Recent Advances in Lipid-Based Nanosystems for Gemcitabine and Gemcitabine–Combination Therapy
Abstract
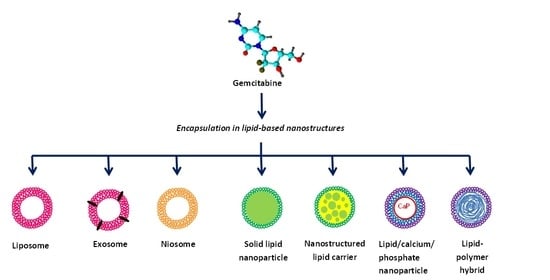
:1. Introduction
2. Liposomes
3. Niosomes
4. Solid Lipid Nanoparticles
5. Lipid/Calcium/Phosphate Nanoparticles
6. Nanostructured Lipid Carriers
7. Exosomes
8. Lipid-Polymer Hybrid Nanoparticles
9. Miscellaneous Lipid Nanoparticles
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moysan, E.; Bastiat, G.; Benoit, J.-P. Gemcitabine versus Modified Gemcitabine: A Review of Several Promising Chemical Modifications. Mol. Pharm. 2013, 10, 430–444. [Google Scholar] [CrossRef] [Green Version]
- Bianchi, V.; Borella, S.; Calderazzo, F.; Ferraro, P.; Bianchi, L.C.; Reichard, P. Inhibition of ribonucleotide reductase by 2’-substituted deoxycytidine analogs: Possible application in AIDS treatment. Proc. Natl. Acad. Sci. USA 1994, 91, 8403–8407. [Google Scholar] [CrossRef] [Green Version]
- Hoang, T.; Kim, K.; Jaslowski, A.; Koch, P.; Beatty, P.; McGovern, J.; Quisumbing, M.; Shapiro, G.; Witte, R.; Schiller, J.H. Phase II study of second-line gemcitabine in sensitive or refractory small cell lung cancer. Lung Cancer 2003, 42, 97–102. [Google Scholar] [CrossRef]
- Carmichael, J.; Fink, U.; Russell, R.C.; Spittle, M.F.; Harris, A.L.; Spiessi, G.; Blatter, J. Phase II study of gemcitabine in patients with advanced pancreatic cancer. Br. J. Cancer 1996, 73, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Yardley, D.A. Gemcitabine Plus Paclitaxel in Breast Cancer. In Seminars in Oncology; Elsevier: Amsterdam, The Netherlands, 2005; Volume 32, pp. 14–21. [Google Scholar] [CrossRef]
- Ozols, R. Gemcitabine and Carboplatin in Second-Line Ovarian Cancer. In Seminars in Oncology; Elsevier: Amsterdam, The Netherlands, 2005; Volume 32, pp. 4–8. [Google Scholar] [CrossRef]
- Parra, H.S.; Cavina, R.; Latteri, F.; Sala, A.; Dambrosio, M.; Antonelli, G.; Morenghi, E.; Alloisio, M.; Ravasi, G.; Santoro, A. Three-week versus four-week schedule of cisplatin and gemcitabine: Results of a randomized phase II study. Ann. Oncol. 2002, 13, 1080–1086. [Google Scholar] [CrossRef]
- Mackey, J.R.; Mani, R.S.; Selner, M.; Mowles, D.; Young, J.D.; Belt, J.A.; Crawford, C.R.; Cass, C.E. Functional nucleoside transporters are required for gemcitabine influx and manifestation of toxicity in cancer cell lines. Cancer Res. 1998, 58, 4349–4357. [Google Scholar]
- Bouffard, D.Y.; Laliberté, J.; Momparler, R.L. Kinetic studies on 2′,2′-difluorodeoxycytidine (gemcitabine) with purified human deoxycytidine kinase and cytidine deaminase. Biochem. Pharmacol. 1993, 45, 1857–1861. [Google Scholar] [CrossRef]
- Van Rompay, A.R.; Johansson, M.; Karlsson, A. Phosphorylation of Deoxycytidine Analog Monophosphates by UMP-CMP Kinase: Molecular Characterization of the Human Enzyme. Mol. Pharmacol. 1999, 56, 562–569. [Google Scholar] [CrossRef] [Green Version]
- Wong, A.; Soo, R.A.; Yong, W.-P.; Innocenti, F. Clinical pharmacology and pharmacogenetics of gemcitabine. Drug Metab. Rev. 2009, 41, 77–88. [Google Scholar] [CrossRef]
- Huang, P.; Chubb, S.; Hertel, L.W.; Grindey, G.B.; Plunkett, W. Action of 2′,2′-difluorodeoxycytidine on DNA synthesis. Cancer Res. 1991, 51, 6110–6117. [Google Scholar]
- Heinemann, V.; Xu, Y.Z.; Chubb, S.; Sen, A.; Hertel, L.W.; Grindey, G.B.; Plunkett, W. Inhibition of ribonucleotide reduction in CCRF-CEM cells by 2’,2’-difluorodeoxycytidine. Mol. Pharmacol. 1990, 38, 567–572. [Google Scholar]
- Heinemann, V.; Xu, Y.Z.; Chubb, S.; Sen, A.; Hertel, L.W.; Grindey, G.B.; Plunkett, W. Cellular elimination of 2’,2’-difluorodeoxycytidine 5’-triphosphate: A mechanism of self-potentiation. Cancer Res. 1992, 52, 533–539. [Google Scholar] [PubMed]
- Ferreira, C.G.; Span, S.W.; Peters, G.J.; Kruyt, F.A.; Giaccone, G. Chemotherapy triggers apoptosis in a caspase-8-dependent and mitochondria-controlled manner in the non-small cell lung cancer cell line NCI-H460. Cancer Res. 2000, 60, 7133–7141. [Google Scholar] [PubMed]
- Chandler, N.M.; Canete, J.J.; Callery, M.P. Caspase-3 drives apoptosis in pancreatic cancer cells after treatment with gemcitabine. J. Gastrointest. Surg. 2004, 8, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Xi, L.; Li, Q.; Wang, S.; Cai, Z.; Zhang, X.; Yu, C. Combination of PI3K/Akt Pathway Inhibition and Plk1 Depletion Can Enhance Chemosensitivity to Gemcitabine in Pancreatic Carcinoma. Transl. Oncol. 2018, 11, 852–863. [Google Scholar] [CrossRef]
- Cavalcante, L.D.S.; Monteiro, G. Gemcitabine: Metabolism and molecular mechanisms of action, sensitivity and chemoresistance in pancreatic cancer. Eur. J. Pharmacol. 2014, 741, 8–16. [Google Scholar] [CrossRef]
- Birhanu, G.; Javar, H.A.; Seyedjafari, E.; Zandi-Karimi, A. Nanotechnology for delivery of gemcitabine to treat pancreatic cancer. Biomed. Pharmacother. 2017, 88, 635–643. [Google Scholar] [CrossRef]
- Maiyo, F.; Singh, M. Selenium nanoparticles: Potential in cancer gene and drug delivery. Nanomedicine 2017, 12, 1075–1089. [Google Scholar] [CrossRef] [PubMed]
- Samanta, K.; Setua, S.; Kumari, S.; Jaggi, M.; Yallapu, M.M.; Chauhan, S.C. Gemcitabine Combination Nano Therapies for Pancreatic Cancer. Pharmaceutics 2019, 11, 574. [Google Scholar] [CrossRef] [Green Version]
- Akinyelu, J.; Singh, M. Folate-tagged chitosan-functionalized gold nanoparticles for enhanced delivery of 5-fluorouracil to cancer cells. Appl. Nanosci. 2018, 9, 7–17. [Google Scholar] [CrossRef]
- Maney, V.; Singh, M. An in vitro assessment of Chitosan/ Bimetallic PtAu nanocomposites as delivery vehicles for Doxorubicin. Nanomedicine 2017, 12, 2625–2640. [Google Scholar] [CrossRef]
- Moodley, T.; Singh, M. Polymeric Mesoporous Silica Nanoparticles for Combination Drug Delivery In vitro. Biointerface Res. Appl. Chem. 2020, 11, 11905–11919. [Google Scholar] [CrossRef]
- Zylberberg, C.; Matosevic, S. Pharmaceutical liposomal drug delivery: A review of new delivery systems and a look at the regulatory landscape. Drug Deliv. 2016, 23, 3319–3329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamam, H.; Park, J.; Gadalla, H.H.; Masters, A.R.; Abdel-Aleem, J.A.; Abdelrahman, S.I.; Abdelrahman, A.A.; Lyle, L.T.; Yeo, Y. Development of Liposomal Gemcitabine with High Drug Loading Capacity. Mol. Pharm. 2019, 16, 2858–2871. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Im, B.N.; Hwang, H.S.; Na, K. Gemcitabine-loaded DSPE-PEG-PheoA liposome as a photomediated immune modulator for cholangiocarcinoma treatment. Biomaterials 2018, 183, 139–150. [Google Scholar] [CrossRef]
- Ding, N.; Wang, Y.; Wang, X.; Chu, W.; Yin, T.; Gou, J.; He, H.; Zhang, Y.; Wang, Y.; Tang, X. Improving plasma stability and antitumor effect of gemcitabine via PEGylated liposome prepared by active drug loading. J. Drug Deliv. Sci. Technol. 2020, 57, 101538. [Google Scholar] [CrossRef]
- Yalcin, T.E.; Ilbasmis-Tamer, S.; Ibisoglu, B.; Özdemir, A.; Ark, M.; Takka, S. Gemcitabine hydrochloride-loaded liposomes and nanoparticles: Comparison of encapsulation efficiency, drug release, particle size, and cytotoxicity. Pharm. Dev. Technol. 2018, 23, 76–86. [Google Scholar] [CrossRef]
- Kanamala, M.; Palmer, B.D.; Ghandehari, H.; Wilson, W.R.; Wu, Z. PEG-Benzaldehyde-Hydrazone-Lipid Based PEG-Sheddable pH-Sensitive Liposomes: Abilities for Endosomal Escape and Long Circulation. Pharm. Res. 2018, 35, 154. [Google Scholar] [CrossRef]
- Mu, Y.; Wang, D.; Bie, L.; Luo, S.; Mu, X.; Zhao, Y. Glypican-1-targeted and gemcitabine-loaded liposomes enhance tumor-suppressing effect on pancreatic cancer. Aging (Albany NY) 2020, 12, 19585–19596. [Google Scholar] [CrossRef] [PubMed]
- Li, P.-W.; Luo, S.; Xiao, L.-Y.; Tian, B.-L.; Wang, L.; Zhang, Z.-R.; Zeng, Y.-C. A novel gemcitabine derivative-loaded liposome with great pancreas-targeting ability. Acta Pharmacol. Sin. 2019, 40, 1448–1456. [Google Scholar] [CrossRef]
- Tucci, S.T.; Kheirolomoom, A.; Ingham, E.S.; Mahakian, L.M.; Tam, S.M.; Foiret, J.; Hubbard, N.E.; Borowsky, A.D.; Baikoghli, M.; Cheng, R.H.; et al. Tumor-specific delivery of gemcitabine with activatable liposomes. J. Control. Release 2019, 309, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Fuse, T.; Tagami, T.; Tane, M.; Ozeki, T. Effective light-triggered contents release from helper lipid-incorporated liposomes co-encapsulating gemcitabine and a water-soluble photosensitizer. Int. J. Pharm. 2018, 540, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Paxton, J.W.; Wu, Z. Development of Long-Circulating pH-Sensitive Liposomes to Circumvent Gemcitabine Resistance in Pancreatic Cancer Cells. Pharm. Res. 2016, 33, 1628–1637. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Paxton, J.W.; Wu, Z. Enhanced pH-Responsiveness, Cellular Trafficking, Cytotoxicity and Long-circulation of PEGylated Liposomes with Post-insertion Technique Using Gemcitabine as a Model Drug. Pharm. Res. 2015, 32, 2428–2438. [Google Scholar] [CrossRef] [PubMed]
- Kanamala, M.; Palmer, B.D.; Jamieson, S.M.; Wilson, W.R.; Wu, Z. Dual pH-sensitive liposomes with low pH-triggered sheddable PEG for enhanced tumor-targeted drug delivery. Nanomedicine 2019, 14, 1971–1989. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, M.; Pandya, T.; Gandhi, R.; Patel, S.; Mashru, R.; Misra, A.; Tandel, H. Inhalable liposomal dry powder of gemcitabine-HCl: Formulation, in vitro characterization and in vivo studies. Int. J. Pharm. 2015, 496, 886–895. [Google Scholar] [CrossRef]
- Yang, W.; Hu, Q.; Xu, Y.; Liu, H.; Zhong, L. Antibody fragment-conjugated gemcitabine and paclitaxel-based liposome for effective therapeutic efficacy in pancreatic cancer. Mater. Sci. Eng. C 2018, 89, 328–335. [Google Scholar] [CrossRef]
- Urey, C.; Hilmersson, K.S.; Andersson, B.; Ansari, D.; Andersson, R. Development and In Vitro Characterization of a Gemcitabine-loaded MUC4-targeted Immunoliposome against Pancreatic Ductal Adenocarcinoma. Anticancer Res. 2017, 37, 6031–6039. [Google Scholar] [CrossRef] [Green Version]
- Shin, D.H.; Lee, S.-J.; Kim, J.S.; Ryu, J.-H.; Kim, J.-S. Synergistic Effect of Immunoliposomal Gemcitabine and Bevacizumab in Glioblastoma Stem Cell-Targeted Therapy. J. Biomed. Nanotechnol. 2015, 11, 1989–2002. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Zhao, Z.; Kim, J.; Razmi, A.; Wang, L.L.; Kapate, N.; Gao, Y.; Peng, K.; Ukidve, A.; Mitragotri, S. Gemcitabine and doxorubicin in immunostimulatory monophosphoryl lipid A liposomes for treating breast cancer. Bioeng. Transl. Med. 2021, 6, 10188. [Google Scholar] [CrossRef]
- Unnam, S.; Panduragaiah, V.M.; Sidramappa, M.A.; Eswara, B.R.M.; Sambamoorthy, U.; Venkataraju, M.P.; Manjappa, A.S.; Rao, M.E.B. Gemcitabine-loaded Folic Acid Tagged Liposomes: Improved Pharmacokinetic and Biodistribution Profile. Curr. Drug Deliv. 2019, 16, 111–122. [Google Scholar] [CrossRef]
- Tang, M.; Svirskis, D.; Leung, E.; Kanamala, M.; Wang, H.; Wu, Z. Can intracellular drug delivery using hyaluronic acid functionalized pH-sensitive liposomes overcome gemcitabine resistance in pancreatic cancer? J. Control. Release 2019, 305, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Han, N.-K.; Shin, D.H.; Kim, J.S.; Weon, K.Y.; Jang, C.-Y.; Kim, J.-S. Hyaluronan-conjugated liposomes encapsulating gemcitabine for breast cancer stem cells. Int. J. Nanomed. 2016, 11, 1413–1425. [Google Scholar] [CrossRef] [Green Version]
- Tang, Z.; Feng, W.; Yang, Y.; Wang, Q. Gemcitabine-loaded RGD modified liposome for ovarian cancer: Preparation, characterization and pharmacodynamic studies. Drug Des. Dev. Ther. 2019, 13, 3281–3290. [Google Scholar] [CrossRef] [Green Version]
- Cai, W.; Geng, C.; Jiang, L.; Sun, J.; Chen, B.; Zhou, Y.; Yang, B.; Lu, H. Encapsulation of gemcitabine in RGD-modified nanoliposomes improves breast cancer inhibitory activity. Pharm. Dev. Technol. 2020, 25, 640–648. [Google Scholar] [CrossRef]
- Affram, K.; Udofot, O.; Agyare, E. Cytotoxicity of gemcitabine-loaded thermosensitive liposomes in pancreatic cancer cell lines. Integr. Cancer Sci. Ther. 2015, 2, 133–142. [Google Scholar] [PubMed]
- Affram, K.; Udofot, O.; Singh, M.; Krishnan, S.; Reams, R.; Rosenberg, J.; Agyare, E. Smart thermosensitive liposomes for effective solid tumor therapy and in vivo imaging. PLoS ONE 2017, 12, e0185116. [Google Scholar] [CrossRef] [PubMed]
- Kirui, D.K.; Celia, C.; Molinaro, R.; Bansal, S.S.; Cosco, D.; Fresta, M.; Shen, H.; Ferrari, M. Mild hyperthermia enhances transport of liposomal gemcitabine and improves in vivo therapeutic response. Adv. Healthc. Mater. 2015, 4, 1092–1103. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, R.V.; Martins, T.M.D.M.; Goes, A.M.; Fabris, J.D.; Cavalcante, L.C.; Outon, L.E.F.; Domingues, R.Z. Thermosensitive gemcitabine-magnetoliposomes for combined hyperthermia and chemotherapy. Nanotechnology 2016, 27, 085105. [Google Scholar] [CrossRef] [Green Version]
- Hylander, B.L.; Sen, A.; Beachy, S.H.; Pitoniak, R.; Ullas, S.; Gibbs, J.F.; Qiu, J.; Prey, J.D.; Fetterly, G.J.; Repasky, E.A. Tumor priming by Apo2L/TRAIL reduces interstitial fluid pressure and enhances efficacy of liposomal gemcitabine in a patient derived xenograft tumor model. J. Control. Release 2015, 217, 160–169. [Google Scholar] [CrossRef] [Green Version]
- Emamzadeh, M.; Emamzadeh, M.; Pasparakis, G. Dual Controlled Delivery of Gemcitabine and Cisplatin Using Polymer-Modified Thermosensitive Liposomes for Pancreatic Cancer. ACS Appl. Bio Mater. 2019, 2, 1298–1309. [Google Scholar] [CrossRef]
- Ribeiro, R.F.L.; Ferreira, R.V.; Pedersoli, D.C.; Paiva, P.R.P.; Cunha, P.D.S.; Goes, A.M.; Domingues, R.Z. Cytotoxic effect of thermosensitive magnetoliposomes loaded with gemcitabine and paclitaxel on human primary breast cancer cells (MGSO-3 line). J. Nanopart. Res. 2020, 22, 1–16. [Google Scholar] [CrossRef]
- Fan, Y.; Wang, Q.; Lin, G.; Shi, Y.; Gu, Z.; Ding, T. Combination of using prodrug-modified cationic liposome nanocomplexes and a potentiating strategy via targeted co-delivery of gemcitabine and docetaxel for CD44-overexpressed triple negative breast cancer therapy. Acta Biomater. 2017, 62, 257–272. [Google Scholar] [CrossRef] [PubMed]
- Caliskan, Y.; Dalgic, A.D.; Gerekci, S.; Gulec, E.A.; Tezcaner, A.; Ozen, C.; Keskin, D. A new therapeutic combination for osteosarcoma: Gemcitabine and Clofazimine co-loaded liposomal formulation. Int. J. Pharm. 2019, 557, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.H.; Koo, M.-J.; Kim, J.S.; Kim, J.-S. Herceptin-conjugated temperature-sensitive immunoliposomes encapsulating gemcitabine for breast cancer. Arch. Pharmacal Res. 2016, 39, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Tong, J.; Liu, J.; Lin, W.; Zhang, C.; Chen, K.; Zhao, J.; Zhu, W. Combination of gemcitabine-containing magnetoliposome and oxaliplatin-containing magnetoliposome in breast cancer treatment: A possible mechanism with potential for clinical application. Oncotarget 2016, 7, 43762–43778. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Tamam, H.; Yeo, Y. Mixed Liposome Approach for Ratiometric and Sequential Delivery of Paclitaxel and Gemcitabine. AAPS PharmSciTech 2018, 19, 693–699. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Zhang, Z. Nanoformulation of Apolipoprotein E3-Tagged Liposomal Nanoparticles for the co-Delivery of KRAS-siRNA and Gemcitabine for Pancreatic Cancer Treatment. Pharm. Res. 2020, 37, 1–11. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, F.; Jiang, X.; Zhao, X.; Wang, Y.; Kuai, Q.; Nie, G.; He, M.; Pan, Y.; Shi, W.; et al. Co-Delivery of Gemcitabine and Mcl-1 SiRNA via Cationic Liposome-Based System Enhances the Efficacy of Chemotherapy in Pancreatic Cancer. J. Biomed. Nanotechnol. 2019, 15, 966–978. [Google Scholar] [CrossRef]
- Lalani, R.A.; Bhatt, P.; Rathi, M.; Misra, A. Improved sensitivity and in vitro efficacy of RGD grafted PEGylated gemcitabine liposomes in RRM1 siRNA pretreated cancer cells. In Proceedings of the AACR 107th Annual Meeting 2016, New Orleans, LA, USA, 16–20 April 2016; 16 April 2016; Volume 76, p. 2063. [Google Scholar] [CrossRef]
- Zhao, L.-P.; Xue, C.; Zhang, J.-W.; Hu, Z.-H.; Zhao, Y.-Y.; Zhang, J.; Huang, Y.; Zhao, H.-Y.; Zhang, L. Expression of RRM1 and its association with resistancy to gemcitabine-based chemotherapy in advanced nasopharyngeal carcinoma. Chin. J. Cancer 2012, 31, 476–483. [Google Scholar] [CrossRef] [Green Version]
- Bulanadi, J.C.; Xue, A.; Gong, X.; Bean, P.A.; Julovi, S.M.; De Campo, L.; Smith, R.C.; Moghaddam, M.J. Biomimetic Gemcitabine–Lipid Prodrug Nanoparticles for Pancreatic Cancer. ChemPlusChem 2020, 85, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Seleci, D.A.; Seleci, M.; Walter, J.-G.; Stahl, F.; Scheper, T. Niosomes as Nanoparticular Drug Carriers: Fundamentals and Recent Applications. J. Nanomater. 2016, 2016, 1–13. [Google Scholar] [CrossRef]
- Maniam, G. Preparation, Characterization and Anti-Pancreatic Cancer Effects of Gemcitabine-Tocotrienols Entrapped Niosomes. Ph.D. Thesis, International Medical University, Kuala Lumpur, Malaysia, 2019. [Google Scholar]
- Saimi, N.M.; Salim, N.; Ahmad, N.; Abdulmalek, E.; Rahman, M.A. Aerosolized Niosome Formulation Containing Gemcitabine and Cisplatin for Lung Cancer Treatment: Optimization, Characterization and In Vitro Evaluation. Pharmaceutics 2021, 13, 59. [Google Scholar] [CrossRef]
- Mohammadi-Samani, S.; Ghasemiyeh, P. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: Applications, advantages and disadvantages. Res. Pharm. Sci. 2018, 13, 288–303. [Google Scholar] [CrossRef]
- Nandini, P.T.; Doijad, R.C.; Shivakumar, H.N.; Dandagi, P.M. Formulation and evaluation of gemcitabine-loaded solid lipid nanoparticles. Drug Deliv. 2015, 22, 647–651. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Affram, K.O.; Smith, T.; Ofori, E.; Krishnan, S.; Underwood, P.; Trevino, J.G.; Agyare, E. Cytotoxic effects of gemcitabine-loaded solid lipid nanoparticles in pancreatic cancer cells. J. Drug Deliv. Sci. Technol. 2020, 55, 101374. [Google Scholar] [CrossRef]
- Soni, N.; Soni, N.; Ramteke, P.W.; Pandey, H. A validated RP-HPLC assay method for determination of gemcitabine loaded nanosized solid lipid nanoparticles. J. Drug Deliv. Ther. 2018, 8, 308–313. [Google Scholar] [CrossRef]
- Wang, C.; Zheng, Y.; Oval, M.A.S.; Valdes, S.A.; Chen, Z.; Lansakara-P, D.S.; Du, M.; Shi, Y.; Cui, Z. Oral 4-(N)-stearoyl gemcitabine nanoparticles inhibit tumor growth in mouse models. Oncotarget 2017, 8, 89876–89886. [Google Scholar] [CrossRef]
- Sloat, B.R.; Sandoval, M.A.; Li, D.; Chung, W.-G.; Lansakara-P, D.S.; Proteau, P.J.; Kiguchi, K.; DiGiovanni, J.; Cui, Z. In vitro and in vivo anti-tumor activities of a gemcitabine derivative carried by nanoparticles. Int. J. Pharm. 2011, 409, 278–288. [Google Scholar] [CrossRef] [Green Version]
- Lansakara, -P.D.S.; Rodriguez, B.L.; Cui, Z. Synthesis and in vitro evaluation of novel lipophilic monophosphorylated gemcitabine derivatives and their nanoparticles. Int. J. Pharm. 2012, 429, 123–134. [Google Scholar] [CrossRef] [Green Version]
- Chen, Z.; Zheng, Y.; Shi, Y.; Cui, Z. Overcoming tumor cell chemoresistance using nanoparticles: Lysosomes are beneficial for (stearoyl) gemcitabine-incorporated solid lipid nanoparticles. Int. J. Nanomed. 2018, 13, 319–336. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Bush, X.; Yan, B.; Chen, J.A. Gemcitabine nanoparticles promote antitumor immunity against melanoma. Biomaterials 2019, 189, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, P.; Zou, Q.; Li, X.; Fu, J.; Luo, Y.; Liang, X.; Jin, Y. Co-Delivery of Gemcitabine and Paclitaxel in cRGD-Modified Long Circulating Nanoparticles with Asymmetric Lipid Layers for Breast Cancer Treatment. Molecules 2018, 23, 2906. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haider, M.; Abdin, S.M.; Kamal, L.; Orive, G. Nanostructured Lipid Carriers for Delivery of Chemotherapeutics: A Review. Pharmaceutics 2020, 12, 288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, Y.; Tian, B.; Zhang, J.; Li, K.; Wang, L.; Han, J.; Wu, Z. Tumor-targeted polymeric nanostructured lipid carriers with precise ratiometric control over dual-drug loading for combination therapy in non-small-cell lung cancer. Int. J. Nanomed. 2017, 12, 1699–1715. [Google Scholar] [CrossRef] [Green Version]
- Liang, Y.; Zhang, J.; Tian, B.; Wu, Z.; Svirskis, D.; Han, J. A NAG-Guided Nano-Delivery System for Redox- and pH-Triggered Intracellularly Sequential Drug Release in Cancer Cells. Int. J. Nanomed. 2020, 15, 841–855. [Google Scholar] [CrossRef] [Green Version]
- Lu, Z.; Su, J.; Li, Z.; Zhan, Y.; Ye, D. Hyaluronic acid-coated, prodrug-based nanostructured lipid carriers for enhanced pancreatic cancer therapy. Drug Dev. Ind. Pharm. 2017, 43, 160–170. [Google Scholar] [CrossRef]
- Ni, S.; Qiu, L.; Zhang, G.; Zhou, H.; Han, Y. Lymph cancer chemotherapy: Delivery of doxorubicin–gemcitabine prodrug and vincristine by nanostructured lipid carriers. Int. J. Nanomed. 2017, 12, 1565–1576. [Google Scholar] [CrossRef] [Green Version]
- Campanella, C.; Bavisotto, C.C.; Logozzi, M.; Gammazza, A.M.; Mizzoni, D.; Cappello, F.; Fais, S. On the Choice of the Extracellular Vesicles for Therapeutic Purposes. Int. J. Mol. Sci. 2019, 20, 236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.-J.; Wu, J.-Y.; Wang, J.-M.; Hu, X.-B.; Cai, J.-X.; Xiang, D.-X. Gemcitabine loaded autologous exosomes for effective and safe chemotherapy of pancreatic cancer. Acta Biomater. 2020, 101, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Date, T.; Nimbalkar, V.; Kamat, J.; Mittal, A.; Mahato, R.I.; Chitkara, D. Lipid-polymer hybrid nanocarriers for delivering cancer therapeutics. J. Control. Release 2018, 271, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Yalcin, T.E.; Ilbasmis-Tamer, S.; Takka, S. Development and characterization of gemcitabine hydrochloride loaded lipid polymer hybrid nanoparticles (LPHNs) using central composite design. Int. J. Pharm. 2018, 548, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Yalcin, T.E.; Ilbasmis-Tamer, S.; Takka, S. Antitumor activity of gemcitabine hydrochloride loaded lipid polymer hybrid nanoparticles (LPHNs): In vitro and in vivo. Int. J. Pharm. 2020, 580, 119246. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Li, F.; Li, Y.; Wang, H.; Ren, H.; Chen, J.; Nie, G.; Hao, J. Co-delivery of HIF1α siRNA and gemcitabine via biocompatible lipid-polymer hybrid nanoparticles for effective treatment of pancreatic cancer. Biomaterials 2015, 46, 13–25. [Google Scholar] [CrossRef]
- Dora, C.P.; Kushwah, V.; Katiyar, S.S.; Kumar, P.; Pillay, V.; Suresh, S.; Jain, S. Improved metabolic stability and therapeutic efficacy of a novel molecular gemcitabine phospholipid complex. Int. J. Pharm. 2017, 530, 113–127. [Google Scholar] [CrossRef]
- Bastiancich, C.; Vanvarenberg, K.; Ucakar, B.; Pitorre, M.; Bastiat, G.; Lagarce, F.; Préat, V.; Danhier, F. Lauroyl-gemcitabine-loaded lipid nanocapsule hydrogel for the treatment of glioblastoma. J. Control. Release 2016, 225, 283–293. [Google Scholar] [CrossRef]
- Sasso, M.S.; Lollo, G.; Pitorre, M.; Solito, S.; Pinton, L.; Valpione, S.; Bastiat, G.; Mandruzzato, S.; Bronte, V.; Marigo, I.; et al. Low dose gemcitabine-loaded lipid nanocapsules target monocytic myeloid-derived suppressor cells and potentiate cancer immunotherapy. Biomaterials 2016, 96, 47–62. [Google Scholar] [CrossRef]
- Gaudin, A.; Song, E.; King, A.R.; Saucier-Sawyer, J.K.; Bindra, R.; Desmaële, D.; Couvreur, P.; Saltzman, W.M. PEGylated squalenoyl-gemcitabine nanoparticles for the treatment of glioblastoma. Biomaterials 2016, 105, 136–144. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Nogales, C.; Sebastián, V.; Irusta, S.; Desmaële, D.; Couvreur, P.; Blanco-Prieto, M. A unique multidrug nanomedicine made of squalenoyl-gemcitabine and alkyl-lysophospholipid edelfosine. Eur. J. Pharm. Biopharm. 2019, 144, 165–173. [Google Scholar] [CrossRef]
- Rodríguez-Nogales, C.; Mura, S.; Couvreur, P.; Blanco-Prieto, M. Squalenoyl-gemcitabine/edelfosine nanoassemblies: Anticancer activity in pediatric cancer cells and pharmacokinetic profile in mice. Int. J. Pharm. 2020, 582, 119345. [Google Scholar] [CrossRef]
- Comparetti, E.J.; Lins, P.P.; Quitiba, J.V.B.; Zucolotto, V. Cancer cell membrane-derived nanoparticles improve the activity of gemcitabine and paclitaxel on pancreatic cancer cells and coordinate immunoregulatory properties on professional antigen-presenting cells. Mater. Adv. 2020, 1, 1775–1787. [Google Scholar] [CrossRef]
- Singh, R.; Lillard, J.W. Nanoparticle-based targeted drug delivery. Exp. Mol. Pathol. 2009, 86, 215–223. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| Lipid-Based Nanoparticle | Advantages | Disadvantages |
|---|---|---|
| Liposomes | Biocompatible; biodegradable High loading capacity Flexibility of composition Targetable | Drug leakage High production cost Special storage conditions required |
| Niosomes | Biocompatible; non-immunogenic Osmotically active and stable Low production cost; long shelf-life | Drug leakage No human safety data available |
| Solid lipid nanoparticles | Biocompatible; biodegradable Low toxicity High bioavailability of drugs Targetable Amenable to large-scale production | Low drug loading efficiency Risk of drug expulsion upon storage No human safety data available |
| Lipid/calcium/phosphate nanoparticles | High encapsulation efficiency Efficient endosomal escape Sustained drug release Blood-brain barrier permeability Targetable | Complex structure and synthesis No human safety data available |
| Nanostructured lipid carriers | Biodegradable Increased drug loading Prevents drug expulsion Improved stability Targetable | No human safety data available |
| Exosomes | High biocompatibility High drug encapsulation efficiency Natural carriers Small size-tissue penetration Slightly negative zeta potential-extended circulation | Lack of standardized techniques for isolation and purification No human safety data available |
| Lipid-polymer hybrids | Robust delivery Well-defined release kinetics Good serum stability Targetable | Suboptimal drug loading and entrapment efficiency No human safety data available |
| Lipid nanocapsules | Biocompatible; small size Long-term stability Manufactured by low energy, organic, solvent-free process | No human safety data available |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Habib, S.; Singh, M. Recent Advances in Lipid-Based Nanosystems for Gemcitabine and Gemcitabine–Combination Therapy. Nanomaterials 2021, 11, 597. https://doi.org/10.3390/nano11030597
Habib S, Singh M. Recent Advances in Lipid-Based Nanosystems for Gemcitabine and Gemcitabine–Combination Therapy. Nanomaterials. 2021; 11(3):597. https://doi.org/10.3390/nano11030597
Chicago/Turabian StyleHabib, Saffiya, and Moganavelli Singh. 2021. "Recent Advances in Lipid-Based Nanosystems for Gemcitabine and Gemcitabine–Combination Therapy" Nanomaterials 11, no. 3: 597. https://doi.org/10.3390/nano11030597