Abstract
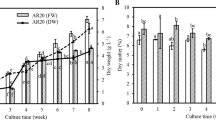
We investigated suitability of media and the types and concentrations of growth regulators necessary for establishing bioreactor cultures of mountain-grown ginseng. Effects of cytokinin and auxin on root growth differed with culture and accession. Low levels of cytokinin and high levels of auxin proved to be superior root-growth regulators. SH (Sheck & Hildebrant) culture medium produced better growth than blends of MS (Murashige & Skoog) and WPM (Wood plant medium) media. The best suspension culture for induction of roots was SH medium supplemented with 4 mg/L indole-3-butyric acid (IBA). Root growth under 1700 lux illuminance was reduced by as much as 10% relative to that under 3500 lux.
Similar content being viewed by others
Abbreviations
- IBA:
-
indole-3-butyric acid
- MS:
-
Murashige & Skoog
- NAA:
-
α-naphthaleneacetic acid
- SH:
-
Sheck & Hildebrant
References
Adams P (2002) Nutrotional control in hydroponics. In Hydroponic production of vegetable and ornamentals, Savvas D and Passam H (eds.), pp 211–262. Embryo Pub, Athens, Greece.
Akoachere JF, Chenwi EB, Njock TE, and Anong DN (2002) Antibacterial effect of Zingiber officinale and Garcinia kola on respiratory tract pathogens. East Afr Med J 79, 588–592.
Baily DA (1996) Alkalinity, pH, and acidification. In Water, media, and nutrition for greenhouse crops, Reed DW (9th ed.), pp 69–91. Ball Pub, Batavia, IL.
Bonfill M, Cusido RM, Palazon J, Pinol MT, and Morales C (2002) Influence of auxins on organogenesis and gin-senoside production in Panax ginseng calluses. Plant Cell Tiss Org Cult 68, 73–78.
Borkird C, Choi JH, and Sung R (1986) Effect of 2,4-D on the expression of embriogenic program in carrot. Plant Physiol 81, 1143–1146.
Bugbee B (1995) Nutrient management in recirculating hydroponic culture. In Proceedings of the 16 th Annual Conference on Hydroponics, pp. 525–529. Hydroponic Society of America, San Ramon CA.
Choi SM, Son SH, Yun SR, Kwon OW, Seon JH, and Paek KY (2000) Pilot-scale culture of adventitious roots of ginseng in a bioreactor system. Plant Cell Tiss Org Cult 62, 187–193.
Ficker C, Smith ML, and Akpagana K (2003) Bioassay-guided isolation and identification of antifungal compounds from ginger. Phytother Res 17, 897–902.
Flores HE, Yao-Rem D, and Cuello JL (1993) Green roots: photosynthesis and photoautotrophy in an underground plant organ. Plant Physiol 101, 363–371.
Ippoushi K, Azuma K, Ito H, Horie H, and Higashio H (2003) 6-Gingerol inhibits nitric oxide synthesis in activated J774.1 mouse macrophages and prevents peroxyni-trite-induced oxidation and nitration reactions. Life Sci 73, 3427–3437.
Murashige T and Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15, 473–497.
Nagasawa H, Watanabe K, and Inatomi H (2002) Effects of bitter melon (Momordica charantia l.) or ginger rhizome (Zingiber officinale Rosc) on spontaneous mammary tumorigenesis in SHN mice. Am J Chin Med 30, 195–205.
Paek KY, Kim KR, and Cheon KT (2000) Industrial production of saponin extract from cell and tissue of Panax ginseng cultured in bioreactors. Annual reports. Department of Science and Technology, Seoul, Korea.
Paek KY, Chakrabarty D, and Hahn EJ (2005) Application of bioreactor systems for large scale production of horticultural and medicinal plants. Plant Cell Tiss Org Cult 81, 287–300.
Pierik RLM (1987) In vitro Culture of Higher Plants. Martinus Nijhoff (ed), The Hague.
Roh KH, Kim TS, Lee JH, Choi IL, Choi YH, and Jang YS (1996) In vitro propagation and tuberization of plantlet regeneration from shoot-tip culture in ginger. Korean J Plant Tiss Cult 23, 129–134.
Sandnes JM, Källqvist T, Wenner D, and Gislerød HR (2005) Combined influence of light and temperature on growth rates of Nannochloropsis oceanica: linking cellular responses to large-scale biomass production. J Appl Phycol 17, 515–525.
Schwarz M (1995) Soiless culture management, Springer Verlag, 31–56. Berlin, Germany
Sharma TR, Singh BM, and Chauhan RS (1994) Production of disease-free encapsulated buds of Zingiber officinale Rosc. Plant Cell Rep 13, 300–302.
Sharma TR and Singh BM (1997) In vitro microrhizome production in Zingiber officinale Rosc. Plant Cell Rep 15, 274–277.
Sheck RU and Hildebrant AC (1972) Medium and techniques for induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Can J Bot 50, 199–204.
Taya M, Sato H, Masahiro K, and Tone S (1994) Characterization of pak-bung green hairy roots cultivated under light irradiation. J Ferment Bioeng 78, 42–48.
Thanh NT, Murthy HN, Yu KW, Jeong CS, and Hahn EJ (2004) Effect of inoculum size on biomass accumulation and ginsenoside production by large-scale cell suspension cultures of Panax ginseng. J Plant Biotech 6, 265–268.
William A, John G, and Hendel J (1996) Reversed-phase high-performance liquid chromatographic determination of ginsenosides of Panax quinquefolium. J Chromatogr 775, 11–17.
Yang JS (1992) Plant regeneration from adventitious rootd-erived calli of ginseng (Panax ginseng C. A. Meyer). J Agric Assoc China 159, 41–48.
Yoshikawa T and Furuya T (1987) Saponin production by cultures of Panax ginseng transformed with Agrobacterium rhizogenes. Plant Cell Rep 6, 449–453.
Zhou S and Brown DC (2006) High efficiency plant production of North American ginseng via somatic embryogenesis from cotyledon explants. Plant Cell Rep 25, 166–173.
Ziv M (2005) Simple bioreactors for mass propagation of plants. Plant Cell Tiss Org Cult 81, 277–285.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, J.G., Seong, E.S., Goh, E.J. et al. Factors involved in masspropagation of ginseng (Panax ginseng C. A. Meyer) using bioreactor system. J. Korean Soc. Appl. Biol. Chem. 52, 466–471 (2009). https://doi.org/10.3839/jksabc.2009.080
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.3839/jksabc.2009.080