Early structural and functional plasticity alterations in a susceptibility period of DYT1 dystonia mouse striatum
Abstract
The onset of abnormal movements in DYT1 dystonia is between childhood and adolescence, although it is unclear why clinical manifestations appear during this developmental period. Plasticity at corticostriatal synapses is critically involved in motor memory. In the Tor1a+/Δgag DYT1 dystonia mouse model, long-term potentiation (LTP) appeared prematurely in a critical developmental window in striatal spiny neurons (SPNs), while long-term depression (LTD) was never recorded. Analysis of dendritic spines showed an increase of both spine width and mature mushroom spines in Tor1a+/Δgag neurons, paralleled by an enhanced AMPA receptor (AMPAR) accumulation. BDNF regulates AMPAR expression during development. Accordingly, both proBDNF and BDNF levels were significantly higher in Tor1a+/Δgag mice. Consistently, antagonism of BDNF rescued synaptic plasticity deficits and AMPA currents. Our findings demonstrate that early loss of functional and structural synaptic homeostasis represents a unique endophenotypic trait during striatal maturation, promoting the appearance of clinical manifestations in mutation carriers.
https://doi.org/10.7554/eLife.33331.001Introduction
Early-onset generalized torsion dystonia (DYT1) is an autosomal dominant movement disorder, commonly caused by a GAG base-pair deletion in the TOR1A gene coding for torsinA protein, without gross brain structural defects or other detectable neuropathology (Ozelius et al., 1997; Ledoux et al., 2013). Intriguingly, only 30–40% of DYT1 mutation carriers develop dystonia, typically in childhood-early adolescence (Bressman et al., 2000). However, what triggers the clinical onset of symptoms is currently unknown, although the presence of a critical developmental period of susceptibility is highly probable, since mutation carriers that do not develop symptoms in that time-window remain unaffected for their entire life (Pappas et al., 2014).
Plasticity changes include functional and structural synaptic specialization, leading to experience-dependent acquisition of motor skills. However, genetic or acquired alterations may lead to maladaptive plasticity changes. Accordingly, human studies indicate neural processing and synaptic plasticity alterations as major determinants in dystonia pathophysiology (Quartarone and Hallett, 2013). A significantly enhanced responsiveness to plasticity protocols has been reported in dystonic patients (Edwards et al., 2006; Weise et al., 2006; Quartarone et al., 2009). Moreover, patterns of impaired motor learning have been described even in clinically unaffected DYT1 mutation carriers (Ghilardi et al., 2003), further supporting the notion that aberrant plasticity represents a unique endophenotype in dystonia.
Of note, an impairment of striatal plasticity has been demonstrated in a number of different DYT1 models, including transgenic mice and rats overexpressing mutant torsinA (Martella et al., 2009; Grundmann et al., 2012), knock-in mice heterozygous for Δgag-torsinA (Dang et al., 2012; Martella et al., 2014; Rittiner et al., 2016), revealing an impressive similarity with studies of synaptic plasticity in human dystonia. Collectively, these observations support the hypothesis that DYT1 dystonia is a complex neurodevelopmental disorder of abnormal neurochemistry, wiring, and physiology (Goodchild et al., 2013; Pappas et al., 2014).
However, these alterations were observed in adult rodents, and to date, a relationship between age and corticostriatal plasticity in dystonia is still lacking. Furthermore, the question as to whether functional and structural plasticity abnormalities occur early in life or later as adaptive changes remains unknown. We report structural and functional abnormalities occurring in a defined postnatal time-window in Tor1a+/Δgag mice, indicative of a ‘premature’ and abnormal functional and structural plasticity, which is paralleled by a time-dependent increase in both BDNF levels and AMPAR-mediated currents.
Our findings reveal molecular, functional and structural changes in DYT1 striatal spiny projection neurons (SPNs), emphasizing the link between abnormal plasticity and dystonia. Understanding the key stages at which synaptic circuits are affected could suggest new routes to prevent or treat the disorder.
Results
The critical period for symptom onset in DYT1 dystonia matches a time-window of postnatal life when motor memories are shaped by activity-dependent changes in the striatum. Thus, in order to characterize plasticity changes in the early adolescence, Tor1a+/Δgag mice were recorded from postnatal day P15 to P35, in good agreement with the approximate life phase equivalencies between humans and mice, predicting that ~4 weeks of mouse age correspond to ~14 years in humans (Flurkey et al., 2007).
Electrophysiological characterization of SPNs
Properties of adult Tor1a+/Δgag SPNs have been extensively characterized (Maltese et al., 2014; Martella et al., 2014). Here, we focused on intrinsic and synaptic properties of juvenile Tor1a+/Δgag neurons. SPNs recorded at P26 from both Tor1a+/+ and Tor1a+/Δgag mice did not display firing activity at rest and exhibited no significant differences in their intrinsic membrane properties (data not shown). Depolarizing and hyperpolarizing current steps caused tonic action potential discharge and strong inward membrane rectification (Figure 1A). Short ISI (25–50 ms) of paired synaptic stimulation induced PPF in both genotypes (Figure 1B; p<0.05). At longer ISI (100–1000 ms), PPF was not observed in juvenile Tor1a+/+ and Tor1a+/Δgag mice (Figure 1B; p>0.05). To explore potential differences in neurotransmitter release, we recorded spontaneous glutamate- and GABA-mediated currents in P26 SPNs from both Tor1a+/+ and Tor1a+/Δgag mice. Glutamatergic sEPSCs did not differ between genotypes (Figure 1C; p>0.05). However, we found a significant increase in the amplitude, but not in the frequency, of mEPSCs recorded from Tor1a+/Δgag mice compared to wild types (Figure 1D; p<0.05). Conversely, GABAergic sIPSCs were unchanged in Tor1a+/Δgag with respect to Tor1a+/+ littermates (Figure 1E; p>0.05). Also, mIPSCs were similar in both genotypes (Figure 1F; p>0.05).
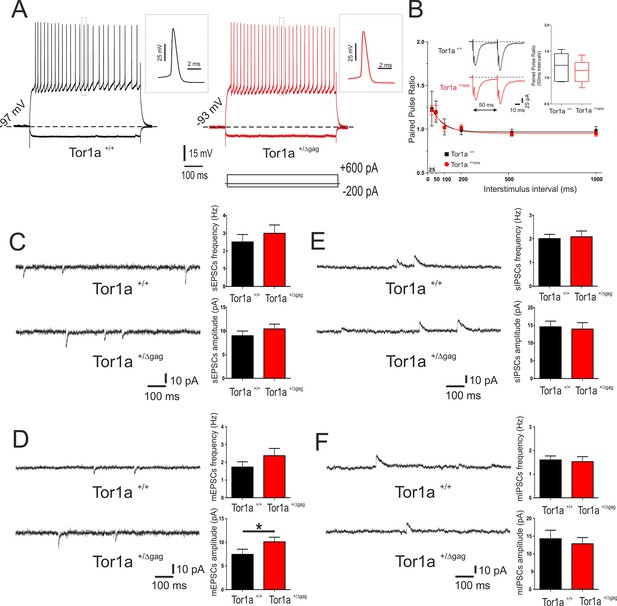
Electrophysiological and synaptic properties of striatal SPNs.
(A) Superimposed traces showing voltage responses to both depolarizing (+600 pA) and hyperpolarizing (−200 pA) current steps in SPNs recorded from P26 Tor1a+/+ (black) and Tor1a+/Δgag (red) mice. The insets display single action potentials (amplitude: Tor1a+/+69.62 ± 1.14 mV, N = 11, n = 11; Tor1a+/Δgag66.65 ± 1.68 mV, N = 8, n = 11; Student’s t test p>0.05). (B) Summary plot of paired-pulse ratio values showing similar facilitation in both genotypes. Each data point represents mean ± SEM. P26 Tor1a+/+ mice N = 3, 25 ms: 1.24 ± 0.20, n = 5; 50 ms: 1.20 ± 0.12, n = 5, Student’s t test p<0.05; P26 Tor1a+/Δgag mice N = 3, 25 ms: 1.22 ± 0.05, n = 5; 50 ms: 1.19 ± 0.08, n = 5; Student’s t test p<0.05. Insets represent sample traces showing facilitation at ISI = 50 ms in both genotypes. (C) Representative sEPSCs recordings in PTX from SPNs of P26 Tor1a+/+ and Tor1a+/Δgag mice. HP: −70 mV. The summary plots show no significant difference between genotypes in sEPSCs frequency and amplitude (Student’s t test p>0.05). (D) Representative whole-cell recordings in PTX plus TTX of mEPSC from P26 Tor1a+/+ and Tor1a+/Δgag SPNs. HP: −70 mV. Plots show a significant difference in the amplitude of mEPSCs recorded from Tor1a+/Δgag mice compared to wild-types (Tor1a+/+, 7.45 ± 1.09, N = 9, n = 9; Tor1a+/Δgag, 10.11 ± 0.97, N = 8, n = 9; Student’s t test *p<0.05). (E) Representative recordings in MK-801 and CNQX of sIPSCs from P26 Tor1a+/+ and Tor1a+/Δgag SPNs. HP:+10 mV. The summary plots show no significant difference in sIPSC frequency and amplitude (Student’s t test p>0.05). (F) Representative traces of mIPSCs recorded in MK-801, CNQX and TTX. HP:+10 mV. The summary plots show no difference in frequency and amplitude between genotypes (Student’s t test p>0.05). Data are presented as mean ± SEM.
-
Figure 1—source data 1
Electrophysiological and synaptic properties of striatal SPNs.
- https://doi.org/10.7554/eLife.33331.003
Premature expression of corticostriatal synaptic plasticity
We previously demonstrated a marked impairment of bidirectional synaptic plasticity in adult (P60-P75) Tor1a+/Δgag striatum (Martella et al., 2014). However, it remains unclear whether these patterns of abnormal plasticity are core pathologic features in an early developmental period, or occur later as maladaptive changes. Thus, we performed a detailed characterization of LTD and LTP from P15 to P35 in Tor1a+/+ and Tor1a+/Δgag mice. In Tor1a+/+ SPNs, HFS failed to induce LTD from P15 to P27 (Figure 2A; p>0.05). Conversely, the HFS protocol elicited a robust LTD from P28 to P35 (Figure 2A; 59.63 ± 2.63% of control; p<0.05). Surprisingly, in slices from Tor1a+/Δgag mice, HFS stimulation failed to cause a synaptic depression, independently from the postnatal day of recording (Figure 2B; p>0.05).
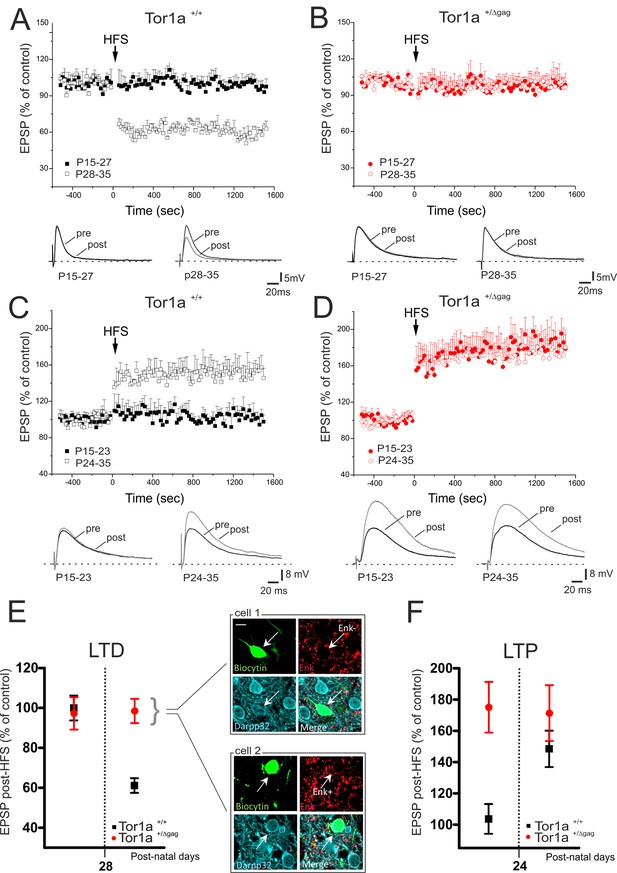
Altered developmental profile of corticostriatal long-term synaptic plasticity expression in Tor1a+/Δgag mice.
(A) (Top) Developmental time-course of LTD expression in Tor1a+/+ mice. HFS protocol (arrow) induces LTD in SPNs recorded from Tor1a+/+ mice after P28 (59.63 ± 2.63% of control; N = 8, n = 8; paired Student’s t test p<0.05), but not from P15 to P27 (99.46 ± 4.65, N = 9, n = 10; paired Student’s t test p>0.05). (Bottom) Representative EPSP traces recorded before (pre) and 20 min after (post) HFS protocol delivery. (B) (Top) In Tor1a+/Δgag mice, HFS protocol fails to induce any LTD, irrespective of the postnatal age (P15-27, 96.85 ± 11.35% of control; N = 8, n = 12; P28-35, 100.29 ± 4.16% of control, N = 8, n = 12; paired Student’s t test p>0.05). (Bottom) Representative traces of EPSPs recorded pre- and post-HFS. (C) (Top) Time-course of corticostriatal LTP expression during postnatal development in Tor1a+/+ mice. HFS of corticostriatal afferents (arrow) induces LTP expression in Tor1a+/+ mice after P24 (148.80 ± 15.39% of control; N = 6, n = 10; paired Student’s t test p<0.05), but not at P15-23 (104.68 ± 8.99% of control; N = 6, n = 10; paired Student’s t test p>0.05). (Bottom) Sample EPSPs recorded pre- and post-HFS protocol in Tor1a+/+ mice. (D) (Top) SPNs recorded from Tor1a+/Δgag mice exhibit a premature LTP (P15-23, 174.68 ± 22.59% of control; N = 6, n = 10; P24-35, 172.35 ± 11.06% of control; N = 9, n = 10; paired Student’s t test p<0.05). (Bottom) EPSP traces recorded pre- and post-LTP induction. (E) Mean plot comparing LTD expression at different postnatal days in Tor1a+/+ and Tor1a+/Δgag SPNs. (Inset) Confocal imaging of two SPNs recorded from Tor1a+/Δgag slices filled with biocytin (green) and immunolabelled for ENK (red) and DARPP-32 (cyano), marker of SPNs. Both ENK-positive and ENK-negative biocytin-labeled SPNs showed lack of LTD (scale bar: 10 µm). (F) Mean plot comparing LTP expression at different postnatal days in Tor1a+/+ and Tor1a+/Δgag SPNs. Values are presented as mean ± SEM.
-
Figure 2—source data 1
Altered developmental profile of corticostriatal long-term synaptic plasticity expression in Tor1a+/Δgag mice.
- https://doi.org/10.7554/eLife.33331.005
The LTP induction protocol failed to elicit a potentiation in Tor1a+/+ mice from P15 to P23 (Partridge et al., 2000) (Figure 2C; p>0.05), whereas a stable LTP occurred from P24 to P35 (Figure 2C; 148.80 ± 15.39% of control; p<0.05). Unexpectedly, in Tor1a+/Δgag SPNs LTP could be evoked as early as P15, revealing a premature onset, and showed a tendency to increase, compared to wild types (Figure 2D; Tor1a+/Δgag P15-23, 174.68 ± 22.59% of control; P24-35, 172.35 ± 11.06% of control; p<0.05).
The pattern of torsinA expression is common to all striatal DARPP-32-labeled neurons (Martella et al., 2009). To unmask potential differences between direct- and indirect-pathway SPNs, recording electrodes were filled with biocytin. Enkephalin staining revealed that neither ENK-positive nor ENK-negative SPNs exhibited LTD, ruling out a possible segregation to a specific population of SPNs (Figure 2E).
Collectively, these data demonstrate that LTD appeared at P28 in wild-type mice, whereas it could not be elicited during the entire postnatal period of observation in Tor1a+/Δgag mice (Figure 2E). Moreover, while in Tor1a+/+ mice LTP could not be evoked before P24, in SPNs from Tor1a+/Δgag LTP appeared prematurely at P15 (Figure 2F).
Increased AMPA receptor function and abundance at corticostriatal synapses during development
Changes in synaptic strength during learning and memory processes implicate an accurate regulation of AMPARs and NMDARs expression at postsynaptic membranes (Bassani et al., 2013; Czöndör and Thoumine, 2013). Thus, we performed an electrophysiological and biochemical characterization of AMPARs and NMDARs of SPNs in both Tor1a+/+ and Tor1a+/Δgag mice.
To investigate the relative abundance of postsynaptic AMPARs and NMDARs, NMDAR/AMPAR current ratios at corticostriatal synapses were evaluated in both juvenile (P26) and adult (P60) Tor1a+/+ and Tor1a+/Δgag SPNs (Figure 3A–B). We found that, at P26, the NMDAR/AMPAR ratio was significantly reduced in Tor1a+/Δgag SPNs compared to wild types (Figure 3A; p<0.05). Conversely, no significant differences were recorded in P60 SPNs of both genotypes (Figure 3B; p>0.05). A reduced NMDAR/AMPAR ratio could reflect an increase in AMPAR function or number, a decrease in NMDARs function, or even a combination of both. To detect possible differences in the composition of postsynaptic glutamate receptors in P26 SPNs, a IV relationship of AMPAR-EPSC was recorded (Figure 3C). Tor1a+/Δgag SPNs showed a significantly increased current at hyperpolarized voltage ranges (Figure 3C; 2-way ANOVA, p<0.01;HP= −70 mV). The GluA2 subunit reduces AMPAR permeability to Ca2+. Therefore, depending on the subunit composition, AMPAR-EPSC may show a linear or an inward-rectifying IV relationship (Cull-Candy et al., 2006). Thus, we measured the rectification index (RI), calculated as the ratio between the AMPAR-EPSC at −70 mV and at +40 mV (Isaac et al., 2007). We observed no significant difference in RI between genotypes (Figure 3C; p>0.05), suggesting that the enhanced AMPAR current involves an increased surface expression of AMPARs, rather than an altered receptor composition. Moreover, the AMPAR-EPSC IV relationship was also recorded in the presence of the selective antagonist of GluA2-lacking AMPARs, NASPM (100 μM). No significant difference in the RI of P26 SPNs was measured in the presence of NASPM (Figure 3D; p>0.05); yet, at hyperpolarized voltage ranges AMPAR-mediated current was still increased in Tor1a+/Δgag SPNs (Figure 3D; two-way ANOVA, p<0.01 at HP =−70 mV). These results further excluded possible alterations of AMPAR surface composition.
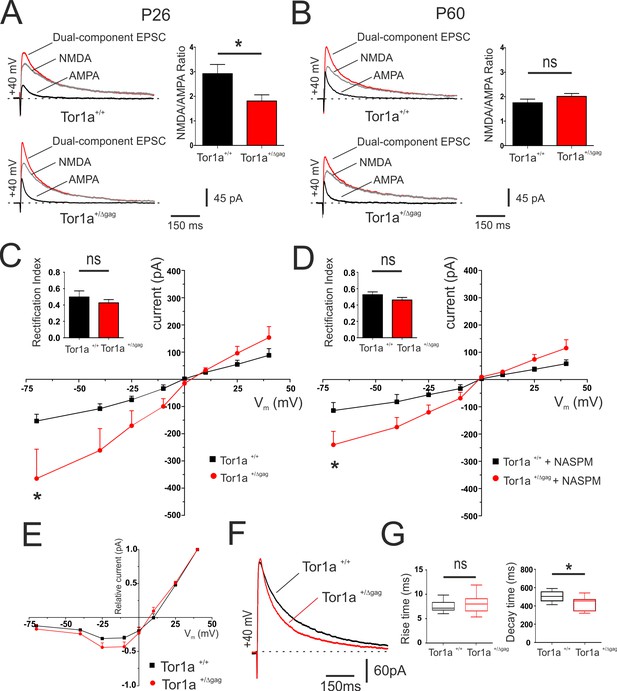
Electrophysiological characterization of AMPAR and NMDAR currents at corticostriatal synapses of SPNs in both Tor1a+/+ and Tor1a+/Δgag mice.
(A) (Left) Representative EPSCs traces recorded at HP=+40 mV from SPNs of juvenile Tor1a+/+ and Tor1a+/Δgag mice. The NMDAR antagonist MK-801 isolates the AMPAR-mediated EPSC component (black trace), while the NMDAR-EPSC (grey trace) is obtained by digital subtraction of the AMPAR EPSC from the dual-component EPSC (red). (Right) Summary plot of NMDA/AMPA current ratio calculated in SPNs from P26 Tor1a+/+ and Tor1a+/Δgag mice. A significant decrease of NMDA/AMPA ratio was detected in P26 Tor1a+/Δgag mice, compared to Tor1a+/+ (Tor1a+/+, 2.92 ± 0.38, N = 3, n = 8; Tor1a+/Δgag, 1.81 ± 0.25, N = 3, n = 6; Student’s t test, p<0.05). (B) (Left) Representative EPSCs traces recorded at HP =+40 mV from SPNs of adult Tor1a+/+ and Tor1a+/Δgag mice. (Right) Summary plot of NMDA/AMPA current ratio showing no significant difference between genotypes (Tor1a+/+, 1.75 ± 0.15, N = 3, n = 7; Tor1a+/Δgag, 2.01 ± 0.12, N = 3, n = 7; Student’s t test, p>0.05). (C) AMPAR-mediated currents recorded at different HP in P26 Tor1a+/+ and Tor1a+/Δgag SPNs. The IV relationship shows a significant increase in the current recorded at more hyperpolarized range from P26 Tor1a+/Δgag SPNs (HP=−70 mV: two-way ANOVA, *p<0.01). (Left) Summary plot of rectification index values of P26 Tor1a+/+ and Tor1a+/Δgag SPNs (Tor1a+/+, 0.50 ± 0.07, n = 7; Tor1a+/Δgag, 0.43 ± 0.04, n = 8; Student’s t test p>0.05). (D) AMPAR-mediated currents recorded in the presence of the GluA2-lacking AMPAR antagonist NASPM at P26. HP =−70 mV; to-way ANOVA, *p<0.01). (Left) Summary plots of the rectification index measured at P26 (Tor1a+/+, 0.53 ± 0.04, n = 5, N = 6; Tor1a+/Δgag, 0.46 ± 0.03, n = 7; Student’s t test, p>0.05). (E) Normalized IV relationships of NMDAR-mediated currents show no difference between genotypes at P26 (two-way ANOVA, p>0.05). (F) Representative NMDA-mediated EPSCs recorded at HP =+40 mV from P26 SPNs. (G) Summary plots display rise and decay time of NMDA-EPSCs recorded at HP =+40 mV in SPNs from P26 Tor1a+/+ and Tor1a+/Δgag mice (rise time: Tor1a+/+, 7.78 ± 0.42, n = 9; Tor1a+/Δgag, 9.23 ± 1.37, n = 7; Student’s t test p>0.05; decay time: Tor1a+/+, 502.50 ± 20.06, n = 9; Tor1a+/Δgag, 422.10 ± 30.15, n = 7, Student’s t test, *p<0.05). Values are presented as mean ± SEM.
-
Figure 3—source data 1
Electrophysiological characterization of AMPAR and NMDAR currents at corticostriatal synapses of SPNs in both Tor1a+/+ and Tor1a+/Δgag mice.
- https://doi.org/10.7554/eLife.33331.007
The normalized IV relationship of NMDAR-EPSCs showed the characteristic ‘J-shape’ (Mayer et al., 1984) in SPNs recorded at P26 from both genotypes (Figure 3E). No significant difference was found in the voltage-dependence of NMDARs (p>0.05). By analyzing the kinetics of the response at HP =+ 40 mV, we detected a significantly decreased decay time in Tor1a+/Δgag mice compared to controls (Figure 3F,G; p<0.05), despite a comparable rise time, suggesting a modification of NMDAR subunit composition (Paoletti et al., 2013). In particular, it is well-established that the decay time of NMDAR currents is correlated to the amount of GluN2-type subunits. GluN2A and GluN2B represent the most abundant NMDAR regulatory subunits expressed in SPNs (Chen and Reiner, 1996; Dunah and Standaert, 2003) and are characterized by a fast and slow decay time, respectively (Sanz-Clemente et al., 2013).
Taking into account all the above-described electrophysiological results, we evaluated the levels of AMPAR and NMDAR subunits into TIF fractions purified from striata of both juvenile (P26) and adult (P60) mice by means of WB analysis. We found a significant increase in the levels of both GluA1 and GluA2 AMPAR subunits in the postsynaptic compartment of P26 Tor1a+/Δgag mice compared to controls (Figure 4A,B; p<0.05), consistent with the observed reduction of the NMDA/AMPA ratio and the absence of any alteration of the RI (see Figure 3). Interestingly, we also found an increase of phosphorylation at GluA1-Ser845 (Figure 4A,B;, p<0.05), which is known to be correlated with LTP expression and to prevent endocytosis of GluA1-containing AMPARs (Oh et al., 2006; Bassani et al., 2013). Moreover, in agreement with the reduction of the NMDAR decay time, we observed an increase of GluN2A but not GluN2B subunit at postsynaptic sites of P26 Tor1a+/Δgag mice compared to Tor1a+/+ (Figure 4C,D; p<0.05). Finally, no modifications of PSD-95, the most abundant scaffolding protein at the excitatory synapse, was observed (Figure 4C,D). Notably, these alterations of AMPAR and NMDAR subunits were not present in SPNs from P60 Tor1a+/Δgag mice (Figure 4; p>0.05).
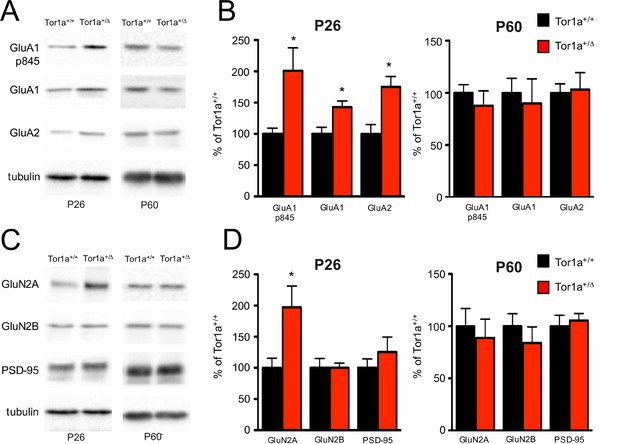
Molecular analysis of the SPNs postsynaptic compartment in P26 and P60 Tor1a+/Δgag compared to age-matched wild-type mice.
WB analyses were performed on the post-synaptic TIF fraction in a minimum of three different animals per genotype. (A) WB analysis for GluN2A, GluN2B, PSD-95 and tubulin in P26 (left panel) and P60 (right panel) Tor1a+/Δgag and age-matched Tor1a+/+ mice. (C) WB analysis for GluA1, GluA1p845, GluA2 and tubulin in P26 (left panel) and P60 (right panel) Tor1a+/Δgag and age-matched Tor1a+/+ mice. (B,D) The histogram shows the quantification of protein levels following normalization on tubulin (P26 Tor1a+/Δgag compared to Tor1a+/+, GluA1: 142.8 ± 9.8%, n = 5, p<0.05; GluA1-p845: 200.9 ± 36.6%, n = 5, p<0.05; GluA2: 175.1 ± 16.6%, n = 5, p<0.05; GluN2A: 197.3 ± 34.0%, n = 5, p<0.05; P60 Tor1a+/Δgag GluA1: 90.0 ± 23.4%, n = 5, p>0.05; GluA1-p845: 77.7 ± 14.2%, n = 5, p>0.05; GluA2: 103.2 ± 16.2%, n = 5, p>0.05; GluN2A: 88.8 ± 18.0%, n = 5,p>0.05). All values are mean ± SEM expressed as % of Tor1a+/+ mice.
Next, we performed a detailed evaluation of dendritic spine density and morphology in Tor1a+/Δgag SPNs, compared to age-matched Tor1a+/+ mice. P26 Tor1a+/Δgag SPNs (Figure 5A–D) exhibited a higher number of mushroom-type spines (Figure 5C; p<0.05) and, consequently, a concomitant overall increase of dendritic spine width compared to Tor1a+/+ mice (Figure 5B; p<0.05), thus suggesting an advanced stage of spine maturation, in agreement with the observed molecular GluN2A/GluN2B switch (see Figure 4). This event was associated, as expected, to an overall decrease of dendritic spine density (Figure 5A; p<0.05).
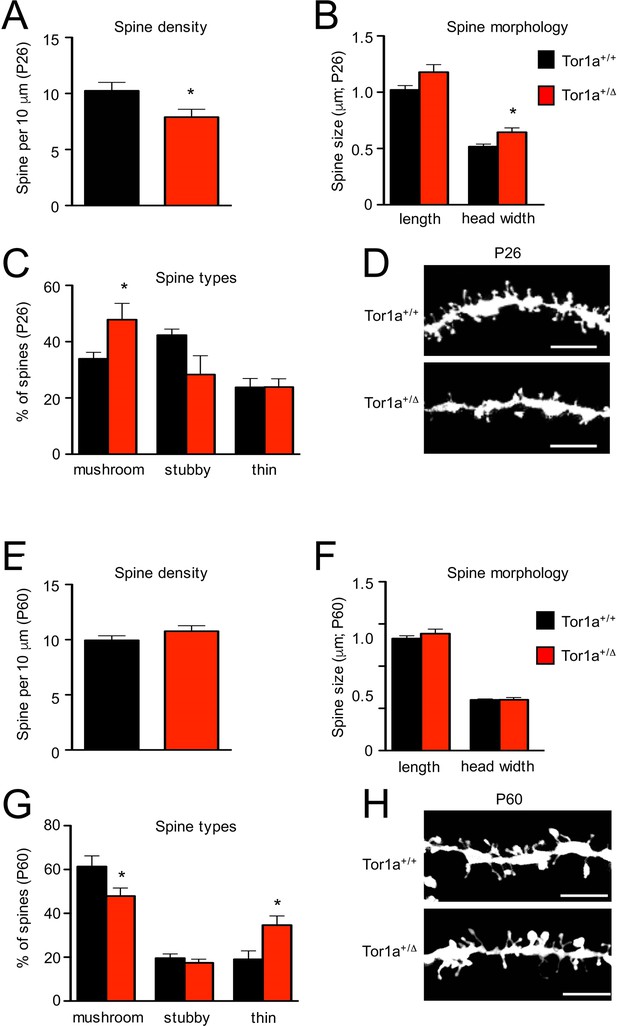
Analysis of dendritic spines morphology in P26 and P60 Tor1a+/Δgag compared to age-matched Tor1a+/+mice.
(A) Histogram representing dendritic spine density in P26 Tor1a+/Δgag and Tor1a+/+ mice (Tor1a+/+, 10.25 ± 0.75 spines/10 μm, n = 10; Tor1a+/Δgag, 7.89 ± 0.70 spines/10 μm, n = 10; unpaired Student’s t test *p<0.05). (B,C) Histograms showing the quantification of dendritic spine size (B, spine length and head width) and dendritic spine type (C, mushroom, stubby, thin) in P26 Tor1a+/Δgag compared to Tor1a+/+ mice (dendritic spine width Tor1a+/+, 0.51 ± 0.02 μm, n = 10; Tor1a+/ Δgag, 0.64 ± 0.04 μm, n = 10, unpaired Student’s t-test *p<0.05; mushroom-type spines Tor1a+/+, 33.92 ± 2.32%, n = 10; Tor1a+/Δgag, 47.81 ± 5.79%, n = 10, unpaired Student’s t-test *p<0.05). (D) Representative images show dendrites of P26 Tor1a+/Δgag and Tor1a+/+ mice. (E) Histogram representing dendritic spine density in P60 Tor1a+/Δgag and Tor1a+/+ mice (Tor1a+/+, 9.94 ± 0.41 spines/10 μm, n = 10; Tor1a+/ Δgag, 10.76 ± 0.50 spines/10 μm, n = 10; unpaired Student’s t-test p>0.05). (F,G) Histograms showing the quantification of dendritic spine size (F, spine length and head width) and dendritic spine type (G, mushroom, stubby, thin) in P60 Tor1a+/Δgag, compared to Tor1a+/+ mice (spine width Tor1a+/+, 0.600 ± 0.012 μm, n = 10; Tor1a+/Δgag, 0.602 ± 0.027 μm, n = 10; p>0.05; mushroom-type spines Tor1a+/+, 61.40 ± 4.81%, n = 10; Tor1a+/Δgag, 47.92 ± 3.67%, n = 10; *p<0.05; thin spines Tor1a+/+, 19.04 ± 3.85%, n = 10; Tor1a+/ Δgag, 34.64 ± 4.16%, n = 10; *p<0.05; unpaired Student’s t-test). (H) Representative images show dendrites of P60 Tor1a+/Δgag and Tor1a+/+ mice. Data were collected in a minimum of three different animals per genotype.
-
Figure 5—source data 1
Analysis of dendritic spines morphology in P26 Tor1a+/Δgag compared to age-matched Tor1a+/+ mice.
- https://doi.org/10.7554/eLife.33331.010
-
Figure 5—source data 2
Analysis of dendritic spines morphology in P60 Tor1a+/Δgag compared to age-matched Tor1a+/+ mice.
- https://doi.org/10.7554/eLife.33331.011
Conversely, P60 Tor1a+/Δgag mice (Figure 5E–H) showed a normalization of dendritic spine density (Figure 5E; p>0.05) and of spine width (Figure 5F; p>0.05) compared to Tor1a+/+ mice. Furthermore, with respect to P26, at P60 the number of mushrooms remained unchanged in Tor1a+/Δgag mice but increased in Tor1a+/+ (Figure 5G; p<0.05). Yet, at P60 Tor1a+/Δgag mice showed an increase of thin spines compared to Tor1a+/+ mice (Figure 5G; p<0.05).
Increased BDNF protein expression in Tor1a+/Δgag striatum at P26
Neurotrophic factors play a fundamental role in the development of SPNs and synaptic plasticity maturation (Altar et al., 1997; Rauskolb et al., 2010). Particularly, BDNF contributes to the developmental expression of AMPAR subunits at postsynaptic compartments (Jourdi et al., 2003; Jourdi and Kabbaj, 2013). The majority of BDNF, anterogradely transported to the striatum, originates from the cortex, where its expression begins in the first postnatal days (Baydyuk and Xu, 2014). We first performed a WB time-course analysis of BDNF protein level in P15, P26 and adult (P60-P75) striatum. BDNF expression profile showed a similar age-dependent time-course in both genotypes (Figure 6A,B; P15 vs P26: Tor1a+/+ p<0.05; Tor1a+/Δgagp<0.01). As indicated by the BDNF/proBDNF ratio, in line with previous evidence (Zermeño et al., 2009), BDNF was highly expressed at P15 in both strains. At P26 the signal decreased, and then reached intermediate values in adults (Figure 6A,B).
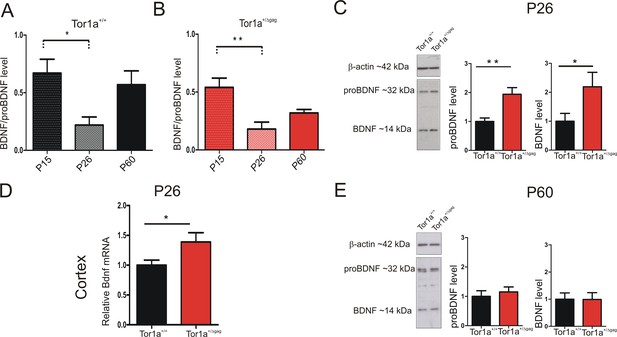
BDNF protein expression in the striatum of Tor1a+/+and Tor1a+/Δgag mice.
(A, B) Striatal BDNF protein expression in Tor1a+/+ and Tor1a+/Δgag mice at postnatal stages (P15, P26, P60). The graphs show the quantification of BDNF/proBDNF ratio at the various ages. Data are represented as mean ± SEM (Tor1a+/+ P15: 0.67 ± 0.12, N = 4; P26: 0.22 ± 0.08, N = 4; P60: 0.57 ± 0.14, N = 3; Tor1a+/Δgag P15: 0.54 ± 0.08, N = 4; P26: 0.18 ± 0.06, N = 4; P60: 0.32 ± 0.03, N = 4; one-way ANOVA, *p<0.05; **p<0.01). (C) (Left) Representative WB of proBDNF and BDNF protein levels relative to β-actin in striatal extracts (30 μg) derived from P26 Tor1a+/+ and Tor1a+/Δgag mice. (Right) The graphs show the quantitative analysis. The amount of proBDNF and BDNF was quantified relative to β-actin and normalized to wild-type mice. Data are represented as mean ± SEM (proBDNF Tor1a+/+ 1.00 ± 0.12, n = 10; Tor1a+/Δgag1.95 ± 0.29, n = 8; BDNF Tor1a+/+: 1.00 ± 0.28, n = 8, Tor1a+/Δgag2.19 ± 0.50, n = 8, Student’s t test: *p<0.05; **p<0.01). (D) Bdnf mRNA is upregulated in the cortex of Tor1a+/Δgag determined by qRT-PCR. The 2-ΔΔCt method was used to determine the relative expression, and all of the values are expressed relative to the levels of the wild-type mice as mean ± SEM (Tor1a+/+ 1.000 ± 0.084, n = 10; Tor1a+/Δgag1.399 ± 0.163, n = 8; Student’s t test: *p<0.05). (E) (Left) Representative Western blots of proBDNF and BDNF proteins relative to β-actin in striatal extracts (15 μg) derived from Tor1a+/+ and Tor1a+/Δgag adult mice. (Right) The graphs show the quantitative analysis. The amount of proBDNF and BDNF was quantified relative to β-actin and normalized to wild-type mice. Data are represented as mean ± SEM (proBDNF Tor1a+/+ 1.00 ± 0.19, n = 7, Tor1a+/Δgag1.15 ± 0.17, n = 7, p>0.05; BDNF Tor1a+/+: 1.00 ± 0.23 n = 7, Tor1a+/Δgag0.99 ± 0.25, n = 7, Student’s t test: p>0.05).
-
Figure 6—source data 1
BDNF protein expression in the striatum of Tor1a+/+ and Tor1a+/Δgag mice.
- https://doi.org/10.7554/eLife.33331.013
Next, we compared striatal proBDNF and BDNF protein levels between genotypes at P26. In line with previous evidence, proBDNF was detected as a double band at ~32 KDa, whereas mature BDNF as a single band at 14 KDa (Hartog et al., 2009; Koshimizu et al., 2009; Mandel et al., 2009; Tropea et al., 2011) (Figure 6C). Both proBDNF and BDNF levels were increased at P26 in Tor1a+/Δgag striatum (Figure 6C; proBDNF p<0.01, BDNF p<0.05). We therefore examined Bdnf mRNA expression in Tor1a+/Δgag cortex. Quantitative PCR revealed an increased Bdnf expression in Tor1a+/Δgag cortex as compared to Tor1a+/+ (Figure 6D; p<0.05). No significant difference between genotypes was measured in the proBDNF and BDNF striatal protein levels in adult mice (Figure 6E; p>0.05). Collectively, these data indicate an increase of BDNF level in P26 Tor1a+/Δgag striatum.
BDNF regulates surface AMPA receptor expression and synaptic plasticity
BDNF has been shown to contribute to LTP induction in normal mice (Jia et al., 2010). To test whether the increase in BDNF levels was involved in the abnormal regulation of AMPA currents and in the synaptic plasticity deficits, BDNF signalling was selectively blocked by the tropomyosin-related kinase B (TrkB) receptor competitive antagonist, ANA-12 (Cazorla et al., 2011). A single in vivo administration of ANA-12 (0.5 mg/kg, intraperitoneal, 4 hr before the experiment) failed to rescue synaptic plasticity deficits in young Tor1a+/Δgag mice (data not shown). However, repetitive treatment with ANA-12 (0.5 mg/kg, intraperitoneal, 12 hr and 4 hr before the experiment; Cazorla et al., 2011; Stragier et al., 2015), completely rescued corticostriatal LTD expression in P28-35 Tor1a+/Δgag mice (Figure 7A; p<0.05). Additionally, ANA-12 treatment reduced LTP amplitude in P24-35 Tor1a+/Δgag mice (Figure 7B; p>0.05). In vehicle-treated Tor1a+/+ and Tor1a+/Δgag mice, no significant change was observed (data not shown).
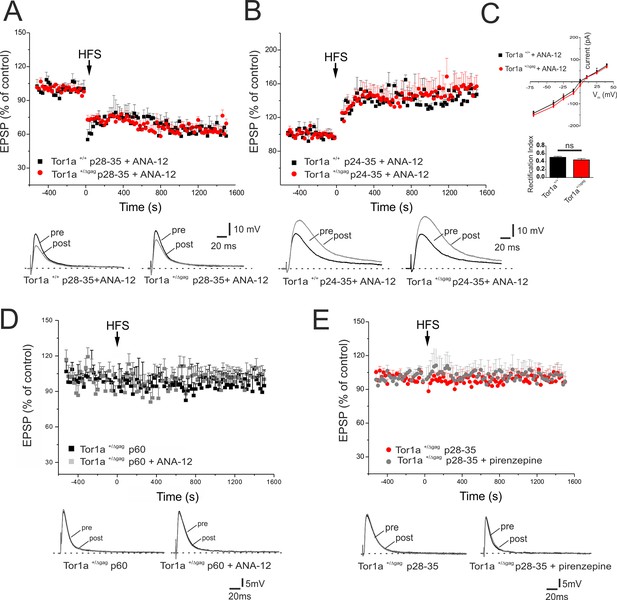
In vivo ANA-12 treatment rescues synaptic plasticity deficits in juvenile Tor1a+/Δgag mice.
(A) Time-course of corticostriatal LTD in juvenile Tor1a+/+ and Tor1a+/Δgag mice (P28-35): after in vivo treatment with the TrkB antagonist ANA-12, the HFS protocol (arrow) induces corticostriatal LTD expression in juvenile Tor1a+/Δgag mice (Tor1a+/+ P28-35, 65.31 ± 1.44% of control; N = 3, n = 12, p<0.05; Tor1a+/Δgag P28-35, 63.41 ± 4.39% of control; N = 3, n = 10; paired Student’s t test p<0.05). (Bottom) Representative EPSPs recorded before (pre) and 20 min after (post) HFS protocol. (B) Time-course of corticostriatal LTP after in vivo ANA-12 treatment: LTP displays a physiological amplitude in SPNs from in P24-35 Tor1a+/Δgag compared to wild-type littermates (Tor1a+/+ P24-35, 144.55 ± 2.67% of control; N = 3, n = 8; Tor1a+/Δgag P24-35, 148.11 ± 10.55% of control; N = 3, n = 9; Tor1a+/Δgag vs. Tor1a+/+ Student’s t test p>0.05). (Bottom) Sample traces recorded pre and post LTP induction. (C) AMPAR-mediated currents recorded from P26 SPNs at HP from −70 mV to + 40 mV after in vivo treatment of Tor1a+/+ and Tor1a+/Δgag mice with ANA-12. The treatment normalizes the current-voltage relationship in Tor1a+/Δgag neurons (HP=−70 mV: 2-way ANOVA p>0.05) and the rectification index (Tor1a+/+, 0.51 ± 0.03, N = 3, n = 3; Tor1a+/Δgag, 0.45 ± 0.04, N = 3, n = 5; Student’s t test p>0.05) (D) In vivo treatment with ANA-12 does not restore corticostriatal LTD in adult (P60) SPNs recorded from Tor1a+/Δgag mice (vehicle: 95.66 ± 9.09% of control, N = 3, n = 8; ANA-12: 98.75 ± 11% of control, N = 3, n = 4; paired Student’s t test p>0.05). (E) Slice pre-treatment with pirenzepine (100 nM) does not rescue LTD expression in P28-35 Tor1a+/Δgag SPNs (vehicle: 101.54 ± 1.07% of control, N = 3, n = 3; pirenzepine: 100.34 ± 8.96% of control; N = 3, n = 3; paired Student’s t test p>0.05). (Bottom) Superimposed traces of EPSPs recorded pre and 20 min post HFS delivery.
-
Figure 7—source data 1
In vivo ANA-12 treatment rescues synaptic plasticity deficits in juvenile Tor1a+/Δgag mice.
- https://doi.org/10.7554/eLife.33331.015
Likewise, in vivo treatment with ANA-12 totally normalized the IV curve of AMPAR-EPSC in P26 Tor1a+/Δgag mice (Figure 7C; p>0.05). Accordingly, also the RI displayed no significant difference between genotypes (Figure 7C; p>0.05). These findings suggest that increased BDNF levels are involved in the abnormal developmental expression of AMPARs on SPN postsynaptic membranes, leading to synaptic plasticity alterations in juvenile mice.
Finally, to demonstrate that BDNF alterations occur in a defined time-window, we tested the effect of ANA-12 on corticostriatal LTD expression in adult Tor1a+/Δgag mice. In vivo treatment with ANA-12 (0.5 mg/kg, intraperitoneal, two administrations at 12 hr and 4 hr before the experiment) failed to restore corticostriatal LTD in P60 Tor1a+/Δgag mice (Figure 7D; p>0.05) confirming that BDNF-dependent alterations are limited to a sensitive period.
Cholinergic transmission is not involved in the early phase of synaptic plasticity alterations
Previous work demonstrated a prominent involvement of cholinergic transmission in the impairment of striatal synaptic plasticity in adult Tor1a+/Δgag mice (Maltese et al., 2014). To verify whether plasticity alterations in juvenile Tor1a+/Δgag mice could also involve cholinergic signaling, slices were pretreated with the M1 muscarinic receptor antagonist, pirenzepine (100 nM, 20 min). Pirenzepine failed to rescue the expression of LTD in Tor1a+/Δgag mice from p28 to p35 (Figure 7E; p>0.05), indicating that distinct mechanisms underlie plasticity alterations at different developmental stages.
Discussion
The critical period for the onset of symptoms in DYT1 dystonia patients matches an early time-window of activity-dependent plastic changes in the striatum, which shape motor memory and learning processes during childhood and early adolescence.
Our systematic analysis of functional and structural synaptic plasticity in DYT1 dystonia demonstrates: (i) The existence of a critical period when SPNs exhibit premature LTP; (ii) A significant increase of AMPAR levels in the postsynaptic compartment which correlates with the reduction of NMDA/AMPA ratio, the increased amplitudes of postsynaptic currents and the rightward shift in the AMPA I-V curve observed in juvenile Tor1a+/Δgag mice; (iii) A BDNF time-dependent increase in expression profile, which parallels the alterations described; (iv) abnormal plasticity is associated with profound changes of dendritic spine density and morphology in juvenile Tor1a+/Δgag; (v) A rescue of the synaptic plasticity deficits is obtained by in vivo administration of a TrkB inhibitor.
It is currently unknown why penetrance is only 30% in DYT1 mutation carriers. One possibility is that at circuit level, motor system is already impaired early during development. The existence of a defined period in which neurons are particularly susceptible to experience-driven modifications is well-established, concurrently with structural modifications, and is currently believed to represent a nodal mechanism both in physiological and pathological conditions (Turrigiano and Nelson, 2004; Johnston, 2004; Meredith, 2015; Calabresi et al., 2016).
Our electrophysiological assessment of synaptic plasticity identifies a rather narrow time-window, between P15 and P26 when striatal SPNs exhibit a premature LTP, whereas LTD cannot be evoked. Although it has to be cautiously reminded that these alterations match those described in adult DYT1 striata (Martella et al., 2014), the time-course performed in the present study indicates their abnormal early appearance, in a developmental phase when normal striatal SPNs do not yet exhibit long-lasting synaptic changes. Of interest, the loss of LTD was observed in a similar time frame in a novel model with a rare missense variant in the Tor1a gene (Bhagat et al., 2016).
Moreover, we describe, along with electrophysiological deficits, molecular and structural changes at striatal synapses that appear to be limited to a specific time-window. Striatal LTP either in mature tissue preparation or in the developing striatum is dependent on the activation of NMDAR (Calabresi et al., 1992b; Partridge et al., 2000), whereas LTD depends on AMPAR (Calabresi et al., 1992a). Our electrophysiological and biochemical characterization demonstrates an increase in currents mediated by AMPAR, consistent with the increased amplitude of mEPSCs, and additionally, the NMDAR/AMPAR ratio was significantly reduced in SPNs from DYT1 mice. A major mechanism regulating synaptic strength involves the balance between synaptic insertion and removal of glutamate receptors into the postsynaptic membrane (Gong and De Camilli, 2008). Specifically, loss of homeostatic regulation of excitatory synapses in distinct neuronal subtypes involve postsynaptic changes in accumulation of AMPAR (Lissin et al., 1998; O'Brien et al., 1998). Accordingly, we observed a significant increase of both GluA1 and GluA2 subunits of AMPARs in the postsynaptic compartment of P26 Tor1a+/Δgag mice compared to controls, suggestive of an increased surface expression of AMPARs. Of interest, the significant increase of the phosphorylation of GluA1-Ser845, a well-established correlate of LTP (Oh et al., 2006; Bassani et al., 2013) is consistent with the abnormal LTP expression measured in DYT1 mice. Moreover, GluA1-Ser845 (Roche et al., 1996) plays a key role in the synaptic delivery of GluA1-containing AMPARs by LTP (Esteban et al., 2003; Bassani et al., 2013) and is involved in surface reinsertion/stabilization of AMPARs (Ehlers, 2000), thus providing a molecular mechanism for the observed increase of AMPARs at postsynaptic membranes in P26 Tor1a+/Δgag mice compared to controls. Thus, we hypothesize that the loss of LTD may be related to the aberrant composition of striatal AMPARs observed in mutant mice.
The identification of increased AMPAR subunit levels in the postsynaptic compartment offers new opportunities to identify potential regulators of AMPAR turnover. Neurotrophins have been implicated in glutamatergic synapse development and plasticity, suggesting a potential role in postsynaptic proteins distribution (Causing et al., 1997; McAllister et al., 1997; Kong et al., 2001). Previous work elucidated the role of BDNF in the regulation of AMPAR expression and function, including synaptic AMPAR subunit trafficking (Narisawa-Saito et al., 1999; Jourdi and Kabbaj, 2013). Indeed, BDNF treatment acutely controls both AMPAR subunits and their scaffolding proteins trafficking, thereby modifying the strength of synaptic activity (Minichiello et al., 1999; Mauceri et al., 2004). Remarkably, we observed an enhancement of pro-BDNF and BDNF protein level in P26 Tor1a+/Δgag mice, which appears critical for the onset of abnormal neurophysiological phenotype in DYT1 dystonia. Consistently, we obtained a functional rescue of synaptic plasticity and AMPA-mediated currents with the competitive antagonist of BDNF TrkB receptor ANA-12 (Cazorla et al., 2011).
Activity-dependent synaptic plasticity as well as composition and activity of NMDARs and AMPARs strictly govern modifications of dendritic spine morphology, leading to a long-lasting structural plasticity. Yet, BDNF also plays a major role in spine maturation in several brain regions, including the striatum (Baquet et al., 2004; Rauskolb et al., 2010). Thus, the abnormal increase in BDNF expression fits with the abnormalities in spine morphology we observed. In P26 Tor1a+/Δgag mice, we measured an increase in mushroom spines, suggestive of a ‘premature’ maturation process accompanied by an overall decrease in the density of dendritic spines. It is well-known that expression patterns of the GluN2 subunits of NMDARs at dendritic spines change during the first postnatal weeks. In particular, GluN2A expression increases from the second postnatal week to become widely expressed and abundant throughout the brain (Bellone and Nicoll, 2007; Gray et al., 2011). Yet, in agreement with the reduction of the NMDAR decay time, we found an increase of postsynaptic GluN2A in P26 Tor1a+/Δgag mice suggesting a ‘premature’ GluN2A/GluN2B switch, thus indicating the existence of a molecular and morphological early maturation of the excitatory synapse in this DYT1 model. Moreover, the existence of a close coordination between spine size and AMPAR levels at synaptic membranes has been previously reported (Kopec et al., 2007; Malinverno et al., 2010) and spine volume has been positively correlated with the strength of AMPAR-mediated synaptic transmission. Accordingly, in Tor1a+/Δgag mice we found a significant increase of spine head width, an increase in mushroom spines and a concomitant increase of both GluA1 and GluA2 subunits of AMPARs.
Most of the molecular and structural alterations described in juvenile DYT1 mice were not confirmed at our analyses performed in adult (P60) mice. Indeed, inhibition of BDNF with ANA-12 did not offset the plasticity deficits in adult mice. Additionally, the anticholinergic agent pirenzepine failed to rescue the plasticity deficits in juvenile animals, contrarily to what reported in adults (Dang et al., 2012; Martella et al., 2014), indicating that distinct mechanisms sustain the abnormal patterns of synaptic activity at different developmental ages. Future work is required to address the precise mechanisms governing this switch.
Collectively, we demonstrate that the rise of BDNF, in a restricted time-window, drives AMPA receptor composition changes and, consequently, structural modifications in spine morphology, resulting in the loss of homeostatic regulation of synaptic plasticity early in postnatal life.
Our hypothesis is also consistent with the clinical observation that the beneficial effects of Deep Brain Stimulation (DBS) in dystonic patients is more effective in young patients, as compared to patients implanted later in life (Isaias et al., 2008). Additionally, compared to the prompt efficacy observed in Parkinson’s disease patients, weeks are commonly required to obtain symptomatic relief following DBS, and improvements may continue to be manifest over time (Vercueil et al., 2001; Krauss, 2002; Vidailhet and Pollak, 2005). It is plausible that severity of abnormal plasticity is related to disease duration, thus justifying the longer time required to erase aberrant plasticity patterns.
In a therapeutic perspective, these sensitive periods might be considered as temporal windows of opportunity, during which specific molecular steps could be targeted to prevent aberrant plasticity to develop.
Materials and methods
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Mus musculus) | Tor1a | MGI:1353568 | Gene ID: 30931 | official full name: torsin family 1, member A (torsin A) |
| Strain, strain background (M. musculus) | C57BL/6J mice | Charles River | catalog number B6JSIFE10SZ - C57BL/6J SPF/VAF; RRID:IMSR_JAX:000664 | |
| Genetic reagent (M. musculus) | heterozygous knock-in Tor1a+/Δgag | Goodchild et al. (2005) | - | maintained on the C57BL/6J background |
| Antibody | monoclonal anti-PSD-95 | Neuromab | clone (k28/43) - catalog number 75–028; RRID:AB_2292909 | dilution 1:2000 in I-Block |
| Antibody | monoclonal anti-GluN2B | Neuromab | clone 59/20 - catalog number 75–097; RRID:AB_10673405 | dilution 1:1000 in I-Block |
| Antibody | polyclonal anti-GluA1 | Merck Millipore | catalog number AB1504; RRID:AB_2113602 | dilution 1:1000 in I-Block |
| Antibody | polyclonal anti-phospho-GluA1 (Ser845) | Merck Millipore | catalog number 04–1073; RRID:AB_1977219 | dilution 1:1000 in I-Block |
| Antibody | polyclonal anti-GluN2A | Sigma-Aldrich | catalog number M264 RRID:AB_260485 | dilution 1:1000 in I-Block |
| Antibody | monoclonal anti-GluA2 | Neuromab | clone L21/32 - catalog number 75–002; RRID:AB_2232661 | dilution 1:1000 in I-Block |
| Antibody | monoclonal anti-α-tubulin | Sigma-Aldrich | clone DM1A - catalog number T9026; RRID:AB_477593 | dilution 1:5000 in I-Block |
| Antibody | goat anti-DARPP-32 | R and D system | catalog number AF6259; RRID:AB_10641854 | dilution 1:500 in I-Block |
| Antibody | mouse anti-Enkephalin | Millipore | catalog number MAB350; RRID:AB_2268028 | dilution 1:1000 in I-Block |
| Antibody | mouse anti-β-actin | Sigma Aldrich | catalog number A5441; RRID:AB_476744 | dilution 1:20000 in I-Block |
| Commercial assay or kit | Clarity Western ECL Substrate | BioRad | - | reagent used to visualize protein bands with Chemidoc Imaging System |
| Commercial assay or kit | ECL reagent | GEHealthcare | catalog number GERPN2232 | reagent used to visualize protein bands with membranes were exposed to film |
| Commercial assay or kit | TRI-reagent | Sigma Aldrich | catalog number T9424 | reagent used to RNA extraction |
| Commercial assay or kit | DNAase I | Invitrogen | catalog number AMPD1-1KT | reagent used for elimination of DNA from RNA |
| Commercial assay or kit | Transcriptor First Strand cDNA Synthesis Kit | Roche | catalog number04379012001 | reagent used to reverse transcribe RNA |
| Commercial assay or kit | Extract-N-Amp™ Tissue PCR Kit | SIGMA | catalog number XNAT2 | genotyping primers UP- AGT CTG TGG CTG GCT CTC C; Low- CCT CAG GCTGCT CAC AAC C |
| Chemical compound, drug | ANA-12 | Sigma-Aldrich | catalog number SML0209 | in vivo administration |
| Chemical compound, drug | CNQX disodium salt | Tocris | catalog number 0190/10 | application in bath during electrophysiology analysis |
| Chemical compound, drug | (+)-MK 801 maleate | Tocris | catalog number 0924/10 | application in bath during electrophysiology analysis |
| Chemical compound, drug | Tetrodotoxin citrate (TTX) | Tocris | catalog number 1069/1 | application in bath during electrophysiology analysis |
| Chemical compound, drug | Picrotoxin | Tocris | catalog number 1128/1 | application in bath during electrophysiology analysis |
| Chemical compound, drug | Biocytin | Tocris | catalog number 3349/10 | electrodes filled with biocytin, versatile marker used for neuroanatomical investigations of neuron IHC |
| Chemical compound, drug | Naspm trihydrochloride | Tocris | catalog number 2766/10 | application in bath during electrophysiology analysis |
| Software, algorithm | ImageLab | BioRad | - | software used for quantification of protein bands in western blotting experiments |
| Software, algorithm | ImageJ software | NIH;Schneider et al. (2012) | RRID:SCR_003070 | software used for the quantification of protein bands in western blotting and confocal laser scanning microscope |
| Software, algorithm | ClampFit 9 | pClamp | Molecular Devices; RRID:SCR_011323 | data analysis |
| Software, algorithm | Origin 8.0 | Microcal | RRID:SCR_002815 | data analysis |
| Software, algorithm | Prism 5.3 | GraphPad | RRID:SCR_002798 | data analysis |
Animal model
Request a detailed protocolStudies were carried out in juvenile (P15-P35) and adult (P60-P75) knock-in Tor1a+/Δgag mice heterozygous for ΔE-torsinA, a mutation that removes a single glutamic acid residue (ΔE) from the torsinA protein, and in their wild-type Tor1a+/+ littermates (Goodchild et al., 2005). Genotyping was performed as described (Ponterio et al., 2018). Animal breeding, on a C57Bl/6J background, and handling were performed in accordance with the guidelines for the use of animals in biomedical research provided by the European Union's directives and Italian laws (2010/63EU, D.lgs. 26/2014; 86/609/CEE, D.Lgs 116/1992). The experimental procedures were approved by Fondazione Santa Lucia and University Tor Vergata Animal Care and Use Committees, and the Italian Ministry of Health (authorization #223/2017-PR).
Experimental design
Request a detailed protocolAge- and sex-matched wild-type and mutant littermates were randomly allocated to experimental groups. Investigators performing experiments and data analysis were blind to knowledge of genotype and treatment. Each observation was obtained from an independent biological sample. For electrophysiology, each cell was recorded from a different brain slice. All data were obtained from at least two animals in independent experiments. Biological replicates are represented with ‘N’ for number of animals and ‘n’ for number of cells. Sample size for any measurement was based on the ARRIVE recommendations on refinement and reduction of animal use in research, as well as on our previous studies.
Electrophysiology
Brain slice preparation
Request a detailed protocolMice were sacrificed by cervical dislocation, brains removed and sliced with a vibratome (Leica Microsystems) in oxygenated Krebs' solution (in mM: 126 NaCl, 2.5 KCl, 1.3 MgCl2, 1.2 NaH2PO4, 2.4 CaCl2, 10 glucose, 18 NaHCO3). Coronal and parasagittal corticostriatal slices (200–300 μm) were incubated in Krebs' solution at room temperature for 30 min. Then, individual slices were transferred into recording chambers continuously superfused with Krebs’ solution (32–33°C) saturated with 95% O2 and 5% CO2.
Patch-clamp recordings
Request a detailed protocolRecordings were performed with AxoPatch 200B amplifiers and pClamp 10.2 software (Molecular Devices). For voltage-clamp experiments, pipettes (2.5–5 MΩ) were filled with Cs+ internal solution (in mM: 120 CsMeSO3, 15 CsCl, 8 NaCl, 10 TEA-Cl, 10 HEPES, 0.2 EGTA, 2 Mg-ATP, and 0.3 Na-GTP; pH 7.3 adjusted with CsOH; 300 mOsm). For whole-cell recordings of glutamatergic sEPSCs, SPNs were clamped at HP=−70 mV in the presence of the GABAA receptor antagonist PTX (50 μM). For GABAergic sIPSCs, SPNs were recorded at HP =+ 10 mV in MK801 (30 μM) and CNQX (10 μM) to block NMDARs and AMPARs, respectively. Both mEPSCs and mIPSCs were measured by adding 1 μM TTX. PPR was measured at HP=−70 mV in PTX by delivering two stimuli at 25–1000 ms ISI. Synaptic strength was analyzed by measuring the NMDAR/AMPAR ratio at HP =+ 40 mV in PTX. The AMPAR–mediated component of EPSC was isolated in MK-801 and the NMDAR component was obtained by digital subtraction of the AMPAR component from the dual-component EPSC (Sciamanna et al., 2012). The AMPAR and NMDAR IV relationships were measured in the presence of PTX plus MK-801 or CNQX, respectively. The RI was calculated as ratio of the mean EPSC amplitudes measured at +40 mV and −70 mV.
Sharp-electrode recordings
Request a detailed protocolCurrent-clamp recordings of SPNs were performed with intracellular electrodes filled with 2M KCl (30–60 MΩ). Corticostriatal EPSPs were recorded in PTX (50 μM). HFS (three trains 100 Hz, 3 s, 20 s apart) was delivered at suprathreshold intensity to induce LTD. Magnesium was omitted to optimize LTP induction (Calabresi et al., 1992b). The EPSP amplitude was averaged and plotted over-time as percentage of control pre-HFS amplitude.
Gene expression analysis
Request a detailed protocolP26 Tor1a+/+ and Tor1a+/Δgag mouse cortex was collected in PCR clean tubes and stored at −80°C. Total RNA was isolated using TRI-reagent (Sigma Aldrich), quantified and treated with DNAase I (Invitrogen). Integrity was confirmed by 1% agarose gel electrophoresis. RNA was reverse-transcribed using random hexamer primer and anchored-oligo (dT)18 primer according to the manufacturer’s instructions (Transcriptor First Strand cDNA Synthesis Kit, Roche). Real-time PCR was performed on 25 ng cDNA by using LightCycler 480 Probes Master (04707494001, Roche) with Roche Light Cycler LC480 system, Bdnf and Hprt1 primers designed on the Roche Universal Probe Library Assay Design Center: https://configurator.realtimeready.roche.com/assaysupply_cp/pages/singleAssays/searchResult.jsf
Raw Ct values for Bdnf gene were normalized to the endogenous control gene Hprt1. Technical triplicates were analyzed for all samples and mean values were utilized for statistical analysis. The relative expression was determined using the 2-ΔΔCt method (Livak and Schmittgen, 2001).
Immunohistochemistry
Request a detailed protocolTo identify direct- and indirect-pathway SPNs electrodes were loaded with biocytin, as described (Martella et al., 2009). Briefly, slices were fixed with 4% PFA in 0.12 M PB and 30 μm thick sections were cut from each slice with a freezing microtome, then dehydrated with serial alcohol dilutions to improve antigen retrieval and reduce background (Buchwalow et al., 2011). We used the following primary antibodies: goat anti-DARPP-32 (1:500 AF6259, R and D system), mouse anti-Enkephalin (1:1000 MAB350, Millipore), and secondary antibodies: anti-goat alexa 647 (Invitrogen), anti-mouse cyanine 3 (Jackson ImmunoResearch) and streptavidin-conjugated alexa 488 (Life Technologies). All sections used for analysis were processed together. Images were acquired with a LSM700 Zeiss confocal laser scanning microscope and analyzed with ImageJ software (NIH; Schneider et al., 2012). Noise was reduced by applying background subtraction in ImageJ.
Subcellular fractionation and western blotting (WB)
Request a detailed protocolTo obtain a preparation that contains selectively proteins of the post-synaptic density (PSD), subcellular fractionation of striatal tissue was performed as reported (Gardoni et al., 2006; Paillé et al., 2010) with minor modifications. Briefly, striata were homogenized with a Teflon-glass potter in ice-cold 0.32M sucrose containing 1 mM HEPES pH 7.4, 1 mM MgCl2,1mM EDTA, 1 mM NaHCO3, 0.1 mM phenylmethanesulfonylfluoride (PMSF) in the presence of a complete set of proteases and phosphatase inhibitors (CompleteTM Protease Inhibitor Cocktail Tablets and PhosSTOPTM Phosphatase Inhibitor Cocktail, Roche Diagnostics). The homogenized tissue was centrifuged at 13,000 g for 15 min. The pellet was re-suspended in a buffer containing 75 mM KCl and 1% Triton X-100 and spun at 100,000 g for 1 hr. The final pellet, referred to as Triton-insoluble postsynaptic fraction (TIF), was homogenized in a glass-glass potter in 20 mM HEPES supplemented with CompleteTM tablets and stored at −80°C until use. Protein samples were separated onto an acrylamide/bisacrylamide gel at the appropriate concentration, transferred to a nitrocellulose membrane and immunoblotted with the appropriate primary and HRP-conjugated secondary antibodies. For WB analysis, the following unconjugated primary antibodies were used: polyclonal anti-GluN2A antibody (Sigma-Aldrich); monoclonal anti-GluN2B antibody (NeuroMab); polyclonal anti-GluA1 antibody (Merck Millipore); polyclonal anti-phospho-GluA1 (Ser845; Merck Millipore); monoclonal anti-GluA2 antibody (NeuroMab); monoclonal anti-PSD-95 antibody (NeuroMab); monoclonal anti-α-tubulin antibody (Sigma-Aldrich). Membrane development was performed with the reagent Clarity Western ECL Substrate (Bio-Rad) and labeling was visualized by Chemidoc Imaging System and ImageLab software (Bio-Rad). For quantification, each protein was normalized against the corresponding α-tubulin band.
WB analysis of BDNF on mouse striatum was performed as described (Sciamanna et al., 2015; Ponterio et al., 2018). Protein extracts (15–30 µg) were loaded with page LDS sample buffer (Invitrogen, Waltham, Massachusetts, USA) containing DTT and denatured at 95°C for 5 min. Proteins were separated on 15% SDS-PAGE, and transferred onto 0.45 µm polyvinylidene fluoride (PVDF) membranes. The following primary antibodies were utilized: rabbit anti-BDNF (1:200 sc-546, SantaCruz Biotechnology) and mouse anti-β-actin (1:20.000 A5441, Sigma Aldrich), as loading control, followed by anti-rabbit or anti-mouse horseradish peroxidase (HRP)-conjugated secondary antibodies. Immunodetection was performed by ECL reagent (GEHealthcare) and membranes were exposed to film (Amersham). Quantification of the band intensity on scanned filters was achieved by ImageJ software.
Spine morphology
Request a detailed protocolCarbocyanine dye DiI (Invitrogen) was used to label neurons as previously described (Kim et al., 2007; Stanic et al., 2015). Images were taken using an inverted LSM510 confocal microscope (Zeiss). For morphological analysis, cells were chosen randomly for quantification from four to eight different coverslips; images were acquired using the same settings/exposure times, and at least 10 cells for each condition were analyzed. Morphological analysis was performed with ImageJ software to measure spine density and size. For each dendritic spine the length, the head and neck width were measured, which was used to classify spines into categories (thin, stubby and mushroom) (Harris et al., 1992).
Statistical analysis
Request a detailed protocolData were analysed with ClampFit 9 (pClamp, Molecular Devices), Origin 8.0 (Microcal) and Prism 5.3 (GraphPad) softwares. All data were obtained from at least two independent experiments and are represented as mean ± SEM. Statistical significance was evaluated, as indicated in figure legends, using paired and unpaired Student's t test, and one-way ANOVA with post-hoc Tukey test and two-way ANOVA with Bonferroni posttest for group comparisons. Statistical tests were two-tailed, the confidence interval was 95%, and the alpha-level used to determine significance was set at p<0.05.
References
-
AMPAR trafficking in synapse maturation and plasticityCellular and Molecular Life Sciences 70:4411–4430.https://doi.org/10.1007/s00018-013-1309-1
-
BDNF signaling and survival of striatal neuronsFrontiers in Cellular Neuroscience 8:254.https://doi.org/10.3389/fncel.2014.00254
-
Long-term synaptic depression in the striatum: physiological and pharmacological characterizationThe Journal of Neuroscience : The Official Journal of the Society for Neuroscience 12:4224–4233.
-
Hyperkinetic disorders and loss of synaptic downscalingNature Neuroscience 19:868–875.https://doi.org/10.1038/nn.4306
-
Regulation of Ca2+-permeable AMPA receptors: synaptic plasticity and beyondCurrent Opinion in Neurobiology 16:288–297.https://doi.org/10.1016/j.conb.2006.05.012
-
Biophysical mechanisms regulating AMPA receptor accumulation at synapsesBrain Research Bulletin 93:57–68.https://doi.org/10.1016/j.brainresbull.2012.11.001
-
An anticholinergic reverses motor control and corticostriatal LTD deficits in Dyt1 ΔGAG knock-in miceBehavioural Brain Research 226:465–472.https://doi.org/10.1016/j.bbr.2011.10.002
-
Subcellular segregation of distinct heteromeric NMDA glutamate receptors in the striatumJournal of Neurochemistry 85:935–943.https://doi.org/10.1046/j.1471-4159.2003.01744.x
-
BookThe Mouse in Aging ResearchIn: Fox J. G, editors. The Mouse in Biomedical Research (2nd Edition). Burlington: American College Laboratory Animal Medicine (Elsevier). pp. 637–672.
-
A critical interaction between NR2B and MAGUK in L-DOPA induced dyskinesiaJournal of Neuroscience 26:2914–2922.https://doi.org/10.1523/JNEUROSCI.5326-05.2006
-
Impaired sequence learning in carriers of the DYT1 dystonia mutationAnnals of Neurology 54:102–109.https://doi.org/10.1002/ana.10610
-
New genetic insights highlight 'old' ideas on motor dysfunction in dystoniaTrends in Neurosciences 36:717–725.https://doi.org/10.1016/j.tins.2013.09.003
-
Generation of a novel rodent model for DYT1 dystoniaNeurobiology of Disease 47:61–74.https://doi.org/10.1016/j.nbd.2012.03.024
-
Three-dimensional structure of dendritic spines and synapses in rat hippocampus (CA1) at postnatal day 15 and adult ages: implications for the maturation of synaptic physiology and long-term potentiationJournal of Neuroscience 12:2685–2705.
-
Critical period regulationAnnual Review of Neuroscience 27:549–579.https://doi.org/10.1146/annurev.neuro.27.070203.144327
-
Presynaptic BDNF promotes postsynaptic long-term potentiation in the dorsal striatumJournal of Neuroscience 30:14440–14445.https://doi.org/10.1523/JNEUROSCI.3310-10.2010
-
Clinical disorders of brain plasticityBrain and Development 26:73–80.https://doi.org/10.1016/S0387-7604(03)00102-5
-
Labeling of dendritic spines with the carbocyanine dye DiI for confocal microscopic imaging in lightly fixed cortical slicesJournal of Neuroscience Methods 162:237–243.https://doi.org/10.1016/j.jneumeth.2007.01.016
-
An evolutionarily conserved transmembrane protein that is a novel downstream target of neurotrophin and ephrin receptorsJournal of Neuroscience 21:176–185.
-
GluR1 links structural and functional plasticity at excitatory synapsesJournal of Neuroscience 27:13706–13718.https://doi.org/10.1523/JNEUROSCI.3503-07.2007
-
Deep brain stimulation for dystonia in adults. Overview and developmentsStereotactic and Functional Neurosurgery 78:168–182.https://doi.org/10.1159/000068963
-
Emerging common molecular pathways for primary dystoniaMovement Disorders 28:968–981.https://doi.org/10.1002/mds.25547
-
Synaptic localization and activity of ADAM10 regulate excitatory synapses through N-cadherin cleavageJournal of Neuroscience 30:16343–16355.https://doi.org/10.1523/JNEUROSCI.1984-10.2010
-
Identification of pro- and mature brain-derived neurotrophic factor in human salivaArchives of Oral Biology 54:689–695.https://doi.org/10.1016/j.archoralbio.2009.04.005
-
Regional specificity of synaptic plasticity deficits in a knock-in mouse model of DYT1 dystoniaNeurobiology of Disease 65:124–132.https://doi.org/10.1016/j.nbd.2014.01.016
-
Calcium/calmodulin-dependent protein kinase II phosphorylation drives synapse-associated protein 97 into spinesJournal of Biological Chemistry 279:23813–23821.https://doi.org/10.1074/jbc.M402796200
-
Sensitive and critical periods during neurotypical and aberrant neurodevelopment: a framework for neurodevelopmental disordersNeuroscience & Biobehavioral Reviews 50:180–188.https://doi.org/10.1016/j.neubiorev.2014.12.001
-
Extrasynaptic membrane trafficking regulated by GluR1 serine 845 phosphorylation primes AMPA receptors for long-term potentiationThe Journal of Biological Chemistry 281:752–758.https://doi.org/10.1074/jbc.M509677200
-
NMDA receptor subunit diversity: impact on receptor properties, synaptic plasticity and diseaseNature Reviews Neuroscience 14:383–400.https://doi.org/10.1038/nrn3504
-
Mouse models of neurodevelopmental disease of the basal ganglia and associated circuitsCurrent Topics in Developmental Biology 109:97–169.https://doi.org/10.1016/B978-0-12-397920-9.00001-9
-
Emerging concepts in the physiological basis of dystoniaMovement Disorders 28:958–967.https://doi.org/10.1002/mds.25532
-
NIH Image to ImageJ: 25 years of image analysisNature Methods 9:671–675.https://doi.org/10.1038/nmeth.2089
-
Rhes regulates dopamine D2 receptor transmission in striatal cholinergic interneuronsNeurobiology of Disease 78:146–161.https://doi.org/10.1016/j.nbd.2015.03.021
-
Brain plasticity and cognitive functions after ethanol consumption in C57BL/6J miceTranslational Psychiatry 5:e696.https://doi.org/10.1038/tp.2015.183
-
Homeostatic plasticity in the developing nervous systemNature Reviews Neuroscience 5:97–107.https://doi.org/10.1038/nrn1327
-
Deep brain stimulation in the treatment of severe dystoniaJournal of Neurology 248:695–700.https://doi.org/10.1007/s004150170116
-
Deep brain stimulation for dystonia: make the lame walkAnnals of Neurology 57:613–614.https://doi.org/10.1002/ana.20491
-
Differential expression of neurotrophins in postnatal C57BL/6 mice striatumInternational Journal of Biological Sciences 5:118–127.https://doi.org/10.7150/ijbs.5.118
Article and author information
Author details
Funding
Ministero dell’Istruzione, dell’Università e della Ricerca (PRIN 2010-2011)
- Fabrizio Gardoni
- Antonio Pisani
Dystonia Medical Research Foundation (2017)
- Antonio Pisani
The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Acknowledgements
We wish to thank Dystonia Medical Research Foundation for funding and for their work helping to increase understanding of this disease. Authors are grateful to Dr Nicole Calakos for critical reading of the manuscript and for helpful comments. We wish to thank also Elisa Zianni and Massimo Tolu for their skillful technical support. This work was supported by a PRIN 2010–2011 grant of the Ministero dell’Istruzione, dell’Università e della Ricerca to AP and FG.
Ethics
Animal experimentation: Animal breeding and handling were performed in accordance with the guidelines for the use of animals in biomedical research provided by the European Union's directives and Italian laws (2010/63EU, D.lgs. 26/2014; 406 86/609/CEE, D.Lgs 116/1992). The experimental procedures were approved by Fondazione Santa Lucia and University Tor Vergata Animal Care and Use Committees and the Italian Ministry of Health (authorization #223/2017-PR).
Version history
- Received: November 3, 2017
- Accepted: March 2, 2018
- Accepted Manuscript published: March 5, 2018 (version 1)
- Version of Record published: March 13, 2018 (version 2)
Copyright
© 2018, Maltese et al.
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 1,718
- views
-
- 302
- downloads
-
- 57
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Neuroscience
Probing memory of a complex visual image within a few hundred milliseconds after its disappearance reveals significantly greater fidelity of recall than if the probe is delayed by as little as a second. Classically interpreted, the former taps into a detailed but rapidly decaying visual sensory or ‘iconic’ memory (IM), while the latter relies on capacity-limited but comparatively stable visual working memory (VWM). While iconic decay and VWM capacity have been extensively studied independently, currently no single framework quantitatively accounts for the dynamics of memory fidelity over these time scales. Here, we extend a stationary neural population model of VWM with a temporal dimension, incorporating rapid sensory-driven accumulation of activity encoding each visual feature in memory, and a slower accumulation of internal error that causes memorized features to randomly drift over time. Instead of facilitating read-out from an independent sensory store, an early cue benefits recall by lifting the effective limit on VWM signal strength imposed when multiple items compete for representation, allowing memory for the cued item to be supplemented with information from the decaying sensory trace. Empirical measurements of human recall dynamics validate these predictions while excluding alternative model architectures. A key conclusion is that differences in capacity classically thought to distinguish IM and VWM are in fact contingent upon a single resource-limited WM store.
-
- Neuroscience
Our ability to recall details from a remembered image depends on a single mechanism that is engaged from the very moment the image disappears from view.






