Abstract
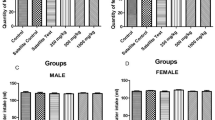
This study was conducted to obtain acute information of the oral dose toxicity of lyophilized water extract of Pinelliae Rhizoma, a dried tuber of Pinellia ternata (PR) in male and female mice. In order to calculated 50% lethal dose (LD50) and approximate lethal dose (ALD), test material was once orally administered to male and female ICR mice at dose levels of 2000, 1000, 500, 250, 125 and 0 (vehicle control) ml/kg (body weight). The mortality and changes in body weight, clinical signs, gross observation, organ weight and histopathology of principle organs were monitored 14 days after treatment with PR extract. We could not find any mortalities, clinical signs, changes in the body and organ weights, gross and histopathological findings except for dose-dependent increases in the hepatic fatty change frequencies detected in PR extract 2000 and 1000mg/kg treated in both male and female mice. The results obtained in this study suggest that LD50 and approximate LD in mice after single oral dose of PR extracts were considered over 2000 mg/kg in both and female male mice, but more than 1000mg/kg of PR extracts treatment could induce slight hepatotoxicity the fatty changes in mice.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Banks, W.J. (1986). Female reproductive system in Applied veterinary histology (Banks, W.J. Ed.). Williams & Wilkins, Baltimore, pp. 330–347, 506–526.
Chen, D., Wu, C.F., Huang, L. and Ning, Z. (2002). Effect of the aqueous extract of xiao-ban-xia-tang on gastric emptying in mice. Am. J. Chin. Med., 30, 207–214.
Chen, Z.X., Zhang, S.J., Lao, S.X., Hu, HT., Zhang, C.Y., Guan, S.H. and Gu, Y.L. (2005). He Jie Tang in the treatment of chronic hepatitis B patients. World J. Gastroenterol., 11, 6638–6643.
Chung, T.W., Koo, B.S., Choi, E.G, Kim, M.G, Lee, I.S. and Kim, C.H. (2006). Neuroprotective effect of a chuk-me-sun-dan on neurons from ischemic damage and neuronal cell toxicity. Neurochem. Res., 31, 1–9.
Dourish, C.T (1987). Effects of drugs on spontaneous motor activity in experimental psychopharmacology (Greenshaw, A.J. and Dourish, C.T. Eds.). Humana Press, Clifton, pp. 325–334.
Hodge, H.C. and Sterner, J.H. (1949). Tabulation of toxicity classes. Am. Ind. Hyg. Q., 10, 93.
Ho, S.T. and Liu, T.S. (1975). Studies on Plnellia ternate Breitenbach. Report I. Antiemetic action of Pinellla ternata Breitenbach and its components. Taiwan Yao Hsueh Tsa Chih, 27, 41–46
Irwin, S. (1968). Comprehensive observational assessment: la. A systemic, quantitative procedure for assessing the behavioral and physiological state of the mouse, Psychopharmacology, 13, 222–257.
Katagiri, F., Inoue, S., Sato, Y, Itoh, H. and Takeyama, M. (2004). Comparison of the effects of Sho-hange-ka-buku-ryo-to and Nichin-to on human plasma adrenocorticotropic hormone and Cortisol levels with continual stress exposure. Biol. Pharm. Bull., 27, 1679–1682.
Kim, Y.J., Shin, Y.O., Ha, Y.W., Lee, S., Oh, J.K. and Kim, Y.S. (2006). Anti-obesity Effect of Plnellia ternata Extract in Zucker Rats. Biol. Pharm. Bull., 29, 1278–1281.
Koo, B.S., Choi, E.G, Park, J.B., Cho, C.H., Chung, K.H. and Kim, C.H. (2005). Neuroprotective effect of Chuk-Me-Sun-Dan on NMDA- and AMPA-evoked nitric oxide synthase activity in mouse brain. Immunopharmacol. Immunotoxicol., 27, 499–514.
Korea Food and Drug Administration. Testing Guidelines for Safety Evaluation of Drugs (Notification No. 2005-60, issued by the Korea Food and Drug Administration on October 21, 2005), 2005.
Kurata, K., Takaaki, T., Ye, Y., Kaoru, K., Kiyotaka, K., Kunio, T., Kazuo, W. and Yoshiki, N. (1998). Quantitative analysis of anti-emetic principle in the tubers of Pinellia ternata by enzyme immunoassay. Plant Med., 64, 645–648
Lee, H.S., Lee, I.G and Ku, S.K. (2006). Single oral dose toxicity study of water extracts of Picrorrhiza Rhizoma in mice. J. Toxicol. Pub. Health, 22, 117–126.
Lee, J.E., Kim, H.J., Choi, E.K., Chai, H.Y, Yun, Y.W., Kim, D.J., Nam, S.Y., Lee, B.J., Ahn, B.W., Kang, H.G and Kim, Y.B. (2003). Four-week repeated-dose toxicity study on pinellia extract. Korean J. Lab. Anim. Sci., 19, 127–141.
Lee, J.H., Yang, K.J., Shin, H.D., Park, B.R., Son, C.W., Jang, H.J., Park, D.C., Lee, H.S. amd Ku, S.K. (2005). Single subcutaneous dose toxicity of Polycan®, a ß-glucan originated from Aureobasidium in mice. Lab. Anim. Res, 21, 299–305.
Lee, S.B., Kim, D.J., Kim, K.J., Jeon, B.H. and Woo, W.H. (1993). Study on the cytotoxic effect of Pinelliae Tuber extracts according to Prebrewed methods. J. Won Kwang Oriental Med., 3, 65–80.
Li, Y.L., Gan, G.P, Zhang, H.Z., Wu, H.Z., Li, C.L., Huang, Y.P, Liu, YW. and Liu, J.W. (2007). A flavonoid glycoside isolated from Smilax china L. rhizome in vitro anticancer effects on human cancer cell lines. J. Ethnopharmacol., 113, 115–124.
Maclachlan, N.J. and Cullen, J.M. (1995). Liver, biliary system, and exocrine pancreas in Thomson’s Special Veterinary Pathology (Carlton, M.W. and McGavin, M.D. Eds.). Mosby, St. Louis, pp. 81–115.
Maki, T., Takahashi, K. and Shibata, S. (1987). An antiemetic principle of Pinellia ternata Tuber. Plant Med., 53, 410–414
Organization for Economic Co-Operation and Development (Ed.). OECD guideline (423) for testing of chemicals -acute oral toxicity-acute toxic class method. 2001.
Park, J.H. and Seo, B.I. (2000). A philological study on poisoning of Pinelliae Rhizoma. J. East-West Med., 25, 47–55.
Pineda, M.H. (1989). Female reproductive system in Veterinary endocrinology and reproduction (McDonald, L.E. and Pineda, M.H. Eds.). Lea & Febiger, Philadelphia, pp. 303–354.
Plata, E.J. and Murphy, W.H. (1972). Growth and haemato-logic properties of the BALB/wm strain of inbred mice. Lab. Anim. Sci., 22, 712–720.
Shi, Y.L., Xu, Y.F. and Zhang, H. (1994). Effect of Pinellia ternata lectin on membrane currents of mouse motor nerve terminals. Sci. China B., 37, 448–453.
U.S. Environmental Protection Agency. Health Effects Test Guidelines OPPTS 870.100, Acute Toxicity Testing Background. US EPA August, Washington, USA (1998).
Yamaguchi, C., Fujita, S., Obara, T. and Ueda, T (1983). Effects of room temperature on reproduction, body weight and organ weights, food and water intakes, and hematology in mice. Exp. Anim., 32, 1–11.
Yang, H.C, Chung, M.H. and Chang, I.M. (2000). Study on the micronucleus test of Pinelliae Tuber Extracts according to Prebrewed methods. Poster presentation PB13. Kor J. Pharmacogn., 21, 258–259.
Zhao, Y.J., Ji, Z.Q., Zhang, X.N., Zhang, Y.Y., Wu, J.F., Fang, X. and Yue, W. (2005). Effect of Rhizoma pinelliae on vomiting in minks. Zhongguo Zhong Yao Za Zhi, 30, 277–279.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lim, YK., Park, JH., Seo, Bl. et al. Single Oral Dose Toxicity Study of Pinelliae Rhizoma Aqueous Extract in ICR Mice. Toxicol Res. 25, 147–157 (2009). https://doi.org/10.5487/TR.2009.25.3.147
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2009.25.3.147