddhi U (2013) Cyclodextrin mediated controlled release of naproxen from pH-sensitive chitosan/poly(vinyl alcohol) hydrogels for colon targeted delivery. Ind Eng Chem Res 52:14192–14200. https://doi.org/10.1021/ie402121f
Don T-M, King C-F, Chiu W-Y (2002) Synthesis and properties of chitosan-modified poly(vinyl acetate). J Appl Polym Sci 86:3057–3063. https://doi.org/10.1002/app.11329
Cui L, Jia J, Guo Y, Liu Y, Zhu P (2014) Preparation and characterization of IPN hydrogels composed of chitosan and gelatin cross-linked by genipin. Carbohydr Polym 99:31–38. https://doi.org/10.1016/j.carbpol.2013.08.048
Lohani A, Singh G, Bhattacharya SS, Verma A (2014) Interpenetrating polymer networks as innovative drug delivery systems. J Drug Deliv 2014:583612. https://doi.org/10.1155/2014/583612
Peng Z, Peng Z, Shen Y (2011) Fabrication and properties of gelatin/chitosan composite hydrogel. Polym Plast Technol Eng 50:1160–1164. https://doi.org/10.1080/03602559.2011.574670
Das S, Subuddhi U (2014) Exploring poly(vinyl alcohol) hydrogels containing drug-cyclodextrin complexes as controlled drug delivery systems. J Appl Polym Sci 131:1–13. https://doi.org/10.1002/app.40318
Kaith BS, Sharma R, Kalia S, Bhatti MS (2014) Response surface methodology and optimized synthesis of guar gum-based hydrogels with enhanced swelling capacity. RSC Adv 4:40339–40344. https://doi.org/10.1039/C4RA05300A
Ganji F, Vasheghani-Farahani S, Vasheghani-Farahani E (2010) Theoretical description of hydrogel swelling: a review, Iran. Polym J 19:375–398. https://doi.org/10.1007/s12303-009-0004-6
Sukriti JS, Sukriti P, Pruthi V, Chaddha AS, Bhatia J, Kaith BS (2017) RSM-CCD optimized adsorbent for the sequestration of carcinogenic rhodamine-B: kinetics and equilibrium studies. Mater Chem Phys 196:270–283. https://doi.org/10.1016/j.matchemphys.2017.04.042
Das S, Subuddhi U (2015) Studies on the complexation of diclofenac sodium with β-cyclodextrin: influence of method of preparation. J Mol Struct 1099:482–489. https://doi.org/10.1016/j.molstruc.2015.07.001
Sukriti JS, Chadha AS, Pruthi V, Anand P, Bhatia J, Kaith BS (2017) Sequestration of dyes from artificially prepared textile effluent using RSM-CCD optimized hybrid backbone based adsorbent-kinetic and equilibrium studies. J Environ Manage 190:176–187. https://doi.org/10.1016/j.jenvman.2016.12.065
Sukriti S, Sharma J, Pruthi V, Anand P, Singh Chaddha AP, Bhatia J, Kaith BS (2016) Surface response methodology–central composite design screening for the fabrication of a Gx-psy-g-polyacrylicacid adsorbent and sequestration of auramine-O dye from a textile effluent. RSC Adv 6:74300–74313. https://doi.org/10.1039/c6ra12715k
Saruchi S, Kaith BS, Jindal R, Kumar V, Bhatti MS (2014) Optimal response surface design of Gum tragacanth-based poly[(acrylic acid)-co-acrylamide] IPN hydrogel for the controlled release of the antihypertensive drug losartan potassium. RSC Adv 4:39822–39829. https://doi.org/10.1039/C4RA02803A
Bakravi A, Ahamadian Y, Hashemi H, Namazi H (2018) Synthesis of gelatin-based biodegradable hydrogel nanocomposite and their application as drug delivery agent. Adv Polym Technol. https://doi.org/10.1002/adv.21938
Yadollahi M, Gholamali I, Namazi H, Aghazadeh M (2015) Synthesis and characterization of antibacterial carboxymethylcellulose/CuO bio-nanocomposite hydrogels. Int J Biol Macromol 73:109–114. https://doi.org/10.1016/j.ijbiomac.2014.10.063
Sharma P, Jindal R, Maiti M, Jana AK (2016) Novel organic–inorganic composite material as a cation exchanger from a triterpenoidal system of dammar gum: synthesis, characterization and application, Iran. Polym J. https://doi.org/10.1007/s13726-016-0456-2
Rasoulzadeh M, Namazi H (2017) Carboxymethyl cellulose/graphene oxide bio-nanocomposite hydrogel beads as anticancer drug carrier agent. Carbohydr Polym 168:320–326. https://doi.org/10.1016/j.carbpol.2017.03.014
Bibby DC, Davies NM, Tucker IG (2000) Mechanisms by which cyclodextrins modify drug release from polymeric drug delivery systems. Int J Pharm 197:1–11. https://doi.org/10.1016/S0378-5173(00)00335-5
Kanjickal D, Lopina S, Evancho-Chapman MM, Schmidt S, Donovan D (2005) Improving delivery of hydrophobic drugs from hydrogels through cyclodextrins. J Biomed Mater Res Part A 74A:454–460. https://doi.org/10.1002/jbm.a.30374
Vimala K, Sivudu K, Mohan Y, Sreedhar B, Raju K (2009) Controlled silver nanoparticles synthesis in semi-hydrogel networks of poly(acrylamide) and carbohydrates: a rational methodology for antibacterial application. Elsevier, Amsterdam
Higuchi T (1963) Mechanism of sustained-action medication. Theoretical analysis of rate of release of solid drugs dispersed in solid matrices. J Pharm Sci 52:1145–1149. https://doi.org/10.1002/jps.2600521210
Ritger PL, Peppas NA (1987) A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. J Control Release 5:37–42. https://doi.org/10.1016/0168-3659(87)90035-6
Peppas NA, Sahlin JJ (1989) A simple equation for the description of solute release. III. Coupling of diffusion and relaxation. Int J Pharm 57:169–172. https://doi.org/10.1016/0378-5173(89)90306-2
Das S, Subuddhi U, Kulkarni RV, Boppana R, Mohan GK, Mutalik S, Kalyane NV, Ma L, Liu M, Liu H, Chen J, Gao C, Cui D, Vats A, Pathak K, Van den Mooter G, Chourasia MK, Jain SK (2014) Controlled delivery of dexamethasone to the intestine from poly(vinyl alcohol)–poly(acrylic acid) microspheres containing drug-cyclodextrin complexes: influence of method of preparation of inclusion complex. RSC Adv 4:24222. https://doi.org/10.1039/c4ra02736a
Yen M-T, Yang J-H, Mau J-L (2009) Physicochemical characterization of chitin and chitosan from crab shells. Carbohydr Polym 75:15–21. https://doi.org/10.1016/J.CARBPOL.2008.06.006
Namazi H, Rakhshaei R, Hamishehkar H, Kafil HS (2016) Antibiotic loaded carboxymethylcellulose/MCM-41 nanocomposite hydrogel films as potential wound dressing. Int J Biol Macromol 85:327–334. https://doi.org/10.1016/j.ijbiomac.2015.12.076
Rakhshaei R, Namazi H (2017) A potential bioactive wound dressing based on carboxymethyl cellulose/ZnO impregnated MCM-41 nanocomposite hydrogel. Mater Sci Eng C 73:456–464. https://doi.org/10.1016/j.msec.2016.12.097
Namazi H, Babazadeh M, Sarabi A, Entezami A (2001) Synthesis and hydrolysis of acrylic type polymers containing non steroidal anti-inflammatory drugs. J Polym Mater 18:301–311
Cárdenas A, Argüelles-Monal W, Goycoolea FM, Higuera-Ciapara I, Peniche C (2003) Diffusion through membranes of the polyelectrolyte complex of chitosan and alginate. Macromol Biosci 3:535–539. https://doi.org/10.1002/mabi.200300031
Logan BK (2009) Combined dextromethorphan and chlorpheniramine intoxication in impaired drivers. J Forensic Sci 54:1176–1180. https://doi.org/10.1111/j.1556-4029.2009.01127.x
Monte AA, Chuang R, Bodmer M (2010) Dextromethorphan, chlorphenamine and serotonin toxicity: case report and systematic literature review. Br J Clin Pharmacol 70:794–798. https://doi.org/10.1111/j.1365-2125.2010.03747.x
Szejtli J, Szente L (2005) Elimination of bitter, disgusting tastes of drugs and foods by cyclodextrins. Eur J Pharm Biopharm 61:115–125. https://doi.org/10.1016/j.ejpb.2005.05.006
Acknowledgements
One of the authors is highly grateful to MHRD for providing financial assistance to carry out research. The author is also thankful to Instrumentation Center, IIT Roorkee, Punjab University, Chandigarh, for different characterization of samples and DST-FIST for providing financial assistance for procurement of equipment like FTIR and UV–Vis spectrophotometer used in the characterization of the samples.
Funding
Funding was provided by MHRD [Grant No. 15520006 (Reg No.)].
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kaur, K., Jindal, R. Exploring RSM-CCD-optimized chitosan-/gelatin-based hybrid polymer network containing CPM–β-CD inclusion complexes as controlled drug delivery systems. Polym. Bull. 76, 3569–3592 (2019). https://doi.org/10.1007/s00289-018-2555-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-018-2555-z