Helical-Ribbon and Tape Formation of Lipid Packaged [Ru(bpy)3]2+ Complexes in Organic Media
Abstract
:1. Introduction
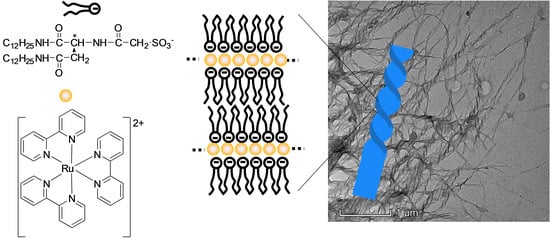
2. Results and Discussion
3. Materials and Instrumentation
3.1. Synthesis of Amphiphilic Lipids 1–6 [20,61,62,63,64,65,66,67,68]
3.1.1. Synthesis of N-Benzyloxycarbonyl-L-Aspartic Acid (1a), N-Benzyloxycarbonyl-L-Glutamic Acid (2a)
3.1.2. Synthesis of L-2-N-(Benzyloxycarbonylamino) Adipic Acid (3a)
3.1.3. Synthesis of N’,N”-Didodecyl-Nα-Benzyloxycarbonyl-L-Aspartamide (1b), N’,N”-Didodecyl-Nα-Benzyloxycarbonyl-L-Glutamide (2b), and N’,N”-Didodecyl- L-2-Nα-(benzyloxycarbonylamino) Adipamide (3b)
3.1.4. Synthesis of N’,N”-Didodecyl-L-Aspartamide (1c), N’,N”-Didodecyl-L-Glutamide (2c), and N’,N”-Didodecyl-L-2- Aminoadipamide (3c)
3.1.5. Synthesis of N’,N”-Didodecyl-Nα-Sulfoacetyl-L-Aspartamide Sodium Salt (1(Na)), N’,N”-Didodecyl- Nα-Sulfoacetyl-L-Glutamide Sodium Salt (2(Na)), N’,N”-Didodecyl- L-2-Nα-(Sulfoacetylamino) Adipamide Sodium Salt (3(Na))
3.1.6. Synthesis of N’,N”-Didodecyl-Nα-4-Sulfobenzoyl-L-Aspartamide Sodium Salt (4(Na)), N’,N”-Didodecyl- Nα-4-Sulfobenzoyl-L-Glutamide Sodium Salt (5(Na)), N’,N”-Didodecyl- L-2-Nα-(4-Sulfobenzoylamino) Adipamide Sodium Salt (6(Na))
3.2. General Preparation of [RuII(bpy)3](Lipid)2 [14]
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Balzoni, V.; Scandola, F. Supramolecular Photochemistry; Ellis Horwood: Chichester, UK, 1991; pp. 1–300. ISBN 9780138775315. [Google Scholar]
- Kalyanasundaram, K. Photochemistry of Polypyridine and Porphyrin Complexes; Academic Press: London, UK, 1992; pp. 1–624. ISBN 9780123949929. [Google Scholar]
- Kane-Maguire, N.A.P. Photochemistry and Photophysics of Coordination Compounds: Chromium. Top. Curr. Chem. 2007, 280, 37–67. [Google Scholar]
- Kumaresan, D.; Shankar, K.; Vaidya, S.; Schmehl, R.H. Photochemistry and Photophysics of Coordination Compounds: Osmium. Top. Curr. Chem. 2007, 281, 101–142. [Google Scholar]
- Indelli, M.T.; Chiorboli, C.; Scandola, F. Photochemistry and Photophysics of Coordination Compounds: Rhodium. Top. Curr. Chem. 2007, 280, 215–255. [Google Scholar]
- Kirk, A.D. Photochemistry and Photophysics of Chromium(III) Complexes. Chem. Rev. 1999, 99, 1607–1640. [Google Scholar] [CrossRef] [PubMed]
- Wrighton, M.S. The Photochemistry of Metal Carbonyls. Chem. Rev. 1974, 74, 401–430. [Google Scholar] [CrossRef]
- Demas, J.N.; Adamson, A.W. A New Photosensitizer. Tris(2,2‘-bipyridine)ruthenium(II) Chloride. J. Am. Chem. Soc. 1971, 93, 1800–1801. [Google Scholar]
- Kalyanasundaram, K. Photophysics, photochemistry and solar energy conversion with tris(bipyridyl)ruthenium(II) and its analogues. Coord. Chem. Rev. 1982, 46, 159–244. [Google Scholar] [CrossRef]
- Hara, M.; Waraksa, C.C.; Lean, J.T.; Lewis, B.A.; Mallouk, T.E. Photocatalytic Water Oxidation in a Buffered Tris(2,2′-bipyridyl)ruthenium Complex-Colloidal IrO2 System. J. Phys. Chem. A 2000, 104, 5275–5280. [Google Scholar] [CrossRef]
- Teply, F. Photoredox catalysis by [Ru(bpy)3]2+ to trigger transformations of organic molecules. Organic synthesis using visible-light photocatalysis and its 20th century roots. Collect. Czech. Chem. Commun. 2011, 76, 859–917. [Google Scholar] [CrossRef]
- Nicewicz, D.A.; MacMillan, D.W.C. Merging Photoredox Catalysis with Organocatalysis: The Direct Asymmetric Alkylation of Aldehydes. Science 2008, 322, 77–80. [Google Scholar] [CrossRef] [Green Version]
- Ischay, M.A.; Anzovino, M.E.; Du, J.; Yoon, T.P. Efficient Visible Light Photocatalysis of [2+2] Enone Cycloadditions. J. Am. Chem. Soc. 2008, 130, 12886–12887. [Google Scholar] [CrossRef] [PubMed]
- Narayanam, J.M.R.; Tucker, J.W.; Stephenson, C.R.J. Electron-Transfer Photoredox Catalysis: Development of a Tin-Free Reductive Dehalogenation Reaction. J. Am. Chem. Soc. 2009, 131, 8756–8757. [Google Scholar] [CrossRef] [PubMed]
- Narayanam, J.M.R.; Stephenson, C.R.J. Visible light photoredox catalysis: Applications in organic synthesis. Chem. Soc. Rev. 2011, 40, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Zeitler, K. Photoredox Catalysis with Visible Light. Angew. Chem. Int. Ed. 2009, 48, 9785–9789. [Google Scholar] [CrossRef] [PubMed]
- Neumann, M.; Füldner, S.; König, B.; Zeitler, K. Metal-Free, Cooperative Asymmetric Organophotoredox Catalysis with Visible Light. Angew. Chem. Int. Ed. 2011, 50, 951–954. [Google Scholar] [CrossRef] [PubMed]
- Juris, A.; Balzani, V.; Barigellitti, F.; Campagna, S.; Belser, P.; von Zelewsky, A. Ru(II) Polypyridine Complexes: Photophysics, Photochemistry, Electrochemistry, and Chemiluminescence. Coord. Chem. Rev. 1988, 84, 85–277. [Google Scholar] [CrossRef]
- Campagna, S.; Puntoriero, F.; Nastasi, F.; Bergamini, G.; Balzani, V. Photochemistry and Photophysics of Coordination Compounds: Ruthenium. Top. Curr. Chem. 2007, 280, 177–214. [Google Scholar]
- Kuroiwa, K.; Yoshida, M.; Masaoka, S.; Kaneko, K.; Sakai, K.; Kimizuka, N. Self-assembly of Tubular Microstructures from Mixed-valence Metal Complexes and their Reversible Transformation via External Stimuli. Angew. Chem. Int. Ed. 2012, 51, 656–659. [Google Scholar] [CrossRef]
- Fuhrhop, J.-H.; Demoulin, C.; Boettcher, C.; Koning, J.; Siggel, U. Chiral Micellar Porphyrin Fibers with 2-Aminoglycosamide Head Groups. J. Am. Chem. Soc. 1992, 114, 4159–4165. [Google Scholar] [CrossRef]
- Fuhrhop, J.-H.; Bindig, U.; Siggel, U. Micellar Rods and Vesicular Tubules Made of 14”‘, 16”‘-Diaminoporphyrins. J. Am. Chem. Soc. 1993, 115, 11036–11037. [Google Scholar] [CrossRef]
- Bingid, U.; Schulz, A.; Fuhrhop, J.-H. Micellar Fibers Made Of Porphyrin And Metalloporphyrin Amides. New J. Chem. 1995, 19, 427–435. [Google Scholar]
- Fuhrhop, J.-H.; Bindig, U.; Demoulin, C.; Rosengarten, B. Polymeric Porphyrin and Metalloporphyrin Assemblies In Bulk Solution. Macromol. Symp. 1994, 80, 63–82. [Google Scholar] [CrossRef]
- Shirakawa, M.; Fujita, N.; Shinkai, S. A Stable Single Piece of Unimolecularly π-Stacked Porphyrin Aggregate in a Thixotropic Low Molecular Weight Gel: A One-Dimensional Molecular Template for Polydiacetylene Wiring up to Several Tens of Micrometers in Length. J. Am. Chem. Soc. 2005, 127, 4164–4165. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Zhang, X.; Gai, X.; Jiang, J. Tuning the morphology of self-assembled nanostructures of amphiphilic tetra(p-hydroxyphenyl)porphyrins with hydrogen bonding and metal–ligand coordination bonding. J. Mater. Chem. 2009, 19, 2417–2424. [Google Scholar] [CrossRef]
- Engelkamp, H.; Middelbeek, S.; Nolte, R.J.M. Self-Assembly of Disk-Shaped Molecules to Coiled-Coil Aggregates with Tunable Helicity. Science 1999, 284, 785–788. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.; Muto, T.; Takimoto, H.; Wada, K.; Ohta, K.; Hanabusa, K.; Shirai, H.; Kobayashi, N. Fibrous Assemblies Made of Amphiphilic Metallophthalocyanines. Langmuir 2000, 16, 2078–2082. [Google Scholar] [CrossRef]
- Rai, R.; Saxena, A.; Ohira, A.; Fujiki, M. Programmed Hyperhelical Supramolecular Assembly of Nickel Phthalocyanine Bearing Enantiopure 1-(p-Tolyl)ethylaminocarbonyl Groups. Langmuir 2005, 21, 3957–3962. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Ma, X.; Liu, M. Optically Active Phthalocyaninato-Polysiloxane Constructed from Achiral Monomers: From Noncovalent Assembly to Covalent Polymer. Macromolecules 2007, 40, 4780–4784. [Google Scholar] [CrossRef]
- Wu, L.; Wang, Q.; Lu, J.; Bian, Y.; Jiang, J.; Zhang, X. Helical Nanostructures Self-Assembled from Optically Active Phthalocyanine Derivatives Bearing Four Optically Active Binaphthyl Moieties: Effect of Metal-Ligand Coordination on the Morphology, Dimension, and Helical Pitch of Self-Assembled Nanostructures. Langmuir 2010, 26, 7489–7497. [Google Scholar] [CrossRef]
- Zhang, W.; Ishimaru, A.; Onouchi, H.; Rai, R.; Saxena, A.; Ohira, A.; Ishikawa, M.; Naito, M.; Fujiki, M. Ambidextrous optically active copper(II) phthalocyanine supramolecules induced by peripheral group homochirality. New J. Chem. 2010, 34, 2310–2318. [Google Scholar] [CrossRef]
- Zhang, W.; Fujiki, M.; Zhu, X. Chiroptical Nanofibers Generated from Achiral Metallophthalocyanines Induced by Diamine Homochirality. Chem. Eur. J. 2011, 17, 10628–10635. [Google Scholar] [CrossRef] [PubMed]
- Garifullin, R.; Erkal, T.S.; Tekin, S.; Ortaç, B.; Gürek, A.G.; Ahsen, V.; Yaglioglu, H.G.; Elmali, A.; Guler, M.O. Encapsulation of a zinc phthalocyanine derivative in self-assembled peptide nanofibers. J. Mater. Chem. 2012, 22, 2553–2559. [Google Scholar] [CrossRef]
- Lu, J.; Wang, S.; Tian, J. Helical nano-fibers of an optical active phthalocyaninato zinc complex: Chiral information transfer, I–V properties and nonlinear optical properties. Inorg. Chem. Commun. 2015, 57, 36–39. [Google Scholar] [CrossRef]
- Hui, J.K.-H.; Yu, Z.; MacLachlan, M.J. Supramolecular Assembly of Zinc Salphen Complexes: Access to Metal-Containing Gels and Nanofibers. Angew. Chem. Int. Ed. 2007, 46, 7980–7983. [Google Scholar] [CrossRef] [PubMed]
- Hui, J.K.-H.; Yu, Z.; Mirfakhrai, T.; MacLachlan, M.J. Supramolecular Assembly of Carbohydrate-Functionalized Salphen–Metal Complexes. Chem. Eur. J. 2009, 15, 13456–13465. [Google Scholar] [CrossRef] [PubMed]
- Po, C.; Tam, A.Y.-Y.; Wong, K.M.-C.; Yam, V.W.-W. Supramolecular Self-Assembly of Amphiphilic Anionic Platinum(II) Complexes: A Correlation between Spectroscopic and Morphological Properties. J. Am. Chem. Soc. 2011, 133, 12136–12143. [Google Scholar] [CrossRef]
- Wang, J.; Chen, Y.; Law, Y.-C.; Li, M.; Zhu, M.-X.; Lu, W.; Chui, S.S. –Y.; Zhu, N.; Che, C.-M. Organo- and Hydrogelators Based on Luminescent Monocationic Terpyridyl Platinum(II) Complexes with Biphenylacetylide Ligands. Chem. Asian J. 2011, 6, 3011–3019. [Google Scholar] [CrossRef]
- Strassert, C.A.; Chien, C.-H.; Lopez, M.D.G.; Kourkoulos, D.; Hertel, D.; Meerholz, K.; Cola, L.D. Switching On Luminescence by the Self-Assembly of a Platinum(II) Complex into Gelating Nanofibers and Electroluminescent Films. Angew. Chem. Int. Ed. 2011, 50, 946–950. [Google Scholar] [CrossRef]
- Leung, S.Y.-L.; Yam, V.W.-W. Chem. Sci., 4(11), 4228–4234 (2013); Po, C.; Tam, A.Y. –Y.; Yam, V.W. –W.; Tuning of spectroscopic properties via variation of the alkyl chain length: A systematic study of molecular structural changes on self-assembly of amphiphilic sulfonate-pendant platinum(II) bzimpy complexes in aqueous medium. Chem. Sci. 2014, 5, 2688–2695. [Google Scholar]
- Chung, C.Y.-S.; Tamaru, S.-i.; Shinkai, S.; Yam, V.W.-W. Supramolecular Assembly of Achiral Alkynylplatinum(II) Complexes and Carboxylic b-1,3-Glucan into Different Helical Handedness Stabilized by Pt···Pt and/or π–π Interactions. Chem. Eur. J. 2015, 21, 5447–5458. [Google Scholar] [CrossRef]
- Li, Y.; Wong, K.M.-C.; Wong, H.-L.; Yam, V.W.-W. Helical Self-Assembly and Photopolymerization Properties of Achiral Amphiphilic Platinum(II) Diacetylene Complexes of Tridentate 2,6-Bis(1-alkylpyrazol-3-yl)pyridines. ACS Appl. Mater. Interfaces 2016, 8, 17445–17453. [Google Scholar] [CrossRef] [PubMed]
- Leunga, S.Y.-L.; Wonga, K.M.-C.; Yam, V.W.-W. Self-assembly of alkynylplatinum(II) terpyridine amphiphiles into nanostructures via steric control and metal–metal interactions. Proc. Nat. Acad. Sci. USA. 2016, 113, 2845–2850. [Google Scholar] [CrossRef] [PubMed]
- Zhanga, K.; Yeunga, M.C.-L.; Leunga, S.Y.-L.; Yam, V.W.-W. Living supramolecular polymerization achieved by collaborative assembly of platinum(II) complexes and block copolymers. Proc. Nat. Acad. Sci. USA. 2017, 114, 11844–11849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, V.C.-H.; Po, C.; Leung, S.Y.-L.; Chan, A.K.-W.; Yang, S.; Zhu, B.; Cui, X.; Yam, V.W.-W. Formation of 1D Infinite Chains Directed by Metal−Metal and/or π−π Stacking Interactions of Water-Soluble Platinum(II) 2,6-Bis(benzimidazol-2′-yl)pyridine Double Complex Salts. J. Am. Chem. Soc. 2018, 140, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Kuroiwa, K.; Masaki, Y.; Koga, Y.; Deming, T.J. Self-assembly of Discrete Metal Complexes in Aqueous Solution via Block Copolypeptide Amphiphiles. Int. J. Mol. Sci. 2013, 14, 2022–2035. [Google Scholar] [CrossRef]
- Toohara, S.; Tanaka, Y.; Sakurai, S.; Ikeda, T.; Tanaka, K.; Gon, M.; Chujo, Y.; Kuroiwa, K. Self-assembly of [Au(CN)2]- complexes with tomato (Solanum lycopersicum) steroidal alkaloid glycosides to form sheet or tubular structures. Chem. Lett. 2018, 47, 1010–1013. [Google Scholar] [CrossRef]
- Kurth, D.G.; Lehmann, P.; Schütte, M. A route to hierarchical materials based on complexesof metallosupramolecular polyelectrolytesand amphiphiles. Proc. Natl. Acad. Sci. USA 2000, 97, 5704–5707. [Google Scholar] [CrossRef]
- Kurth, D.G.; Severin, N.; Rabe, J.P. Perfectly Straight Nanostructures of Metallosupramolecular Coordination-Polyelectrolyte Amphiphile Complexes on Graphite. Angew. Chem. Int. Ed. 2002, 41, 3681–3683. [Google Scholar] [CrossRef]
- Bodenthin, Y.; Pietsch, U.; Mçhwald, H.; Kurth, D.G. Inducing Spin Crossover in Metallo-supramolecular Polyelectrolytes through an Amphiphilic Phase Transition. J. Am. Chem. Soc. 2005, 127, 3110–3114. [Google Scholar] [CrossRef]
- Bodenthin, Y.; Schwarz, G.; Tomkowicz, Z.; Geue, T.; Haase, W.; Pietsch, U.; Kurth, D.G. Liquid Crystalline Phase Transition Induces Spin Crossover in a Polyelectrolyte Amphiphile Complex. J. Am. Chem. Soc. 2009, 131, 2934–2941. [Google Scholar] [CrossRef]
- Schwarz, G.; Bodenthin, Y.; Tomkowicz, Z.; Haase, W.; Geue, T.; Kohlbrecher, J.; Pietsch, U.; Kurth, D.G. Tuning the Structure and the Magnetic Properties of Metallo-supramolecular Polyelectrolyte−Amphiphile Complexes. J. Am. Chem. Soc. 2011, 133, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Kimizuka, N. Towards Self-Assembling Inorganic Molecular Wires. Adv. Mater. 2000, 12, 1461–1463. [Google Scholar] [CrossRef]
- Kimizuka, N. Soluble Amphiphilic Nanostructures and Potential Applications. In Supramolecular Polymers, 2nd ed.; Ciferri, A., Ed.; Taylor &Francis: Boca Raton, UK, 2005; Chapter 13; pp. 481–507. [Google Scholar]
- Kimizuka, N. Self-Assembly of Supramolecular Nanofibers. Adv. Polym. Sci. 2008, 219, 1–26. [Google Scholar]
- Kimizuka, N.; Oda, N.; Kunitake, T. Supramolecular Assemblies Comprised of One-Dimensional Mixed Valence Platinum Complex and Anionic Amphiphiles in Organic Media. Chem. Lett. 1998, 695–696. [Google Scholar] [CrossRef]
- Kimizuka, N.; Oda, N.; Kunitake, T. Self-Assembling Molecular Wires of Halogen-Bridged Platinum Complexes in Organic Media. Mesoscopic Supramolecular Assemblies Consisting of a Mixed Valent Pt(II)/Pt(IV) Complex and Anionic Amphiphiles. Inorg. Chem. 2000, 39, 2684–2689. [Google Scholar] [CrossRef] [PubMed]
- Kimizuka, N.; Lee, S.-H.; Kunitake, T. Molecular Dispersion of Hetero-Metallic Mixed Valence Chains of [M(en)2][MCl2(en)2] (M: Pt, Pd, Ni) and Anionic Amphiphiles in Organic Media. Angew. Chem. Int. Ed. 2000, 39, 389–391. [Google Scholar] [CrossRef]
- Kimizuka, N.; Yamada, K.; Kunitake, T. Organization of One-dimensional Mixed-Valence Platinum Complexes at the Air-Water Interface and in Langmuir-Blodgett Films. Mol. Cryst. Liq. Cryst. 2000, 342, 103–110. [Google Scholar] [CrossRef]
- Lee, C.-S.; Kimizuka, N. Solvatochromic Nanowires Self-assembled from Cationic, Chloro-bridged Linear Platinum Complexes and Anionic Amphiphiles. Chem. Lett. 2002, 1252–1253. [Google Scholar] [CrossRef]
- Lee, C.-S.; Hatanaka, Y.; Kimizuka, N. Effect of Lipid-packaging on the Charge Transfer Characteristics of One-dimensional, Mixed-valence Platinum Complexes. Int. J. Nanosci. 2002, 1, 391–395. [Google Scholar] [CrossRef]
- Lee, C.-S.; Kimizuka, N. Pillared honeycomb nano-architectures formed on solid surfaces by the self-assembly of lipid-packaged one-dimensional Pt complexes. Proc. Natl. Acad. Sci. USA 2002, 99, 4922–4926. [Google Scholar]
- Kuroiwa, K.; Oda, N.; Kimizuka, N. Supramolecular solvatochromism. Effect of solvents on the self-assembly and charge transfer absorption characteristics of lipid-packaged, linear mixed valence platinum complexes. Sci. Tech. Adv. Mater. 2006, 7, 629–634. [Google Scholar] [CrossRef]
- Matsukizono, H.; Kuroiwa, K.; Kimizuka, N. Lipid-Packaged Linear Iron(II) Trizaole Complexes in Solution: Controlled Spin Conversion via Solvophobic Self-Assembly. J. Am. Chem. Soc. 2008, 130, 5622–5623. [Google Scholar] [CrossRef] [PubMed]
- Ihara, H.; Hachisako, H.; Hirayama, C.; Yamada, K. Specific bindings of methyl orange to chiral bilayer membranes with β-alanyl-L-glutamoyl head groups [1]. Liq. Cryst. 1987, 2, 215–221. [Google Scholar] [CrossRef]
- Hachisako, H.; Ryu, N.; Murakami, R. Molecular structural requirements, dye specificity, and application of anionic peptide amphiphiles that induce intense fluorescence in cationic dyes. Org. Biomol. Chem. 2009, 7, 2327–2337. [Google Scholar] [CrossRef] [PubMed]
- Hachisako, H.; Murata, Y.; Ihara, H. Supramolecular receptors from -amino acid-derived lipids. J. Chem. Soc., Perkin Tras. 2 1999, 12, 2569–2577. [Google Scholar] [CrossRef]
- Kumar, C.V.; Barton, J.K.; Turro, N.J. Photophysics of ruthenium complexes bound to double helical DNA. J. Am. Chem. Soc. 1985, 107, 5518–5523. [Google Scholar] [CrossRef]
- Pyle, A.M.; Rehmann, J.P.; Meshoyrer, R.; Kumar, C.V.; Turro, N.J.; Barton, J.K. Mixed-ligand complexes of ruthenium(II): Factors governing binding to DNA. J. Am. Chem. Soc. 1989, 111, 3051–3058. [Google Scholar] [CrossRef]
- Montalti, M.; Cedi, A.; Prodi, L.; Gandolfi, M.T. Handbook of Photochemistry, 3rd ed.; CRC Press, Taylor & Francis: London, UK, 2006; pp. 379–404. [Google Scholar]
- Yeh, A.T.; Shank, C.V.; McCusker, J.K. Ultrafast Electron Localization Dynamics Following Photo-Induced Charge Transfer. Science 2000, 289, 935–938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, K.; Kobayashi, A.; Kaneko, S.; Takehisa, K.; Yoshihara, T.; Ishida, H.; Shiina, Y.; Oishi, S.; Tobita, S. Reevaluation of absolute luminescence quantum yields of standard solutions using a spectrometer with an integrating sphere and a back-thinned CCD detector. Phys. Chem. Chem. Phys. 2009, 11, 9850–9860. [Google Scholar] [CrossRef] [PubMed]
- Rillema, D.P.; Jones, D.S.; Levy, H.A. Structure of Tris(2,2′-bipyridyl)ruthenium(II) Hexafiuorophosphate, [Ru(bipy)3][PF6]2; X-Ray Crystallographic Determination. J. Chem. Soc. Chem. Commun. 1979, 19, 849–851. [Google Scholar] [CrossRef]
- Broomhead, J.A.; Young, C.G. Tris(2,2″-Bipyridine)Ruthenium(II) Dichloride Hexahydrate. Inorganic Syntheses 1990, 28, 338–340. [Google Scholar]
- Tien, N.-D.; Hoa, T.-P.; Mochizuki, M.; Saijo, K.; Hasegawa, H.; Sasaki, S.; Sakurai, S. Higher-order crystalline structures of poly(oxyethylene) in poly(D,L-lactide)/poly(oxyethylene) blends. Polymer 2013, 54, 4653–4659. [Google Scholar] [CrossRef]
- Tian, N.-D.; Sasaki, S.; Masunaga, H.; Shimizu, N.; Igarashi, N.; Sakurai, S. Small-angle X-ray scattering studies on melting and recrystallization behaviors of poly(oxyethylene) crystallites in poly(D,L-lactide)/poly(oxyethylene) blends. Polymer 2014, 55, 2562–2569. [Google Scholar]






| Composite | λex/nm | λem/nm | Emission Quantum Yield/% | Lifetime/µs |
|---|---|---|---|---|
| [RuII(bpy)3](1)2 | 450 | 600 | 8.4 | 0.68 |
| [RuII(bpy)3](2)2 | 416 | 599 | 9.0 | 0.82 |
| [RuII(bpy)3](3)2 | 420 | 599 | 7.7 | 0.74 |
| [RuII(bpy)3](4)2 | 446 | 601 | 9.0 | 0.79 |
| [RuII(bpy)3](5)2 | 448 | 602 | 8.9 | 0.78 |
| [RuII(bpy)3](6)2 | 435 | 601 | 8.8 | 0.72 |
| [RuII(bpy)3]Cl2 1 | 450 [71] | 608 [71] | 9.5 [71] | 0.89 [73] |
| Composite | Hypochromic Effect | Luminescence | Induced Circular Dichroism (ICD) Signal | Nanostructure |
|---|---|---|---|---|
| [RuII(bpy)3](1)2 | Strong | Phosphorescence | Moderate | Helical ribbon |
| [RuII(bpy)3](2)2 | Strong | Phosphorescence | Moderate | Ribbon |
| [RuII(bpy)3](3)2 | Moderate | Phosphorescence | Weak | Ribbon |
| [RuII(bpy)3](4)2 | Strong | Phosphorescence | Weak | Sheet |
| [RuII(bpy)3](5)2 | Moderate | Phosphorescence | Weak | Sheet |
| [RuII(bpy)3](6)2 | Moderate | Phosphorescence | Weak | Sheet |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hatakeda, M.; Toohara, S.; Nakashima, T.; Sakurai, S.; Kuroiwa, K. Helical-Ribbon and Tape Formation of Lipid Packaged [Ru(bpy)3]2+ Complexes in Organic Media. Int. J. Mol. Sci. 2019, 20, 3298. https://doi.org/10.3390/ijms20133298
Hatakeda M, Toohara S, Nakashima T, Sakurai S, Kuroiwa K. Helical-Ribbon and Tape Formation of Lipid Packaged [Ru(bpy)3]2+ Complexes in Organic Media. International Journal of Molecular Sciences. 2019; 20(13):3298. https://doi.org/10.3390/ijms20133298
Chicago/Turabian StyleHatakeda, Miho, Souta Toohara, Takuya Nakashima, Shinichi Sakurai, and Keita Kuroiwa. 2019. "Helical-Ribbon and Tape Formation of Lipid Packaged [Ru(bpy)3]2+ Complexes in Organic Media" International Journal of Molecular Sciences 20, no. 13: 3298. https://doi.org/10.3390/ijms20133298