Chasing ChEs-MAO B Multi-Targeting 4-Aminomethyl-7-Benzyloxy-2H-Chromen-2-ones
Abstract
:1. Introduction
2. Results
2.1. Chemistry
2.2. In Vitro Screening
3. Discussion
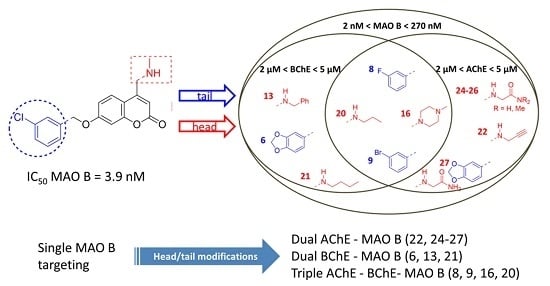
3.1. Structure–Activity Relationships
3.2. AChE Kinetics
3.3. Docking Studies
4. Materials and Methods
4.1. Enzyme Inhibition Assays
4.2. Molecular Modeling
4.3. Chemistry
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACh | acetylcholine |
| AChE | acetylcholinesterase |
| AD | Alzheimer’s disease |
| EtOAc | ethyl acetate |
| EU | European Union |
| BBB | blood-brain barrier |
| BChE | butyrylcholinesterase |
| CAS | catalytic anionic subsite |
| ChE | cholinesterase |
| DIAD | diisopropyl azodicarboxylate |
| DIEA | N,N-diisopropylethylamine |
| DMF | N,N-dimethylformamide |
| DMSO | dimethylsulfoxide |
| FAD | flavin adenine dinucleotide |
| FDA | Food and Drug Administration |
| GABA | γ-amminobutirric acid |
| HBA | hydrogen bond acceptor |
| HBD | hydrogen bond donor |
| HIV | human immunodeficiency virus |
| HRMS | high-resolution mass |
| MAO A | monoamine oxidase A |
| MAO B | monoamine oxidase B |
| MTDL | multi-target-directed ligand |
| PAS | peripheral anionic subsite |
| PD | Parkinson’s disease |
| PDB | protein data bank |
| Q-TOF | quadrupole-time of flight |
| THF | tetrahydrofuran |
| TPSA | topological polar surface area |
References
- Alzheimer’s Association. 2017 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2017, 13, 325–373. [Google Scholar] [CrossRef]
- Prince, M.; Comas-Herrera, A.; Knapp, M.; Guerchet, M.; Karagiannidou, M. World Alzheimer Report 2016: Improving Healthcare for People Living with Dementia; Alzheimer’s Disease International (ADI): London, UK, 2016; pp. 1–140. [Google Scholar]
- Querfurth, H.W.; LaFerla, F.M. Alzheimer’s disease: Mechanism of disease. N. Engl. J. Med. 2010, 362, 329–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scarpini, E.; Scheltens, P.; Feldman, H. Treatment of Alzheimer’s disease: Current status and new perspectives. Lancet. Neurol. 2003, 2, 539–547. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, A.; Ekavali. A review on Alzheimer’s disease pathophysiology and its management: An update. Pharm. Rep. 2015, 67, 195–203. [Google Scholar] [CrossRef]
- Atri, A.; Hendrix, S.B.; Pejović, V.; Hofbauer, R.K.; Edwards, J.; Molinuevo, J.L.; Graham, S.M. Cumulative, additive benefits of memantine-donepezil combination over component monotherapies in moderate to severe Alzheimer’s dementia: A pooled area under the curve analysis. Alzheimers. Res. 2015, 7, 28. [Google Scholar] [CrossRef] [Green Version]
- Oliveira Pedrosa, M.; Duarte da Cruz, R.; Oliveira Viana, J.; de Moura, R.; Ishiki, H.; Barbosa Filho, J.; Diniz, M.; Scotti, M.; Scotti, L.; Bezerra Mendonca, F. Hybrid compounds as direct multitarget ligands: A review. Curr. Top. Med. Chem. 2016, 17, 1044–1079. [Google Scholar] [CrossRef]
- Park, J.-H.; Ju, Y.H.; Choi, J.W.; Song, H.J.; Jang, B.K.; Woo, J.; Chun, H.; Kim, H.J.; Shin, S.J.; Yarishkin, O.; et al. Newly developed reversible MAO-B inhibitor circumvents the shortcomings of irreversible inhibitors in Alzheimer’s disease. Sci. Adv. 2019. [Google Scholar] [CrossRef] [Green Version]
- Riederer, P.; Danielczyk, W.; Grünblatt, E. Monoamine oxidase-B inhibition in Alzheimer’s disease. Neurotoxicology. 2004, 25, 271–277. [Google Scholar] [CrossRef]
- Pisani, L.; Muncipinto, G.; Miscioscia, T.F.; Nicolotti, O.; Leonetti, F.; Catto, M.; Caccia, C.; Salvati, P.; Soto-Otero, R.; Mendez-Alvarez, E.; et al. Discovery of a novel class of potent coumarin monoamine oxidase B inhibitors: Development and biopharmacological profiling of 7-[(3-chlorobenzyl) oxy]-4-[(methylamino)methyl]-2H-chromen-2-one methanesulfonate (NW-1772) as a highly potent, selective, rever. J. Med. Chem. 2009, 52, 6685–6706. [Google Scholar] [CrossRef]
- Binda, C.; Wang, J.; Pisani, L.; Caccia, C.; Carotti, A.; Salvati, P.; Edmondson, D.E.; Mattevi, A. Structures of human monoamine oxidase B complexes with selective noncovalent inhibitors: Safinamide and coumarin analogs. J. Med. Chem. 2007, 50, 5848–5852. [Google Scholar] [CrossRef]
- Pisani, L.; De Palma, A.; Giangregorio, N.; Miniero, D.V.; Pesce, P.; Nicolotti, O.; Campagna, F.; Altomare, C.D.; Catto, M. Mannich base approach to 5-methoxyisatin 3-(4-isopropylphenyl)hydrazone: A water-soluble prodrug for a multitarget inhibition of cholinesterases, beta-amyloid fibrillization and oligomer-induced cytotoxicity. Eur. J. Pharm. Sci. 2017, 109, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Pisani, L.; Catto, M.; De Palma, A.; Farina, R.; Cellamare, S.; Altomare, C.D. Discovery of potent dual binding site acetylcholinesterase inhibitors via homo- and heterodimerization of coumarin-based moieties. ChemMedChem 2017, 12, 1349–1358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pisani, L.; Iacobazzi, R.M.; Catto, M.; Rullo, M.; Farina, R.; Denora, N.; Cellamare, S.; Altomare, C.D. Investigating alkyl nitrates as nitric oxide releasing precursors of multitarget acetylcholinesterase-monoamine oxidase B inhibitors. Eur. J. Med. Chem. 2019, 161, 292–309. [Google Scholar] [CrossRef] [PubMed]
- López-Rojas, P.; Janeczko, M.; Kubiński, K.; Amesty, Á.; Masłyk, M.; Estévez-Braun, A. Synthesis and antimicrobial activity of 4-substituted 1,2,3-triazole-coumarin derivatives. Molecules 2018, 23, 199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frederik, R.; Robert, S.; Charleir, C.; de Ruyck, J.; Wouters, J.; Pirotte, B.; Masereel, B.; Pochet, L. 3,6-Disubstituted coumarins as mechanism-based inhibitors of thrombin and factor Xa. J. Med. Chem. 2005, 48, 7592–7603. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Yan, B.; Peterson, L.B.; Blagg, B.S.J. 3-Arylcoumarin derivatives manifest anti-proliferative activity through Hsp90 inhibition. ACS Med. Chem. Lett. 2012, 3, 327–331. [Google Scholar] [CrossRef]
- Lee, S.; Sivakumar, K.; Shin, W.-S.; Xie, F.; Wang, Q. Synthesis and anti-angiogenesis activity of coumarin derivatives. Bioorg. Med. Chem. Lett. 2006, 16, 4596–4599. [Google Scholar] [CrossRef]
- Yu, D.; Suzuki, M.; Xie, L.; Morris-Natschke, S.L.; Lee, K.-H. Recent progress in the development of coumarin derivatives as potent anti-HIV agents. Med. Res. Rev. 2003, 23, 322–345. [Google Scholar] [CrossRef]
- Reid, G.A.; Chilukuri, N.; Darvesh, S. Butyrylcholinesterase and the cholinergic system. Neuroscience 2013, 234, 53–68. [Google Scholar] [CrossRef] [Green Version]
- Pisani, L.; Catto, M.; Nicolotti, O.; Grossi, G.; Di Braccio, M.; Soto-Otero, R.; Mendez-Alvarez, E.; Stefanachi, A.; Gadaleta, D.; Carotti, A. Fine molecular tuning at position 4 of 2H-chromen-2-one derivatives in the search of potent and selective monoamine oxidase B inhibitors. Eur. J. Med. Chem. 2013, 70, 723–739. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharm. 1961, 7, 88–90. [Google Scholar] [CrossRef]
- Pisani, L.; Farina, R.; Nicolotti, O.; Gadaleta, D.; Soto-Otero, R.; Catto, M.; Di Braccio, M.; Mendez-Alvarez, E.; Carotti, A. In silico design of novel 2H-chromen-2-one derivatives as potent and selective MAO-B inhibitors. Eur. J. Med. Chem. 2015, 89, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Bolognesi, M.L. Harnessing polypharmacology with medicinal chemistry. ACS Med. Chem. Lett. 2019, 10, 273–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nochi, S.; Asakawa, N.; Sato, T. Kinetic study on the inhibition of acetylcholinesterase by 1-benzyl-4-((5,6-dimethoxy-1-indanon)-2-yl)methylpiperidine hydrochloride (E2020). Biol. Pharm. Bull. 1995, 18, 1145–1147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inestrosa, N.C.; Dinamarca, M.C.; Alvarez, A. Amyloid-cholinesterase interactions: Implications for Alzheimer’s disease. Febs. J. 2008, 275, 625–632. [Google Scholar] [CrossRef]
- Gerlits, O.; Ho, K.Y.; Cheng, X.; Blumenthal, D.; Taylor, P.; Kovalevsky, A.; Radić, Z. A new crystal form of human acetylcholinesterase for exploratory room-temperature crystallography studies. Chem. Biol. Interact. 2019, 309, 108698. [Google Scholar] [CrossRef]
- Messerer, R.; Dallanoce, C.; Matera, C.; Wehle, S.; Flammini, L.; Chirinda, B.; Bock, A.; Irmen, M.; Tränkle, C.; Barocelli, E.; et al. Novel bipharmacophoric inhibitors of the cholinesterases with affinity to the muscarinic receptors M 1 and M 2. MedChemComm 2017, 8, 1346–1359. [Google Scholar] [CrossRef]
- Cheung, J.; Rudolph, M.J.; Burshteyn, F.; Cassidy, M.S.; Gary, E.N.; Love, J.; Franklin, M.C.; Height, J.J. Structures of human acetylcholinesterase in complex with pharmacologically important ligands. J. Med. Chem. 2012, 55, 10282–10286. [Google Scholar] [CrossRef]
- Schrödinger Release 2018-4: Maestro; Schrödinger, LLC: New York, NY, USA, 2018.
- Knez, D.; Coquelle, N.; Pišlar, A.; Žakelj, S.; Jukič, M.; Sova, M.; Mravljak, J.; Nachon, F.; Brazzolotto, X.; Kos, J.; et al. Multi-target-directed ligands for treating Alzheimer’s disease: Butyrylcholinesterase inhibitors displaying antioxidant and neuroprotective activities. Eur. J. Med. Chem. 2018, 156, 598–617. [Google Scholar] [CrossRef]
- Cieplak, P.; Bayly, C.I.; Gould, I.R.; Merz, K.M.; Ferguson, D.M.; Spellmeyer, D.C.; Fox, T.; Caldwell, J.W.; Kollman, P.A. A 2nd generation force-field for the simulation of proteins, nucleic-acids, and organic-molecules. J. Am. Chem. Soc. 1995, 117, 5179–5197. [Google Scholar]
- QUACPAC 1.7.0.2: OpenEye Scientific Software, Santa Fe, NM. Available online: http://www.eyesopen.com (accessed on 1 October 2019).
- Morris, G.M.; Goodsell, D.S.; Halliday, R.S.; Huey, R.; Hart, W.E.; Belew, R.K.; Olson, A.J. Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J. Comput. Chem. 1998, 19, 1639–1662. [Google Scholar] [CrossRef] [Green Version]
- Forli, S.; Olson, A.J. A force field with discrete displaceable waters and desolvation entropy for hydrated ligand docking. J. Med. Chem. 2012, 55, 623–638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pisani, L.; Barletta, M.; Soto-Otero, R.; Nicolotti, O.; Mendez-Alvarez, E.; Catto, M.; Introcaso, A.; Stefanachi, A.; Cellamare, S.; Altomare, C.; et al. Discovery, biological evaluation, and structure–activity and -selectivity relationships of 6′-substituted (E)-2-(benzofuran-3(2H)-ylidene)-N-methylacetamides, a novel class of potent and selective monoamine oxidase inhibitors. J. Med. Chem. 2013, 56, 2651–2664. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 3–27 are available from the authors. |







 | ||||||||
| General structure | entry | R1 | R2 | R3 | MAO A a,b | MAO B a,b | AChE a,c | BChE a,d |
| A | 3 | Cl | H | H | 0.65 ± 0.02 | 0.0039 ± 0.0008 | 6.2 ± 0.7 | 43 ± 5% |
| 4 | NMe2 | H | H | 49 ± 1% | 0.57 ± 0.05 | 6.0 ± 1.5 | 34 ± 5% | |
| 5 | OMe | H | OMe | 1.0 ± 0.1 | 0.067 ± 0.005 | 11 ± 2 | 20 ± 4% | |
| 6 | –OCH2O– | H | 1.2 ± 0.1 | 0.0075 ± 0.0001 | 8.5 ± 1.8 | 3.4 ± 2.1 | ||
| 7 | H | H | H | 0.53 ± 0.01 | 0.0021 ± 0.0002 | 26 ± 5% | 17 ± 1% | |
| 8 | Br | H | H | 2.31 ± 0.04 | 0.0022 ± 0.0001 | 2.00 ± 0.04 | 2.9 ± 0.4 | |
| 9 | F | H | H | 1.74 ± 0.04 | 0.0013 ± 0.0001 | 2.7 ± 1.0 | 4.8 ± 0.3 | |
| General structure | entry | R1 | R2 | MAO A | MAO B | AChE | BChE | |
| B | 10 | H | Me | 1.0 ± 0.1 | 0.17 ± 0.01 | 7.2 ± 0.6 | 46 ± 2% | |
| 11 | Me | H | 0.58 ± 0.03 | 0.068 ± 0.002 | 47.1 ± 0.3% | 48.1 ± 0.6% | ||
| 12 | Cl | H | 48 ± 1% | 1.0 ± 0.1 | 7.9 ± 0.4 | 27 ± 2% | ||
| General structure | entry | R1 | R2 | MAO A | MAO B | AChE | BChE | |
| C | 13 | Bn | H | 24 ± 3% | 0.103 ± 0.001 | 17 ± 1 | 3.4 ± 1.3 | |
| 14 | Bn | Me | 28 ± 1% | 0.332 ± 0.026 | 16 ± 1 | 6.3 ± 0.3 | ||
| 15 | pyrrolidin-1-yl | 1.12 ± 0.01 | 0.152 ± 0.020 | 6.6 ± 0.9 | 14 ± 2% | |||
| 16 | 4-methylpiperazin-1-yl | 32 ± 5% | 0.273 ± 0.011 | 3.1 ± 0.6 | 2.3 ± 0.9 | |||
| 17 | morpholin-1-yl | 32 ± 3% | 0.172 ± 0.001 | 6.6 ± 1.1 | 46 ± 2% | |||
| 18 | H | H | 46 ± 3% | 0.066 ± 0.015 | 17 ± 4 | 20 ± 7% | ||
| 19 | Me | Me | 9.2 ± 0.7 | 0.213 ± 0.002 | 8.3 ± 2.0 | 7.0 ± 1.1 | ||
| 20 | nPr | H | 48 ± 1% | 0.0094 ± 0.0035 | 4.9 ± 1.4 | 3.6 ± 0.8 | ||
| 21 | nBu | H | 41 ± 3% | 0.164 ± 0.002 | 39 ± 1% | 4.8 ± 0.3 | ||
| 22 | CH2C≡CH | H | 1.1 ± 0.1 | 0.0041 ± 0.0011 | 2.1 ± 0.2 | 33 ± 6% | ||
| 23 | CH2C≡CH | Me | 1.7 ± 0.1 | 0.084 ± 0.002 | 5.6 ± 0.7 | 28 ± 1% | ||
| 24 | CH2CONH2 | H | 0.077 ± 0.014 | 0.061 ± 0.013 | 2.2 ± 0.7 | 13 ± 2% | ||
| 25 | CH2CONHMe | H | 2.6 ± 0.1 | 0.0011 ± 0.0003 | 5.3 ± 1.4 | <5% | ||
| 26 | CH2CONMe2 | H | 2.5 ± 0.5 | 0.030 ± 0.001 | 4.3 ± 0.6 | 34 ± 2% | ||
| D | 27 | CH2CONH2 | OMe | 5.9 ± 0.5 | 0.013 ± 0.001 | 4.4 ± 0.8 | 41 ± 2% | |
| safinamide | 18 ± 3% | 0.031 ± 0.001 | ||||||
| donepezil | 0.021 ± 0.002 | 2.3 ± 0.2 | ||||||
| Entry | MW a | TPSA (Å2) a | HBA a | HBD a | cLogP b | BBB c | RO5 violation a |
|---|---|---|---|---|---|---|---|
| 6 | 339.34 | 69.93 | 6 | 1 | 2.89 | + | 0 |
| 8 | 374.23 | 51.47 | 4 | 1 | 3.80 | + | 0 |
| 9 | 313.32 | 51.47 | 5 | 1 | 3.08 | + | 0 |
| 16 | 398.88 | 45.92 | 5 | 0 | 3.53 | + | 0 |
| 20 | 357.83 | 51.47 | 4 | 1 | 4.69 | + | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rullo, M.; Catto, M.; Carrieri, A.; de Candia, M.; Altomare, C.D.; Pisani, L. Chasing ChEs-MAO B Multi-Targeting 4-Aminomethyl-7-Benzyloxy-2H-Chromen-2-ones. Molecules 2019, 24, 4507. https://doi.org/10.3390/molecules24244507
Rullo M, Catto M, Carrieri A, de Candia M, Altomare CD, Pisani L. Chasing ChEs-MAO B Multi-Targeting 4-Aminomethyl-7-Benzyloxy-2H-Chromen-2-ones. Molecules. 2019; 24(24):4507. https://doi.org/10.3390/molecules24244507
Chicago/Turabian StyleRullo, Mariagrazia, Marco Catto, Antonio Carrieri, Modesto de Candia, Cosimo Damiano Altomare, and Leonardo Pisani. 2019. "Chasing ChEs-MAO B Multi-Targeting 4-Aminomethyl-7-Benzyloxy-2H-Chromen-2-ones" Molecules 24, no. 24: 4507. https://doi.org/10.3390/molecules24244507