Published online May 14, 2017. doi: 10.3748/wjg.v23.i18.3228
Peer-review started: October 11, 2016
First decision: October 28, 2016
Revised: November 17, 2016
Accepted: March 15, 2017
Article in press: March 15, 2017
Published online: May 14, 2017
Although viral hepatitis treatments have evolved over the years, the resultant liver cirrhosis still does not completely heal. Platelets contain proteins required for hemostasis, as well as many growth factors required for organ development, tissue regeneration and repair. Thrombocytopenia, which is frequently observed in patients with chronic liver disease (CLD) and cirrhosis, can manifest from decreased thrombopoietin production and accelerated platelet destruction caused by hypersplenism; however, the relationship between thrombocytopenia and hepatic pathogenesis, as well as the role of platelets in CLD, is poorly understood. In this paper, experimental evidence of platelets improving liver fibrosis and accelerating liver regeneration is summarized and addressed based on studies conducted in our laboratory and current progress reports from other investigators. In addition, we describe our current perspective based on the results of these studies. Platelets improve liver fibrosis by inactivating hepatic stellate cells, which decreases collagen production. The regenerative effect of platelets in the liver involves a direct effect on hepatocytes, a cooperative effect with liver sinusoidal endothelial cells, and a collaborative effect with Kupffer cells. Based on these observations, we ascertained the direct effect of platelet transfusion on improving several indicators of liver function in patients with CLD and liver cirrhosis. However, unlike the results of our previous clinical study, the smaller incremental changes in liver function in patients with CLD who received eltrombopag for 6 mo were due to patient selection from a heterogeneous population. We highlight the current knowledge concerning the role of platelets in CLD and cancer and anticipate a novel application of platelet-based clinical therapies to treat liver disease.
Core tip: Platelets improve liver fibrosis and accelerate liver regeneration; therefore, patients with liver dysfunction due to chronic liver disease (CLD) and cirrhosis can benefit from platelet transfusion. However, administration of the thrombopoietin receptor agonist eltrombopag for 6 mo did not result in the improvement of liver function in patients with CLD despite its long-term safety and ability to maintain an increased platelet count. We believe that this difference is due to platelet aging. Therefore, we are pursuing novel strategies with thrombopoietin receptor agonists and desialylated formulations to treat liver diseases.
- Citation: Kurokawa T, Ohkohchi N. Platelets in liver disease, cancer and regeneration. World J Gastroenterol 2017; 23(18): 3228-3239
- URL: https://www.wjgnet.com/1007-9327/full/v23/i18/3228.htm
- DOI: https://dx.doi.org/10.3748/wjg.v23.i18.3228
Chronic liver disease (CLD) refers to a long-term pathological process of continuous destruction of liver parenchyma and its gradual substitution with fibrous tissue, which ultimately results in liver cirrhosis associated with a fatal outcome. CLD has diverse etiologies, which include hepatotrophic viruses, chemicals, alcohol and drug abuse, autoimmune disorders, cholestasis and metabolic diseases[1,2], and it is a major cause of morbidity and mortality in many countries[3,4]. Hepatocellular carcinoma (HCC) is a dominant complication of CLD and cirrhosis, with the third highest death rate among malignancies in the world[5].
HCC was shown to significantly correlate with advanced fibrosis, as a sustained wound-healing response to hepatitis C virus infection; however, despite the progress in viral hepatitis therapies, they are unable to completely heal hepatocellular injuries and prevent liver cirrhosis[6]. At present, liver transplantation is the only treatment option for end-stage liver failure, but its clinical availability is hindered by serious problems such as donor shortage, surgical complications, graft rejection and high cost[1,7-11]. Therefore, other therapeutic approaches are being looked into; among them, measures to resolve liver fibrosis have been investigated.
Liver fibrosis is caused by a continuous excessive deposition of the extracellular matrix (ECM) in response to chronic liver injury[1,2]. Although advanced liver fibrosis has been considered irreversible, resulting in permanent substitution of hepatocytes with the ECM components, recent reports indicate that certain immunotherapies may promote partial resolution of liver cirrhosis[12-15]. These studies encourage the development of novel approaches to treat patients with advanced CLD.
Thrombocytopenia, i.e., the reduction of platelet count in blood, is a common hematological complication of CLD caused by decreased production of hormone thrombopoietin (TPO) in the damaged liver and/or increased destruction of platelets through phagocytosis in the enlarged spleen, as well as the loss of hematopoietic function in bone marrow due to alcohol abuse or viral infection[16-19]. Platelets are anuclear blood cells derived from TPO-stimulated megakaryocytes; they not only play a critical role in hemostasis, but also secrete several growth factors, including platelet-derived growth factor (PDGF) and hepatocyte growth factor (HGF), which promote liver regeneration[20-25]. As a feedback response, thrombocytopenia further aggravates hepatic destruction and contributes to the pathogenesis of CLD and cirrhosis, suggesting that measures to prevent platelet loss may be useful as a therapeutic approach to treat CLD.
Indeed, our previous studies in experimental animal models revealed that platelets play a crucial role in promoting liver regeneration after hepatectomy by inducing hepatocyte proliferation[26-29] and stopping the progression of liver fibrosis[30-32]. Furthermore, in patients with CLD and cirrhosis, platelet transfusion and splenectomy used as a platelet increment therapy, positively affected hepatic function[33-35]. Along these lines, we have investigated the safety of long-term administration of the TPO receptor (TPO-R) agonist eltrombopag for treating patients with CLD.
However, the effect of thrombocytopenia on liver damage and exact mechanisms underlying thrombocytopenia in patients with CLD and cirrhosis are still unclear and require further study to confirm the clinical utility of increasing platelets in CLD.
The aim of this editorial is to summarize the current perspectives of novel approaches to treat liver cirrhosis based on augmentation of platelet counts using TPO-R agonists.
Liver fibrogenesis is triggered by destruction of hepatic cells and represents a wound-healing process leading to excessive deposition of the matrix proteins collagens and elastin, glycoproteins, proteoglycans and carbohydrates; in the context of chronic liver injury, fibrosis ultimately results in the substitution of liver tissue with ECM, formation of scar tissue, and gradual ceasing of hepatic functions[1,36]. Histologically, liver consists of parenchymal hepatocytes (70%-80%) and non-parenchymal cells such as Kupffer cells, sinusoidal endothelial cells and stellate cells. Hepatic stellate cells (HSCs) reside in the perisinusoidal space of the liver, also known as the space of Disse, between hepatocytes and sinusoidal endothelial cells and are the major fibrogenic cell type in the liver as they produce a large number of ECM components and secrete transforming growth factor-β (TGF-β), a key mediator of liver fibrogenesis[1,36].
In the normal liver, HSCs have a star-like morphology corresponding to a quiescent state, and their primary function is the storage of vitamin A as retinol ester in lipid droplets[37,38]. In response to liver injury, HSCs undergo activation and change into contractile myofibroblastic cells, which proliferate, secrete TGF-β, and increase matrix production. As a result, collagens IV and VI in the space of Disse are progressively replaced by fibrous collagens I and III and fibronectin, characteristic for ECM remodeling and fibrosis[39,40].
In our previous study, we revealed a link between the activation of human HSCs and platelets by showing that platelets and platelet-derived extracts suppressed transdifferentiation of quiescent HSCs into the myofibroblast-like phenotype as well as the production of collagen type I via cAMP signaling[31]. The underlying mechanism is based on the increase of adenosine concentration in the HSC milieu due to breakdown of ADP and ATP, which are abundant in platelet-dense granules[31]. As a result, adenosine entering HSCs through its cognate receptors prevents their activation and down-regulates their ability to secrete TGF-β and deposit the ECM. In addition, interaction between HSCs and platelets promotes the release of platelet-derived HGF, which was shown to inhibit the expression of type I collagen in cultured HSCs[41] and to attenuate liver fibrosis in mice by decreasing hepatic TGF-β secretion and blocking myofibroblast activation[42]. However, although these findings indicate that platelets can reduce hepatic fibrogenesis through inhibition of HSC activation, it is unclear whether they can be translated to the clinical situation, as the production of HGF by human platelets is lower than that by rodent platelets[43].
TPO is the most important factor in the regulation of megakaryocyte proliferation and differentiation into platelets through activation of its cognate receptor c-Mpl, also known as TPO-R[44]. Several agonists of the c-Mpl receptor, such as eltrombopag and romiplostim, are approved for clinical application as agents by which to increase platelet counts in chronic immune thrombocytopenia[44,45]. Moreover, they are currently undergoing clinical trials as treatment options to reduce thrombocytopenia in patients with CLD and liver cirrhosis[46-48], as the increase in platelet counts could make these patients eligible for interferon-based antiviral therapy[49,50].
The strategy to treat liver fibrosis in CLD through inhibition of thrombocytopenia was proved feasible in studies showing that TPO improved both platelet counts and liver fibrosis, even in conditions of hepatic cirrhosis[30,32]. Thus, in cirrhotic rats with dimethylnitrosamine-induced liver fibrosis and 70% hepatectomy, platelet increase by a single intravenous injection of TPO correlated with the inhibition of HSC activation and decrease of the fibrotic area in the liver, while antiplatelet serum attenuated hepatic regeneration[30]. In another study, mice with liver fibrosis induced by carbon tetrachloride (CCl4) showed improvement after weekly intraperitoneal administration of TPO for 5-8 wk[32].
Although mechanistic insights into the correlation of increased platelet counts with the reversal of liver fibrosis are yet to be provided, it can be suggested that platelets may promote hepatocyte proliferation by secreting HGF, which is a potent mitogen for hepatocytes through activation of the MET receptor that is essential for organogenesis and wound healing. Moreover, HGF may contribute to the resolution of fibrosis by modulating levels of TGF-β and matrix metalloproteinases (MMPs), which are the main ECM enzymes degrading collagen.
The suggested association between platelets, HGF, and hepatic fibrosis is supported by the findings of Takahashi et al[51], who showed that transfused human platelets improved CCl4-induced liver fibrosis in severe combined immune deficiency mice by increasing HGF levels in the mouse liver, which suppressed HSC activation, induced MMP-9 expression and inhibited hepatocyte apoptosis.
Liver regeneration is provided by the proliferation of both parenchymal and non-parenchymal hepatic cells, including hepatocytes, liver sinusoidal endothelial cells (LSECs), biliary epithelial and Kupffer cells, and HSCs, which contribute to the restoration of destroyed hepatic tissue[52]. Cell proliferation is triggered by several growth factors and cytokines, such as HGF, TGF-α, tumor necrosis factor-α (TNF-α), epidermal growth factor (EGF) and interleukin-6 (IL-6), which activate their cognate receptors and, consequently, downstream signaling and transcription of the genes associated with cell cycle progression[52-58]. Among the signaling cascades mediating platelet effects on the process of liver regeneration, the most important are TNF-α/nuclear factor-kappa B (NF-κB), IL-6/signal transducer and activator of transcription 3 STAT3, and phosphatidylinositol-3-kinase (PI3K)/Akt.
Previous studies indicate that platelets can exert positive effects on liver regeneration through cooperation with LSECs and Kupffer cells, and direct interaction with hepatocytes.
LSECs mostly consist of sinusoidal cells which, through formation of a continuous thin layer of the sinusoidal endothelium, create a structural barrier between the hepatic parenchyma and blood flowing through the liver[59,60]. LSECs play an important role in the maintenance of hepatic functions by providing exchange of nutrients between circulating blood and hepatocytes because of the presence of open pores beneath the endothelium[61]. In addition, LSECs secrete immunoregulatory cytokines, including HGF, IL-1, IL-6 and interferons, affecting liver regeneration. Thus, IL-6 secretion increased following hepatectomy[62,63] triggers STAT3 phosphorylation in hepatocytes, which up-regulates the synthesis of acute phase proteins as a part of the mechanism restoring the disturbed physiological homeostasis[64]. The direct contact of platelets with LSECs stimulated LSEC proliferation and accelerated DNA synthesis in hepatocytes by inducing IL-6 secretion, possibly via sphingosine 1-phosphate (S1P), a major bioactive lysophospholipid released from platelets[29]. S1P is known as a regulator of diverse cellular activities, including migration, proliferation and cytoskeletal remodeling, and is known to induce STAT3 activation by stimulating IL-6 secretion[65]. Activated platelets secrete high amounts of S1P which acts on endothelial cells in the processes involving platelet-endothelial interactions, such as thrombosis, angiogenesis and atherosclerosis[65,66].
Another type of non-parenchymal cell interacting with platelets is the Kupffer cells, which constitute over 80% of the tissue macrophages found in the body and act against gastrointestinal bacteria, microbial debris and endotoxins[67]. Upon activation, Kupffer cells secrete important growth-stimulating cytokines that promote hepatocyte proliferation after hepatectomy and induce processes involved in hepatic tissue restoration[68]. Kupffer cells are the most important source of IL-6 and TNF-α; the latter is increased following hepatectomy, suggesting that this cytokine as well as its producers (Kupffer cells) are implicated in the restoration of hepatic function in pathologic conditions. This notion is supported by the observations that anti-TNF-α antibodies suppressed hepatocyte proliferation[69], while TNF-α receptor-deficient mice had delayed liver regeneration after hepatectomy[70,71] because of decreased production of IL-6, which is a key target of TNF-α receptor activation in the regenerating liver[52]. As Kupffer cells are the most active producers of both TNF-α and IL-6 in the liver, it is not surprising that Kupffer cell-depleted mice fail to up-regulate TNF-α and IL-6 secretion after hepatectomy[72].
However, the role of Kupffer cells in liver regeneration is controversial. It has been shown that the interaction among platelets, Kupffer cells and leukocytes promote endothelial cell apoptosis in the liver following ischemia/reperfusion[73]. Depletion of Kupffer cells decreased platelet adherence in sinusoids in rats subjected to ischemia/reperfusion and attenuated damage to liver endothelium[74], which is consistent with the findings that platelets adhering to Kupffer cells during the early period of ischemia/reperfusion promoted hepatocyte apoptosis[75]. Although Nakamura et al[76] reported that in lipopolysaccharide-injected mice, platelets migrate to the space of Disse, which is mediated by their interaction with Kupffer cells, and then enter hepatocytes, the role of this process in hepatic regeneration is unclear. Further studies are needed to elucidate the mechanism underlying the impact of platelet-Kupffer cell interaction in liver fibrosis. However, it is evident that the contact between platelets and Kupffer cells cause activation of both cells.
Finally, platelets can induce hepatic regeneration by directly interacting with hepatocytes. Thus, in thrombocytotic BALB/c mice, platelets accumulate in the liver shortly after liver resection, causing the regeneration of hepatic tissue even following 90% hepatectomy and preventing liver failure by promoting cell cycle progression and metabolic pathways in hepatocytes[77]. Such stimulation of hepatocyte activity is likely a result of platelet accumulation in the sinusoidal space, from where they flow into the space of Disse and directly contact hepatocytes[26-28]. These findings suggest that following hepatic injury, platelets quickly migrate to the liver where they, through direct interaction with hepatocytes, activate cell cycle transition-related pathways and induce rapid hepatocyte proliferation. This notion is supported by the study using a co-culture chamber system, which showed that the contact between platelets and hepatocytes triggered the secretion of growth factors, including HGF, IGF-1 and VEGF from platelets, which induced hepatocyte proliferation[20].
We suggest the following mechanistic model explaining the effect of platelets on liver regeneration. Platelets migrating to the injured liver translocate from the liver sinusoids to the space of Disse, where, upon interaction with hepatocytes, they secrete HGF, IGF-1 and VEGF, which induce hepatocyte proliferation, resulting in liver regeneration. However, this model may not be fully applicable to humans, because human platelets do not secrete sufficient amounts of HGF[43]; therefore, IGF-1 may be the most important platelet-derived growth factor involved in the restoration of the human liver.
A recent study suggested an additional mechanism underlying platelet stimulation of liver regeneration. Thus, Kirschbaum et al[78] showed that transfer of coding or regulatory RNA could occur between platelets and hepatocytes, promoting hepatocyte proliferation. However, the role of both mechanisms, i.e. the release of growth factors and/or RNA transfer from platelets, in liver regeneration needs confirmation in vivo[79,80].
Thrombocytopenia is typically treated by platelet transfusion, which is suggested to improve liver function. However, it has not been established whether platelet transfusion can benefit CLD patients with thrombocytopenia since the pathogenesis of thrombocytopenia in CLD is multifactorial; therefore, the published guidelines on platelet transfusion do not cover CLD-related platelet loss[49]. As animal experiments indicate feasibility of using blood transfusion for thrombocytopenia associated with chronic liver injuries, clinical trials have been conducted.
A recent study included patients with CLD and cirrhosis (Child-Pugh class A or B) who demonstrated thrombocytopenia with platelet counts between 50000 and 100000/μL; the patients were treated with 10 U of platelet concentrate weekly for 12 wk and followed up for 9 mo after the last transfusion[33]. Although the platelet count did not show a significant increase, a marked improvement of liver function was observed, as evidenced by higher serum albumin levels 1 and 3 mo post-transfusion and higher serum cholinesterase concentration 9 mo post-transfusion; at the same time, serum hyaluronic acid levels indicative of liver fibrosis tended to decrease. However, this clinical trial was a non-controlled, non-randomized study based on a small sample size (6 patients); therefore, randomized controlled trials using larger patient cohorts need to be conducted in order to conclusively determine clinical value of platelet transfusion in CLD.
However, on the other hand, platelet preparations, despite recent prolongation of the preservation period, can only be used for 4 d due to the need to prevent proliferation of bacteria by preservation at room temperature. Furthermore, in patients with hematologic diseases who require repeated blood transfusions, alloantibodies are produced for human leukocyte antigens (HLAs) and human platelet antigens (HPAs) that differ from the patient’s own antigens, thereby triggering platelet transfusion refractoriness (PTR). Patients with PTR caused by anti-HLA antibodies or anti-HPA antibodies require HLA/HPA-matched platelet transfusions; however, securing donors is extremely difficult[81].
These factors result in an imbalance between supply and demand, thus generating interest in research that seeks to develop an alternative transfusion source to blood donation. Despite research in which hematopoietic stem cells (which are somatic stem cells) are used as a source to manufacture platelets ex vivo, this research has not been put to practical use due to the absence of an ex vivo human hematopoietic stem cell amplification method in which the cells’ functions can be retained.
Using our independently developed human mesenchymal stromal cells, we have succeeded in cultivating larger numbers of megakaryocytes, the sources of platelet production, than reported in previous studies; however, this method has not produced enough platelets to serve as a substitute for donated blood[82]. Similarly, studies in which umbilical cord blood-derived CD34+ cells are used as a source for red blood cell production have been unable to amplify these cells ex vivo; thus, no technique has yet been developed that can produce 1012 red blood cells, the number of red blood cells needed for a single transfusion[83].
Alternatively, pluripotent embryonic stem cells (ES cells) and induced pluripotent stem cells (iPS cells)[84] can be grown semipermanently in vitro and can resolve the crucial issue of “yielding the number of original cells”. In iPS cell-based regenerative medicine, post-transplantation cancer is a major concern. In response to this concern, safety for platelets and red blood cells can easily be ensured with a combination of anucleated cells, removal of nucleated cells with a filter prior to transfusion, and radiation to eliminate mixed-in lymphocytes. In addition, banking of iPS cells with various HLAs enables the construction of a system for stable provision of HLA-matched platelets[85].
Furthermore, Ono-Uruga et al[86] have discovered that pre-adipocytes endogenously possess the megakaryocyte inducer p45NF-E2 and can induce differentiation of megakaryocytes into platelets while increasing their own expression of p45NF-E2; they are striving to stably and safely produce platelets for transfusion from pre-adipocytes for medical applications. However, the number of platelets produced by this method is currently too low for practical use.
Although platelet increment therapies such as splenectomy and platelet transfusion can ameliorate CLD and liver cirrhosis, they may also cause serious side effects[48,87-90]. Thus, portal vein thrombosis[48,87], hemorrhage, infection and injury to the pancreatic tail[88] are among surgical complications encountered during splenectomy, while the platelet activation frequently observed in platelet transfusion may result in proinflammatory responses, febrile non-hemolytic reactions, and acute lung injury[89,90]. Besides, as we indicated earlier, there are reports about detrimental effects of platelets on hepatocytes[73-75]. Therefore, other approaches to treat thrombocytopenia in CLD should be considered. Among them, TPO-R agonists eltrombopag and romiplostim, already approved as therapeutics for chronic immune thrombocytopenic purpura, may also hold promise as treatment for CLD and liver cirrhosis.
Eltrombopag is a low molecular weight, synthetic nonpeptidyl drug, whereas romiplostim is a peptide containing an IgG Fc fragment in its structure[91]. Eltrombopag has already been tested in a phase II clinical trial involving patients with hepatitis C-associated cirrhosis and concurrent thrombocytopenia to increase platelet counts before starting anti-viral therapy with peg-interferon and ribavirin, which showed promising results[47]. Although eltrombopag is a TPO-R ligand, it caused little effect on platelet function and did not activate the PI3K/Akt pathway, in contrast to TPO, which caused significant platelet activation[92].
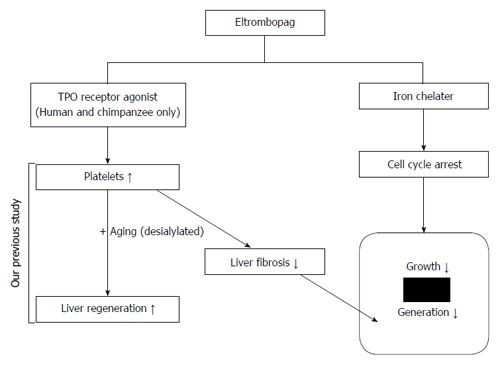
The safety of using TPO-R (c-Mpl) agonists was examined in c-Mpl-expressing leukemia cells. Similar to TPO, eltrombopag did not increase but rather inhibited proliferation of leukemia cells in vitro, suggesting the lack of tumorigenic side effects[93]. Moreover, TPO had no proliferative effect on HCC either in vitro or in vivo[94], while eltrombopag induced cell cycle arrest in HCC, demonstrating that it may act as a chemotherapeutic agent to inhibit the progression of HCC developed as a complication of CLD and liver cirrhosis[95]. Thus, eltrombopag represents a promising novel candidate drug to treat both CLD-related thrombocytopenia and associated malignant neoplasm.
To increase platelets in a continuous manner and avoid platelet transfusion refractoriness, we conducted an exploratory clinical trial and administered eltrombopag for 6 mo. The study included 5 patients with both CLD and a hepatitis C virus infection (Child-Pugh class A) who presented with thrombocytopenia (average platelet count 54 × 109/L). All of the patients administered eltrombopag maintained platelet counts between 10.0 and 15.0 × 1010/L during the 6-mo study, and their serum albumin, cholinesterase, alanine aminotransferase, T-bilirubin, hyaluronic acid and type IV collagen levels, as well as the platelets (%) and liver volumes, were stable throughout the clinical trial. The liver volumes calculated by computed tomography during the clinical trial were also stable, and no new cancerous lesions were observed.
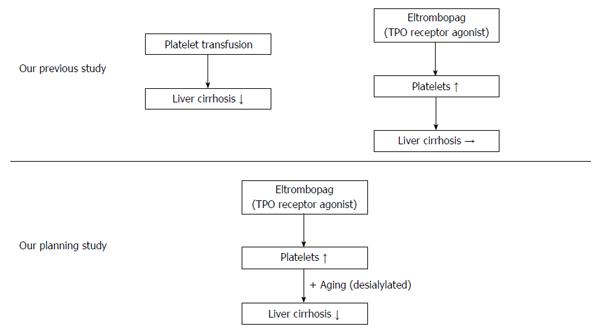
However, in contrast to our previous clinical study[33], we predicted that the increment values would be small because the liver function was representative of a heterogeneous population (Figure 1). Additionally, recent reports stated that the aging of platelets controls TPO production[96]; thus, we concluded that the aging and activation of platelets are involved in liver regeneration. Therefore, we are planning a novel therapy with TPO receptor agonists by using a desialylated formulation. In other words, increasing the platelet count by using TPO and aging the platelets via administration of a desialylated formulation to promote liver regeneration could be an effective treatment of CLD and liver cirrhosis (Figure 1).
Together with the antitumor effects of eltrombopag on hepatocellular carcinoma (HCC) that we previously reported[95], we are preparing for eltrombopag clinical trials for HCC patients with unmet medical needs and who cannot use sorafenib (Figure 2).
Since Trousseau[97] first reported the excessive blood coagulation in cancer patients with elevated platelet counts in 1865, a large number of studies have been conducted on cancer and platelets[98].
Cancer development involves the following progression: (1) separation from the primary tumor and infiltration of blood vessels; (2) transportation through blood flow; (3) adhesion to the capillary walls of distant organs; (4) extravascular migration; and (5) proliferation at the new site to form a metastatic lesion. Of course, platelets are involved in almost every step of this process.
For epithelial cancers to separate from their primary tumor, cells are required to break free from the strong cellular adhesion with the surrounding cells and then separate and migrate. The process of a cell losing its cellular adhesiveness, gaining the mesenchymal functions of migration and invasive capacity, and becoming resistant to apoptosis is known as epithelial-mesenchymal transition (EMT). Labelle et al[99] demonstrated that platelet-secreted TGF-β and direct interactions between cancer cells and platelets can induce EMT and promote metastasis.
Normally, the mass of cancer cells in the circulating blood is destroyed by the attack of the immunocompetent cell and the shear stress in the blood vessel, and it is said that only 0.01% of the cancer cells are involved in metastasis. However, when platelets are activated by cancer cells, if platelets stick around the cancer cells and surround the periphery, the possibility of survival, as protected from shear stress and attack by immunocompetent cells, increases.
Protection from shear stress: Using a shear stress-induced platelet aggregation measuring device, Egan et al[100] demonstrated that elevation of LDH levels induced by cell destruction was significantly lower when both platelets and cancer cells were present compared to the presence of cancer cells alone.
Protection from natural killer cells: Natural killer (NK) cells recognize cells with poor MHC class I expression as non-self and attack those cells. In 1999, Nieswandt et al[101] reported a mechanism by which platelets can surround cancer cells and physically block access of NK cells in order to protect cancer cells from damage. In 2012, Placke et al[102] reported that MHC class I, which is abundantly expressed on platelets, can transfer from the adherent platelets to the cancer cells; therefore, cancer cells are then recognized as “self” by NK cells and escape injury. Besides this, some soluble substances from activated platelets reduce NK cell activity, and TGF-β released from platelets suppresses the expression of NKG2G on NK cells and reduces its cytotoxic activity, as has been reported.
Various membrane proteins that bind to endothelial cells are expressed on platelets; thus, an agglomeration of cancer cells and platelets can bind to the endothelium of blood vessels and is thought to form footholds for metastases. However, it is possible that cancer cells could adhere to von Willebrand factor and P secretin expressed on platelets that have been activated by blood vessel endothelium-mediated interactions with GPIb[103-105]. Additionally, both P secretin and PSGL-1 are expressed on both platelets and blood vessel endothelium, the latter of which may become a foothold for metastases.
Following the agglomeration of cancer cells and platelets has bound to the blood vessel endothelium, release of platelet granules plays an important role during extravascular migration, as demonstrated in experiments using either granule content or knockout mice incapable of granule release[106,107]. It has been reported that ATP released from platelets activated by cancer cells binds with purinergic receptors in the blood vessel endothelium and elevates intracellular calcium via Gq/G11 signaling and increases the permeability of the blood vessel endothelial barrier via activation of protein kinase C, thereby driving the extravascular migration of cancer cells.
As a result of the above-mentioned activation, platelets release various growth factors, such as VEGF, PDGF and bFGF, which are said to promote tumor proliferation[106]. Reports indicate that when mice were injected with stomach cancer cell lines and ovarian cancer cell lines, and subjected to platelet reduction via platelet antibodies, the tumor volume was less pronounced[108,109].
There are also reports describing that platelet infusion into tumor-bearing mice results in a significant increase in the tumor volume and the number of Ki67-positive tumor cells, which are indicative of cell proliferation[109,110]. On the other hand, there are also reports suggesting that the tumor volume does not change when platelet reduction is induced in mice transplanted with melanoma cell lines[111] and, as in the reports described above, that the tumor volume is not affected even when TPO is administered to mice transplanted with liver cancer cell lines[94]. Based on these conflicting data, it is thought that the influence of platelets on tumor proliferation is dependent upon the histological type and the microenvironment.
The above findings suggest that platelets are involved at each stage of cancer metastasis, from separation of cancer cells from the primary tumor to proliferation at the metastatic site. Therefore, inhibiting the platelet-cancer interaction by targeting platelets may lead to the development of novel therapeutic agents to treat cancer metastasis. Recently, there have been reports that aspirin significantly reduced the mortality rates of adenocarcinoma but did not inhibit the mortality rate for other types of cancer[112,113]. Furthermore, the mechanism of action regarding this purported anti-metastatic effect of aspirin can also be observed in cells other than cancer cells, and the increased bleeding tendency with an aspirin regimen is problematic.
In other words, it is important to target not platelets, which have critical physiological activities, but rather cancer-specific platelet activating factors, including integrins, cathepsin B, Necl-5, MMP2, GPIb and podoplanin, all of which are expressed on cancer cells.
This editorial suggests the current perspective of novel treatments for liver cirrhosis by using TPO-R agonists to increase platelet counts in a clinical setting.
There is significant evidence that platelets play a role in improving fibrosis. Upon their release, the ATP and ADP within platelet dense granules are degraded by HSCs into adenosine, which is incorporated into the HSCs.
There are three distinct mechanisms of liver regeneration induced by platelets: a cooperative effect with LSECs; a cooperative effect with Kupffer cells; and a direct effect on hepatocytes. Additionally, platelet transfusion improves liver function in patients with CLD and cirrhosis. Despite its safety and maintenance in increasing platelet counts, administration of the TPO-R agonist eltrombopag for 6 mo did not result in improvements of liver function in patients with CLD. Therefore, we are planning a new approach to develop novel strategies with TPO-R agonists and a desialylated formulation for treating liver diseases for which there are currently no effective treatments except transplantation. Of course, it is necessary to pay sufficient attention to the onset of thrombosis by excessively increasing platelets.
Editorial support, in the form of medical writing was provided by Cactus Communications.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Japan
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): E
P- Reviewer: Ikura Y, Liu ZW, Meyer J, Starlinger P S- Editor: Gong ZM L- Editor: Filipodia E- Editor: Wang CH
| 1. | Bataller R, Brenner DA. Liver fibrosis. J Clin Invest. 2005;115:209-218. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3381] [Cited by in F6Publishing: 3806] [Article Influence: 200.3] [Reference Citation Analysis (3)] |
| 2. | Friedman SL. Liver fibrosis -- from bench to bedside. J Hepatol. 2003;38 Suppl 1:S38-S53. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1199] [Cited by in F6Publishing: 1220] [Article Influence: 58.1] [Reference Citation Analysis (0)] |
| 3. | Jaeschke H. Cellular adhesion molecules: regulation and functional significance in the pathogenesis of liver diseases. Am J Physiol. 1997;273:G602-G611. [PubMed] [Cited in This Article: ] |
| 4. | Pinzani M, Marra F. Cytokine receptors and signaling in hepatic stellate cells. Semin Liver Dis. 2001;21:397-416. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 330] [Cited by in F6Publishing: 326] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 5. | Montalto G, Cervello M, Giannitrapani L, Dantona F, Terranova A, Castagnetta LA. Epidemiology, risk factors, and natural history of hepatocellular carcinoma. Ann N Y Acad Sci. 2002;963:13-20. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 203] [Cited by in F6Publishing: 220] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 6. | Tachi Y, Hirai T, Miyata A, Ohara K, Iida T, Ishizu Y, Honda T, Kuzuya T, Hayashi K, Ishigami M. Progressive fibrosis significantly correlates with hepatocellular carcinoma in patients with a sustained virological response. Hepatol Res. 2015;45:238-246. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 52] [Cited by in F6Publishing: 51] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 7. | Neuberger J, James O. Guidelines for selection of patients for liver transplantation in the era of donor-organ shortage. Lancet. 1999;354:1636-1639. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 134] [Cited by in F6Publishing: 141] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 8. | Guarrera JV, Emond JC. Advances in segmental liver transplantation: can we solve the donor shortage? Transplant Proc. 2001;33:3451-3455. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 9. | Taylor MC, Greig PD, Detsky AS, McLeod RS, Abdoh A, Krahn MD. Factors associated with the high cost of liver transplantation in adults. Can J Surg. 2002;45:425-434. [PubMed] [Cited in This Article: ] |
| 10. | Mueller AR, Platz KP, Kremer B. Early postoperative complications following liver transplantation. Best Pract Res Clin Gastroenterol. 2004;18:881-900. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 36] [Reference Citation Analysis (0)] |
| 11. | Porrett PM, Hsu J, Shaked A. Late surgical complications following liver transplantation. Liver Transpl. 2009;15 Suppl 2:S12-S18. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 46] [Cited by in F6Publishing: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 12. | Benyon RC, Iredale JP. Is liver fibrosis reversible? Gut. 2000;46:443-446. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 189] [Cited by in F6Publishing: 204] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 13. | Dufour JF, DeLellis R, Kaplan MM. Regression of hepatic fibrosis in hepatitis C with long-term interferon treatment. Dig Dis Sci. 1998;43:2573-2576. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 83] [Cited by in F6Publishing: 86] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 14. | Dufour JF, DeLellis R, Kaplan MM. Reversibility of hepatic fibrosis in autoimmune hepatitis. Ann Intern Med. 1997;127:981-985. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 224] [Cited by in F6Publishing: 202] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 15. | Iwamoto T, Terai S, Hisanaga T, Takami T, Yamamoto N, Watanabe S, Sakaida I. Bone-marrow-derived cells cultured in serum-free medium reduce liver fibrosis and improve liver function in carbon-tetrachloride-treated cirrhotic mice. Cell Tissue Res. 2013;351:487-495. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 16. | Ishikawa T, Ichida T, Matsuda Y, Sugitani S, Sugiyama M, Kato T, Miyazaki H, Asakura H. Reduced expression of thrombopoietin is involved in thrombocytopenia in human and rat liver cirrhosis. J Gastroenterol Hepatol. 1998;13:907-913. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 44] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 17. | Aster RH. Pooling of platelets in the spleen: role in the pathogenesis of “hypersplenic” thrombocytopenia. J Clin Invest. 1966;45:645-657. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 581] [Cited by in F6Publishing: 543] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 18. | Peck-Radosavljevic M. Thrombocytopenia in liver disease. Can J Gastroenterol. 2000;14 Suppl D:60D-66D. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 114] [Cited by in F6Publishing: 123] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 19. | Kajihara M, Okazaki Y, Kato S, Ishii H, Kawakami Y, Ikeda Y, Kuwana M. Evaluation of platelet kinetics in patients with liver cirrhosis: similarity to idiopathic thrombocytopenic purpura. J Gastroenterol Hepatol. 2007;22:112-118. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 44] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 20. | Matsuo R, Ohkohchi N, Murata S, Ikeda O, Nakano Y, Watanabe M, Hisakura K, Myronovych A, Kubota T, Narimatsu H. Platelets Strongly Induce Hepatocyte Proliferation with IGF-1 and HGF In Vitro. J Surg Res. 2008;145:279-286. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 91] [Cited by in F6Publishing: 83] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 21. | Nakamura T, Nawa K, Ichihara A, Kaise N, Nishino T. Purification and subunit structure of hepatocyte growth factor from rat platelets. FEBS Lett. 1987;224:311-316. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 417] [Cited by in F6Publishing: 428] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 22. | Harrison P, Cramer EM. Platelet alpha-granules. Blood Rev. 1993;7:52-62. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 389] [Cited by in F6Publishing: 380] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 23. | Diegelmann RF, Evans MC. Wound healing: an overview of acute, fibrotic and delayed healing. Front Biosci. 2004;9:283-289. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1313] [Cited by in F6Publishing: 1278] [Article Influence: 63.9] [Reference Citation Analysis (0)] |
| 24. | Gruber R, Baron M, Busenlechner D, Kandler B, Fuerst G, Watzek G. Proliferation and osteogenic differentiation of cells from cortical bone cylinders, bone particles from mill, and drilling dust. J Oral Maxillofac Surg. 2005;63:238-243. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 25. | Gerard D, Carlson ER, Gotcher JE, Jacobs M. Effects of platelet-rich plasma at the cellular level on healing of autologous bone-grafted mandibular defects in dogs. J Oral Maxillofac Surg. 2007;65:721-727. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 38] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 26. | Murata S, Ohkohchi N, Matsuo R, Ikeda O, Myronovych A, Hoshi R. Platelets promote liver regeneration in early period after hepatectomy in mice. World J Surg. 2007;31:808-816. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 128] [Cited by in F6Publishing: 131] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 27. | Murata S, Matsuo R, Ikeda O, Myronovych A, Watanabe M, Hisakura K, Nakano Y, Hashimoto I, Ohkohchi N. Platelets promote liver regeneration under conditions of Kupffer cell depletion after hepatectomy in mice. World J Surg. 2008;32:1088-1096. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 45] [Cited by in F6Publishing: 44] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 28. | Matsuo R, Nakano Y, Ohkohchi N. Platelet administration via the portal vein promotes liver regeneration in rats after 70% hepatectomy. Ann Surg. 2011;253:759-763. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 84] [Cited by in F6Publishing: 89] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 29. | Kawasaki T, Murata S, Takahashi K, Nozaki R, Ohshiro Y, Ikeda N, Pak S, Myronovych A, Hisakura K, Fukunaga K. Activation of human liver sinusoidal endothelial cell by human platelets induces hepatocyte proliferation. J Hepatol. 2010;53:648-654. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 73] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 30. | Murata S, Hashimoto I, Nakano Y, Myronovych A, Watanabe M, Ohkohchi N. Single administration of thrombopoietin prevents progression of liver fibrosis and promotes liver regeneration after partial hepatectomy in cirrhotic rats. Ann Surg. 2008;248:821-828. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 63] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 31. | Ikeda N, Murata S, Maruyama T, Tamura T, Nozaki R, Kawasaki T, Fukunaga K, Oda T, Sasaki R, Homma M. Platelet-derived adenosine 5’-triphosphate suppresses activation of human hepatic stellate cell: In vitro study. Hepatol Res. 2012;42:91-102. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 32. | Watanabe M, Murata S, Hashimoto I, Nakano Y, Ikeda O, Aoyagi Y, Matsuo R, Fukunaga K, Yasue H, Ohkohchi N. Platelets contribute to the reduction of liver fibrosis in mice. J Gastroenterol Hepatol. 2009;24:78-89. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 75] [Cited by in F6Publishing: 80] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 33. | Maruyama T, Murata S, Takahashi K, Tamura T, Nozaki R, Ikeda N, Fukunaga K, Oda T, Sasaki R, Ohkohchi N. Platelet transfusion improves liver function in patients with chronic liver disease and cirrhosis. Tohoku J Exp Med. 2013;229:213-220. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 34. | Murata K, Ito K, Yoneda K, Shiraki K, Sakurai H, Ito M. Splenectomy improves liver function in patients with liver cirrhosis. Hepatogastroenterology. 2008;55:1407-1411. [PubMed] [Cited in This Article: ] |
| 35. | Ushitora Y, Tashiro H, Takahashi S, Amano H, Oshita A, Kobayashi T, Chayama K, Ohdan H. Splenectomy in chronic hepatic disorders: portal vein thrombosis and improvement of liver function. Dig Surg. 2011;28:9-14. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 36. | Gressner AM, Weiskirchen R. Modern pathogenetic concepts of liver fibrosis suggest stellate cells and TGF-beta as major players and therapeutic targets. J Cell Mol Med. 2006;10:76-99. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 538] [Cited by in F6Publishing: 599] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 37. | Friedman SL. Seminars in medicine of the Beth Israel Hospital, Boston. The cellular basis of hepatic fibrosis. Mechanisms and treatment strategies. N Engl J Med. 1993;328:1828-1835. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 793] [Cited by in F6Publishing: 876] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 38. | Friedman SL, Maher JJ, Bissell DM. Mechanisms and therapy of hepatic fibrosis: report of the AASLD Single Topic Basic Research Conference. Hepatology. 2000;32:1403-1408. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 109] [Cited by in F6Publishing: 114] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 39. | Friedman SL. Mechanisms of hepatic fibrogenesis. Gastroenterology. 2008;134:1655-1669. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1928] [Cited by in F6Publishing: 2053] [Article Influence: 128.3] [Reference Citation Analysis (0)] |
| 40. | Parsons CJ, Takashima M, Rippe RA. Molecular mechanisms of hepatic fibrogenesis. J Gastroenterol Hepatol. 2007;22 Suppl 1:S79-S84. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 216] [Cited by in F6Publishing: 230] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 41. | Kodama T, Takehara T, Hikita H, Shimizu S, Li W, Miyagi T, Hosui A, Tatsumi T, Ishida H, Tadokoro S. Thrombocytopenia exacerbates cholestasis-induced liver fibrosis in mice. Gastroenterology. 2010;138:2487-298, 2487-298. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 96] [Cited by in F6Publishing: 99] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 42. | Xia JL, Dai C, Michalopoulos GK, Liu Y. Hepatocyte growth factor attenuates liver fibrosis induced by bile duct ligation. Am J Pathol. 2006;168:1500-1512. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 159] [Cited by in F6Publishing: 160] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 43. | Nakamura T, Nishizawa T, Hagiya M, Seki T, Shimonishi M, Sugimura A, Tashiro K, Shimizu S. Molecular cloning and expression of human hepatocyte growth factor. Nature. 1989;342:440-443. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1628] [Cited by in F6Publishing: 1606] [Article Influence: 45.9] [Reference Citation Analysis (0)] |
| 44. | Wolber EM, Jelkmann W. Thrombopoietin: the novel hepatic hormone. News Physiol Sci. 2002;17:6-10. [PubMed] [Cited in This Article: ] |
| 45. | Cheng G. Eltrombopag, a thrombopoietin- receptor agonist in the treatment of adult chronic immune thrombocytopenia: a review of the efficacy and safety profile. Ther Adv Hematol. 2012;3:155-164. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 46. | Cooper KL, Fitzgerald P, Dillingham K, Helme K, Akehurst R. Romiplostim and eltrombopag for immune thrombocytopenia: methods for indirect comparison. Int J Technol Assess Health Care. 2012;28:249-258. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 26] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 47. | McHutchison JG, Dusheiko G, Shiffman ML, Rodriguez-Torres M, Sigal S, Bourliere M, Berg T, Gordon SC, Campbell FM, Theodore D. Eltrombopag for thrombocytopenia in patients with cirrhosis associated with hepatitis C. N Engl J Med. 2007;357:2227-2236. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 336] [Cited by in F6Publishing: 352] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 48. | Afdhal NH, Giannini EG, Tayyab G, Mohsin A, Lee JW, Andriulli A, Jeffers L, McHutchison J, Chen PJ, Han KH. Eltrombopag before procedures in patients with cirrhosis and thrombocytopenia. N Engl J Med. 2012;367:716-724. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 239] [Cited by in F6Publishing: 222] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 49. | Afdhal N, McHutchison J, Brown R, Jacobson I, Manns M, Poordad F, Weksler B, Esteban R. Thrombocytopenia associated with chronic liver disease. J Hepatol. 2008;48:1000-1007. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 380] [Cited by in F6Publishing: 372] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 50. | Kawaguchi T, Komori A, Seike M, Fujiyama S, Watanabe H, Tanaka M, Sakisaka S, Nakamuta M, Sasaki Y, Oketani M. Efficacy and safety of eltrombopag in Japanese patients with chronic liver disease and thrombocytopenia: a randomized, open-label, phase II study. J Gastroenterol. 2012;47:1342-1351. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 51. | Takahashi K, Murata S, Fukunaga K, Ohkohchi N. Human platelets inhibit liver fibrosis in severe combined immunodeficiency mice. World J Gastroenterol. 2013;19:5250-5260. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 22] [Cited by in F6Publishing: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 52. | Malik R, Selden C, Hodgson H. The role of non-parenchymal cells in liver growth. Semin Cell Dev Biol. 2002;13:425-431. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 102] [Cited by in F6Publishing: 108] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 53. | FitzGerald MJ, Webber EM, Donovan JR, Fausto N. Rapid DNA binding by nuclear factor kappa B in hepatocytes at the start of liver regeneration. Cell Growth Differ. 1995;6:417-427. [PubMed] [Cited in This Article: ] |
| 54. | Cressman DE, Greenbaum LE, Haber BA, Taub R. Rapid activation of post-hepatectomy factor/nuclear factor kappa B in hepatocytes, a primary response in the regenerating liver. J Biol Chem. 1994;269:30429-30435. [PubMed] [Cited in This Article: ] |
| 55. | Stepniak E, Ricci R, Eferl R, Sumara G, Sumara I, Rath M, Hui L, Wagner EF. c-Jun/AP-1 controls liver regeneration by repressing p53/p21 and p38 MAPK activity. Genes Dev. 2006;20:2306-2314. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 161] [Cited by in F6Publishing: 170] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 56. | Wang GL, Salisbury E, Shi X, Timchenko L, Medrano EE, Timchenko NA. HDAC1 promotes liver proliferation in young mice via interactions with C/EBPbeta. J Biol Chem. 2008;283:26179-26187. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 48] [Cited by in F6Publishing: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 57. | Factor VM, Seo D, Ishikawa T, Kaposi-Novak P, Marquardt JU, Andersen JB, Conner EA, Thorgeirsson SS. Loss of c-Met disrupts gene expression program required for G2/M progression during liver regeneration in mice. PLoS One. 2010;5. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 54] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 58. | Jackson LN, Larson SD, Silva SR, Rychahou PG, Chen LA, Qiu S, Rajaraman S, Evers BM. PI3K/Akt activation is critical for early hepatic regeneration after partial hepatectomy. Am J Physiol Gastrointest Liver Physiol. 2008;294:G1401-G1410. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 71] [Cited by in F6Publishing: 72] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 59. | Knook DL, Sleyster EC. Separation of Kupffer and endothelial cells of the rat liver by centrifugal elutriation. Exp Cell Res. 1976;99:444-449. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 295] [Cited by in F6Publishing: 300] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 60. | Hisakura K, Murata S, Takahashi K, Matsuo R, Pak S, Ikeda N, Kawasaki T, Kohno K, Myronovych A, Nakano Y. Platelets prevent acute hepatitis induced by anti-fas antibody. J Gastroenterol Hepatol. 2011;26:348-355. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 61. | Braet F, Wisse E. Structural and functional aspects of liver sinusoidal endothelial cell fenestrae: a review. Comp Hepatol. 2002;1:1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 539] [Cited by in F6Publishing: 483] [Article Influence: 22.0] [Reference Citation Analysis (1)] |
| 62. | Rai RM, Yang SQ, McClain C, Karp CL, Klein AS, Diehl AM. Kupffer cell depletion by gadolinium chloride enhances liver regeneration after partial hepatectomy in rats. Am J Physiol. 1996;270:G909-G918. [PubMed] [Cited in This Article: ] |
| 63. | Badia JM, Ayton LC, Evans TJ, Carpenter AJ, Nawfal G, Kinderman H, Zografos G, Uemoto S, Cohen J, Habib NA. Systemic cytokine response to hepatic resections under total vascular exclusion. Eur J Surg. 1998;164:185-190. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 28] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 64. | Gauldie J, Richards C, Baumann H. IL6 and the acute phase reaction. Res Immunol. 1992;143:755-759. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 57] [Cited by in F6Publishing: 58] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 65. | Yatomi Y, Ohmori T, Rile G, Kazama F, Okamoto H, Sano T, Satoh K, Kume S, Tigyi G, Igarashi Y. Sphingosine 1-phosphate as a major bioactive lysophospholipid that is released from platelets and interacts with endothelial cells. Blood. 2000;96:3431-3438. [PubMed] [Cited in This Article: ] |
| 66. | Takuwa Y, Okamoto Y, Yoshioka K, Takuwa N. Sphingosine-1-phosphate signaling and biological activities in the cardiovascular system. Biochim Biophys Acta. 2008;1781:483-488. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 105] [Cited by in F6Publishing: 110] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 67. | Bilzer M, Roggel F, Gerbes AL. Role of Kupffer cells in host defense and liver disease. Liver Int. 2006;26:1175-1186. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 586] [Cited by in F6Publishing: 576] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 68. | Meijer C, Wiezer MJ, Diehl AM, Schouten HJ, Schouten HJ, Meijer S, van Rooijen N, van Lambalgen AA, Dijkstra CD, van Leeuwen PA. Kupffer cell depletion by CI2MDP-liposomes alters hepatic cytokine expression and delays liver regeneration after partial hepatectomy. Liver. 2000;20:66-77. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 143] [Cited by in F6Publishing: 149] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 69. | Akerman P, Cote P, Yang SQ, McClain C, Nelson S, Bagby GJ, Diehl AM. Antibodies to tumor necrosis factor-alpha inhibit liver regeneration after partial hepatectomy. Am J Physiol. 1992;263:G579-G585. [PubMed] [Cited in This Article: ] |
| 70. | Yamada Y, Webber EM, Kirillova I, Peschon JJ, Fausto N. Analysis of liver regeneration in mice lacking type 1 or type 2 tumor necrosis factor receptor: requirement for type 1 but not type 2 receptor. Hepatology. 1998;28:959-970. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 212] [Cited by in F6Publishing: 217] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 71. | Yamada Y, Kirillova I, Peschon JJ, Fausto N. Initiation of liver growth by tumor necrosis factor: deficient liver regeneration in mice lacking type I tumor necrosis factor receptor. Proc Natl Acad Sci USA. 1997;94:1441-1446. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 740] [Cited by in F6Publishing: 733] [Article Influence: 27.1] [Reference Citation Analysis (0)] |
| 72. | Abshagen K, Eipel C, Kalff JC, Menger MD, Vollmar B. Loss of NF-kappaB activation in Kupffer cell-depleted mice impairs liver regeneration after partial hepatectomy. Am J Physiol Gastrointest Liver Physiol. 2007;292:G1570-G1577. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 76] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 73. | Sindram D, Porte RJ, Hoffman MR, Bentley RC, Clavien PA. Synergism between platelets and leukocytes in inducing endothelial cell apoptosis in the cold ischemic rat liver: a Kupffer cell-mediated injury. FASEB J. 2001;15:1230-1232. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 77] [Cited by in F6Publishing: 79] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 74. | Nakano Y, Kondo T, Matsuo R, Hashimoto I, Kawasaki T, Kohno K, Myronovych A, Tadano S, Hisakura K, Ikeda O. Platelet dynamics in the early phase of postischemic liver in vivo. J Surg Res. 2008;149:192-198. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 43] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 75. | Tamura T, Kondo T, Pak S, Nakano Y, Murata S, Fukunaga K, Ohkohchi N. Interaction between Kupffer cells and platelets in the early period of hepatic ischemia-reperfusion injury--an in vivo study. J Surg Res. 2012;178:443-451. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 76. | Nakamura M, Shibazaki M, Nitta Y, Endo Y. Translocation of platelets into Disse spaces and their entry into hepatocytes in response to lipopolysaccharides, interleukin-1 and tumour necrosis factor: the role of Kupffer cells. J Hepatol. 1998;28:991-999. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 50] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 77. | Myronovych A, Murata S, Chiba M, Matsuo R, Ikeda O, Watanabe M, Hisakura K, Nakano Y, Kohno K, Kawasaki T. Role of platelets on liver regeneration after 90% hepatectomy in mice. J Hepatol. 2008;49:363-372. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 79] [Cited by in F6Publishing: 87] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 78. | Kirschbaum M, Karimian G, Adelmeijer J, Giepmans BN, Porte RJ, Lisman T. Horizontal RNA transfer mediates platelet-induced hepatocyte proliferation. Blood. 2015;126:798-806. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 79. | Meyer J, Lejmi E, Fontana P, Morel P, Gonelle-Gispert C, Bühler L. A focus on the role of platelets in liver regeneration: Do platelet-endothelial cell interactions initiate the regenerative process? J Hepatol. 2015;63:1263-1271. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 58] [Cited by in F6Publishing: 62] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 80. | Lisman T, Kirschbaum M, Porte RJ. The role of platelets in liver regeneration - What don’t we know? J Hepatol. 2015;63:1537-1538. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 81. | Stroncek DF, Rebulla P. Platelet transfusions. Lancet. 2007;370:427-438. [Cited in This Article: ] |
| 82. | Matsunaga T, Tanaka I, Kobune M, Kawano Y, Tanaka M, Kuribayashi K, Iyama S, Sato T, Sato Y, Takimoto R. Ex vivo large-scale generation of human platelets from cord blood CD34 cells. Stem Cells. 2006;24:2877-2887. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 93] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 83. | Neildez-Nguyen TMA, Wajcman H, Marden MC, Bensidhoum M, Moncollin V, Giarratana MC, Kobari L, Thierry D, Douay L. Human erythroid cells produced ex vivo at large scale differentiate into red blood cells in vivo. Nat Biotechnol. 2002;20:467-472. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 203] [Cited by in F6Publishing: 203] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 84. | Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861-872. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14327] [Cited by in F6Publishing: 13579] [Article Influence: 848.7] [Reference Citation Analysis (0)] |
| 85. | Van der Meer PF, Pietersz RNI. Gamma irradiation does not affect 7-day storage of platelet concentrates. Vox Sang. 2005;89:97-99. [Cited in This Article: ] |
| 86. | Ono-Uruga Y, Tozawa K, Horiuchi T, Murata M, Okamoto S, Ikeda Y, Suda T, Matsubara Y. Human adipose tissue-derived stromal cells can differentiate into megakaryocytes and platelets by secreting endogenous thrombopoietin. J Thromb Haemost. 2016;14:1285-1297. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 87. | Stamou KM, Toutouzas KG, Kekis PB, Nakos S, Gafou A, Manouras A, Krespis E, Katsaragakis S, Bramis J. Prospective study of the incidence and risk factors of postsplenectomy thrombosis of the portal, mesenteric, and splenic veins. Arch Surg. 2006;141:663-669. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 134] [Cited by in F6Publishing: 147] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 88. | Kojouri K, Vesely SK, Terrell DR, George JN. Splenectomy for adult patients with idiopathic thrombocytopenic purpura: a systematic review to assess long-term platelet count responses, prediction of response, and surgical complications. Blood. 2004;104:2623-2634. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 417] [Cited by in F6Publishing: 414] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 89. | Chambers LA, Kruskall MS, Pacini DG, Donovan LM. Febrile reactions after platelet transfusion: the effect of single versus multiple donors. Transfusion. 1990;30:219-221. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 79] [Cited by in F6Publishing: 79] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 90. | Khan SY, Kelher MR, Heal JM, Blumberg N, Boshkov LK, Phipps R, Gettings KF, McLaughlin NJ, Silliman CC. Soluble CD40 ligand accumulates in stored blood components, primes neutrophils through CD40, and is a potential cofactor in the development of transfusion-related acute lung injury. Blood. 2006;108:2455-2462. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 305] [Cited by in F6Publishing: 325] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 91. | Kuter DJ. New thrombopoietic growth factors. Blood. 2007;109:4607-4616. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 251] [Cited by in F6Publishing: 229] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 92. | Erhardt JA, Erickson-Miller CL, Aivado M, Abboud M, Pillarisetti K, Toomey JR. Comparative analyses of the small molecule thrombopoietin receptor agonist eltrombopag and thrombopoietin on in vitro platelet function. Exp Hematol. 2009;37:1030-1037. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 79] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 93. | Erickson-Miller CL, Kirchner J, Aivado M, May R, Payne P, Chadderton A. Reduced proliferation of non-megakaryocytic acute myelogenous leukemia and other leukemia and lymphoma cell lines in response to eltrombopag. Leuk Res. 2010;34:1224-1231. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 94. | Nozaki R, Murata S, Nowatari T, Maruyama T, Ikeda N, Kawasaki T, Fukunaga K, Ohkohchi N. Effects of thrombopoietin on growth of hepatocellular carcinoma: Is thrombopoietin therapy for liver disease safe or not? Hepatol Res. 2013;43:610-620. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 95. | Kurokawa T, Murata S, Zheng YW, Iwasaki K, Kohno K, Fukunaga K, Ohkohchi N. The Eltrombopag antitumor effect on hepatocellular carcinoma. Int J Oncol. 2015;47:1696-1702. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 96. | Grozovsky R, Begonja AJ, Liu K, Visner G, Hartwig JH, Falet H, Hoffmeister KM. The Ashwell-Morell receptor regulates hepatic thrombopoietin production via JAK2-STAT3 signaling. Nat Med. 2015;21:47-54. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 193] [Cited by in F6Publishing: 200] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 97. | Trousseau A. Phlegmasia alba dolens. Clin Med Hotel Dieu De Paris. 1865;3:94. [Cited in This Article: ] |
| 98. | Gay LJ, Felding-Habermann B. Contribution of platelets to tumour metastasis. Nat Rev Cancer. 2011;11:123-134. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1087] [Cited by in F6Publishing: 1164] [Article Influence: 89.5] [Reference Citation Analysis (0)] |
| 99. | Labelle M, Begum S, Hynes RO. Direct signaling between platelets and cancer cells induces an epithelial-mesenchymal-like transition and promotes metastasis. Cancer Cell. 2011;20:576-590. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1180] [Cited by in F6Publishing: 1274] [Article Influence: 98.0] [Reference Citation Analysis (0)] |
| 100. | Egan K, Cooke N, Kenny D. Living in shear: platelets protect cancer cells from shear induced damage. Clin Exp Metastasis. 2014;31:697-704. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 94] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 101. | Nieswandt B, Hafner M, Echtenacher B, Männel DN. Lysis of tumor cells by natural killer cells in mice is impeded by platelets. Cancer Res. 1999;59:1295-1300. [PubMed] [Cited in This Article: ] |
| 102. | Placke T, Örgel M, Schaller M, Jung G, Rammensee HG, Kopp HG, Salih HR. Platelet-derived MHC class I confers a pseudonormal phenotype to cancer cells that subverts the antitumor reactivity of natural killer immune cells. Cancer Res. 2012;72:440-448. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 231] [Cited by in F6Publishing: 275] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 103. | Kim YJ, Borsig L, Varki NM, Varki A. P-selectin deficiency attenuates tumor growth and metastasis. Proc Natl Acad Sci USA. 1998;95:9325-9330. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 340] [Cited by in F6Publishing: 329] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 104. | Borsig L, Wong R, Feramisco J, Nadeau DR, Varki NM, Varki A. Heparin and cancer revisited: mechanistic connections involving platelets, P-selectin, carcinoma mucins, and tumor metastasis. Proc Natl Acad Sci USA. 2001;98:3352-3357. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 506] [Cited by in F6Publishing: 501] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 105. | Jain S, Zuka M, Liu J, Russell S, Dent J, Guerrero JA, Forsyth J, Maruszak B, Gartner TK, Felding-Habermann B. Platelet glycoprotein Ib alpha supports experimental lung metastasis. Proc Natl Acad Sci USA. 2007;104:9024-9028. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 139] [Cited by in F6Publishing: 142] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 106. | Mammadova-Bach E, Mangin P, Lanza F, Gachet C. Platelets in cancer. From basic research to therapeutic implications. Hamostaseologie. 2015;35:325-336. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 107. | Guerrero JA, Bennett C, van der Weyden L, McKinney H, Chin M, Nurden P, McIntyre Z, Cambridge EL, Estabel J, Wardle-Jones H. Gray platelet syndrome: proinflammatory megakaryocytes and α-granule loss cause myelofibrosis and confer metastasis resistance in mice. Blood. 2014;124:3624-3635. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 68] [Cited by in F6Publishing: 72] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 108. | Mikami J, Kurokawa Y, Takahashi T, Miyazaki Y, Yamasaki M, Miyata H, Nakajima K, Takiguchi S, Mori M, Doki Y. Antitumor effect of antiplatelet agents in gastric cancer cells: an in vivo and in vitro study. Gastric Cancer. 2016;19:817-826. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 109. | Yuan L, Liu X. Platelets are associated with xenograft tumor growth and the clinical malignancy of ovarian cancer through an angiogenesis-dependent mechanism. Mol Med Rep. 2015;11:2449-2458. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 110. | Cho MS, Bottsford-Miller J, Vasquez HG, Stone R, Zand B, Kroll MH, Sood AK, Afshar-Kharghan V. Platelets increase the proliferation of ovarian cancer cells. Blood. 2012;120:4869-4872. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 152] [Cited by in F6Publishing: 172] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 111. | Li R, Ren M, Chen N, Luo M, Deng X, Xia J, Yu G, Liu J, He B, Zhang X. Presence of intratumoral platelets is associated with tumor vessel structure and metastasis. BMC Cancer. 2014;14:167. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 59] [Cited by in F6Publishing: 68] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 112. | Rothwell PM, Price JF, Fowkes FG, Zanchetti A, Roncaglioni MC, Tognoni G, Lee R, Belch JF, Wilson M, Mehta Z. Short-term effects of daily aspirin on cancer incidence, mortality, and non-vascular death: analysis of the time course of risks and benefits in 51 randomised controlled trials. Lancet. 2012;379:1602-1612. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 622] [Cited by in F6Publishing: 613] [Article Influence: 51.1] [Reference Citation Analysis (0)] |
| 113. | Rothwell PM, Wilson M, Price JF, Belch JF, Meade TW, Mehta Z. Effect of daily aspirin on risk of cancer metastasis: a study of incident cancers during randomised controlled trials. Lancet. 2012;379:1591-1601. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 698] [Cited by in F6Publishing: 707] [Article Influence: 58.9] [Reference Citation Analysis (0)] |