Abstract
Objective
To elucidate the regulation, function of the chemokine CXC-motif ligand 12 (CXCL12) and its receptors (CXCR) 4 and 7 in prostate cancer tumor microenvironment.
Material
In-silico-analysis of expression in prostate cancer tissues. In-vitro comparison, testing of regulation in human prostate cancer cells LNCaP, DU145, and PC3.
Treatment
Dihydrotestosterone (DHT) treatments (0–10 nM) were for 0–48 h. The inflammatory agent Flagellin treatment (20 ng/ml) was for 2 h. Migration assays were performed for 24 h using 10 ng/ml CXCL12.
Methods
Real-time PCR, western analysis, and migration assays were used to determine mRNA, protein, and functional changes, respectively.
Results
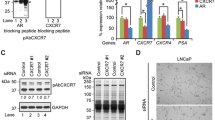
Malignant prostate cancer tissues exhibit higher CXCR4/7 mRNA ratio, and higher CXCR7 mRNA levels were detected in the androgen-responsive LNCaP cells. Putative androgen-responsive elements were identified in CXCR4, 7 gene, and exposure to DHT, flagellin increased CXCR4 mRNA but decreased CXCR7 mRNA levels in LNCaP cells. Androgen receptor siRNA significantly attenuated the effects of DHT on CXCR4, 7 mRNA in LNCaP cells. However, DHT and flagellin only decrease CXCR7 protein and additively increased migration of LNCaP cells towards CXCL12.
Conclusions
Down regulation of CXCR7 protein by DHT and flagellin increased migration, supporting CXCR7 as decoy receptor counteracting CXCL12/CXCR4-mediated migration in prostate cancer cells.





Similar content being viewed by others
References
Brawley OW. Prostate cancer epidemiology in the United States. World J Urol. 2012;30:195–200.
Lubeck DP, Grossfeld GD, Carroll PR. The effect of androgen deprivation therapy on health-related quality of life in men with prostate cancer. Urology. 2001;58:94–9.
Akashi T, Koizumi K, Tsuneyama K, Saiki I, Takano Y, Fuse H. Chemokine receptor CXCR4 expression and prognosis in patients with metastatic prostate cancer. Cancer Sci. 2008;99:539–42.
Chung LW, Baseman A, Assikis V, Zhau H. Molecular insights into prostate cancer progression: the missing link of tumor microenvironment. J Urol. 2005;173:10–20.
Wang JH, Shiozawa Y, Wang JC, et al. The role of CXCR7/RDC1 as a chemokine receptor for CXCL12/SDF-1 in prostate cancer. J Biol Chem. 2007;283:4283–94.
Chinni SR, Yamamoto H, Dong Z, Sabbota A, Bonfil RD, Cher ML. CXCL12/CXCR4 transactivates HER2 in lipid rafts of prostate cancer cells and promotes growth of metastatic deposits in bone. Mol Cancer Res. 2008;6:446–57.
Sánchez-Martín L, Estecha A, Samaniego R, Sánchez-Ramón S, Vega MÁ, Sánchez-Mateos P. The chemokine CXCL12 regulates monocyte-macrophage differentiation and RUNX3 expression. Blood. 2011;117:88–97.
Rigo A, Gottardi M, Zamò A, et al. Macrophages may promote cancer growth via a GM-CSF/HB-EGF paracrine loop that is enhanced by CXCL12. Mol Cancer. 2010;9:273.
Ray P, Stacer AC, Fenner J, et al. CXCL12-γ in primary tumors drives breast cancer metastasis. Oncogene. 2015;34:2043–51.
Sun YX, Wang JC, Shelburne CE, et al. Expression of CXCR4 and CXCL12 (SDF-1) in human prostate cancers (PCa) in vivo. J Cell Biochem. 2003;89:462–73.
Roy I, Zimmerman NP, Mackinnon AC, Tsai S, Evans DB, Dwinell MB. CXCL12 Chemokine expression suppresses human pancreatic cancer growth and metastasis. PLoS ONE. 2014;9:e90400.
Singh S, Singh UP, Grizzle WE, Lillard JW Jr. CXCL12–CXCR4 interactions modulate prostate cancer cell migration, metalloproteinase expression and invasion. Lab Invest. 2004;84:1666–76.
Chinni SR, Sivalogan S, Dong Z, et al. CXCL12/CXCR4 signaling activates Akt-1 and MMP-9 express ion in prostate cancer cells: the role of bone microenvironment-associated CXCL12. Prostate. 2006;66:32–48.
Burns JM, Summers BC, Wang Y, et al. A novel chemokine receptor for SDF-1 and I-TAC involved in cell survival, cell adhesion, and tumor development. J Exp Med. 2006;203:2201–13.
Zheng K, Li HY, Su XL, et al. Chemokine receptor CXCR7 regulates the invasion, angiogenesis and tumor growth of human hepatocellular carcinoma cells. J Exp Clin Canc Res. 2010;29:31.
Rajagopal S, Kim J, Ahn S, et al. β-arrestin- but not G protein-mediated signaling by the “decoy” receptor CXCR7. PNAS. 2010;107:2628–32.
Singh RK, Lokeshwar BL. The IL-8–regulated chemokine receptor CXCR7 stimulates EGFR signaling to promote prostate cancer growth. Cancer Res. 2011;71:3268–77.
Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454:436–44.
Yu L, Chen SW. Toll-like receptors expressed in tumor cells: targets for therapy. Cancer Immunol Immunother. 2008;57:1271–8.
Caruso C, Balistreri CR, Candore G, et al. Polymorphisms of pro-inflammatory genes and prostate cancer risk: a pharmacogenomic approach. Cancer Immunol Immunother. 2009;58:1919–33.
Galli R, Starace D, Busà R, et al. TLR stimulation of prostate tumor cells induces chemokine-mediated recruitment of specific immune cell types. J Immunol. 2010;184:6658–69.
Hajishengallis G. Immune evasion strategies of porphyromonas gingivalis. J Oral Biosci. 2011;53:233–40.
Rieber N, Brand A, Hector A, et al. Flagellin induces myeloid-derived suppressor cells: implications for Pseudomonas aeruginosa infection in cystic fibrosis lung disease. J Immunol. 2013;190:1276–84.
Gann PH, Hennekens CH, Ma J, Longcope C, Stampfer MJ. Study of sex hormone levels and risk of prostate cancer. J Natl Cancer Inst. 1996;88:1118–26.
Wang TTY, Hudson TS, Wang TC, et al. Differential effects of resveratrol on androgen-responsive LNCaP human prostate cancer cells in vitro and in vivo. Carcinogenesis. 2008;29:2001–100.
Kim EK, Kim YS, Milner JA, Wang TTY. Indole-3-carbinol and 3′,3′-diindolylmethane modulate androgen's effect on C-C Chemokine ligand 2 and monocyte attraction to prostate cancer cells. Cancer Prev Res. 2013;6:519–29.
Peng H, Erdmann N, Whitney N, et al. HIV-1-infected and/or immune activated macrophages regulate astrocyte SDF-1 production through IL-1β. GLIA. 2006;54:619–29.
Huang HQ, Fletcher A, Niu YG, Wang TTY, Yu LL. Characterization of lipopolysaccharide-stimulated cytokine expression in macrophages and monocytes. Inflamm Res. 2012;61:1329–38.
Wang TTY, Schoene NW, Kim YS, Mizuno CS, Rimando AM. Differential effects of resveratrol and its naturally occurring methylether analogs on cell cycle and apoptosis in human androgen-responsive LNCaP cancer cells. Mol Nutr Food Res. 2010;54:335–44.
Veldscholte J, Berrevoets CA, Ris-Stalpers C, et al. The androgen receptor in LNCaP cells contains a mutation in the ligand binding domain which affects steroid binding characteristics and response to antiandrogens. J Steroid Biochem Mol Biol. 1992;41:665–9.
Trasino SE, Harrison EH, Wang TTY. Androgen regulation of aldehyde dehydrogenase 1A3 (ALDH1A3) in the androgen-responsive human prostate cancer cell line LNCaP. Exp Biol Med. 2007;232:762–71.
Niu YJ, Altuwaijri S, Lai KP, et al. Androgen receptor is a tumor suppressor and proliferator in prostate cancer. Proc Natl Acad Sci USA. 2008;105:12182–7.
Li T, Li D, Sha JJ, Sun P, Huang YR. MicroRNA-21 directly targets MARCKS and promotes apoptosis resistance and invasion in prostate cancer cells. Biochem Biophys Res Commun. 2009;383:280–5.
Hoy JJ, Kallifatidis G, Smith DK, Lokeshwar BL. Inhibition of androgen receptor promotes CXC-chemokine receptor 7-mediated prostate cancer cell survival. Sci Rep. 2017;7(1):3058.
Wang JH, Loberg R, Taichman RS. The pivotal role of CXCL12 (SDF-1)/CXCR4 axis in bone metastasis. Cancer Metastasis Rev. 2006;25:573–87.
Uygur B, Wu WS. SLUG promotes prostate cancer cell migration and invasion via CXCR4/CXCL12 axis. Mol Cancer. 2011;10:139.
Lee C, Sutkowski DM, Sensibar JA, et al. Regulation of proliferation and production of prostate-specific antigen in androgen-sensitive prostatic cancer cells, LNCaP, by dihydrotestosterone. Endocrinol. 1995;136:796–803.
Fürstenberger G, Senn HJ. Insulin-like growth factors and cancer. Lancet. 2002;3:298–302.
Zhu ML, Kyprianou N. Role of androgens and the androgen receptor in epithelial-mesenchymal transition and invasion of prostate cancer cells. FASEB J. 2009;24:769–79.
Palapattu GS, Sutcliffe S, Bastian PJ, et al. Prostate carcinogenesis and inflammation: emerging insights. Carcinogenesis. 2005;26:1170–81.
Acknowledgements
The authors thank Dr. Norberta Schoene for her comments and editorial assistance. This study was supported by US Department of Agriculture appropriated fund #8040–51530-057-00D
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yu, L., Pham, Q., Yu, L.L. et al. Modulation of CXC-motif chemokine receptor 7, but not 4, expression is related to migration of the human prostate cancer cell LNCaP: regulation by androgen and inflammatory stimuli. Inflamm. Res. 69, 167–178 (2020). https://doi.org/10.1007/s00011-019-01305-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-019-01305-0