Omic insights into various ceftazidime-avibactam-resistant Klebsiella pneumoniae isolates from two southern Italian regions
- 1Microbiology Section, Dept of Biomedical and Biotechnological Science, University of Catania, Catania, Italy
- 2Unit of Clinical Microbiology, Department of Health Sciences, “Magna Graecia” University, Catanzaro, Italy
- 3Unit of Infectious and Tropical Diseases, Department of Medical and Surgical Sciences, “Magna Graecia” University, Catanzaro, Italy
- 4Unità Operativa Complessa (UOC) Laboratory Analysis, University Hospital Policlinico-San Marco, Catania, Italy
Ceftazidime-avibactam (CZA) is one of the best therapeutic options available for infections caused by Klebsiella pneumoniae carbapenemase (KPC)-producing bacteria. However, sporadic reports of CZA-resistant strains have been rapidly increasing in patients. Herein, we provide detailed case reports of the emergence of ceftazidime-avibactam resistance to identify their resistome and virulome using genomic molecular approaches. Sixteen isolates were collected from 13 patients at three hospitals in Catania and Catanzaro (Italy) between 2020-2021. Antimicrobial susceptibility was determined by broth microdiluition. The samples included in study were analyzed for resistome, virulome and Sequence Type (ST) using Whole Genome Sequencing (WGS). All strains were resistant to ceftazidime/avibactam, ciprofloxacin, extended-spectrum cephalosporins and aztreonam, 13/16 to meropenem, 8/16 to colistin and 7/16 to fosfomycin; 15/16 were susceptible to meropenem/vaborbactam; all strains were susceptible to cefiderocol. Molecular analysis showed circulation of three major clones: ST101, ST307 and ST512. In 10/16 strains, we found a blaKPC-3 gene; in 6/16 strains, four different blaKPC variants (blaKPC28-31-34-50) were detected. A plethora of other beta-lactam genes (blaSHV28-45-55-100-106-187-205-212, blaOXA1-9-48, blaTEM-181 and blaCTX-M-15) was observed; blaOXA-9 was found in ST307 and ST512, instead blaOXA48 in one out four ST101 strains. With regard to membrane permeability, ompK35 and ompK36 harbored frameshift mutations in 15/16 strains; analysis of ompK37 gene revealed that all strains harbored a non-functional protein and carry wild-type PBP3. There is an urgent need to characterize the mechanisms underlying carbapenem resistance and the intrinsic bacterial factors that facilitate the rapid emergence of resistance. Furthermore, it is becoming increasingly important to explore feasible methods for accurate detection of different KPC enzymes.
Introduction
For KPC-producing Enterobacterales are categorized as one of the most dangerous pandemics in the history of Gram-negative bacteria, particularly due to Klebsiella pneumoniae ST258 and variants (Patel and Bonomo, 2013).These strains have been reported in many countries, including Italy (Giani et al., 2013; Gona et al., 2014), where the majority of CZA-resistant KPC-producing K. pneumoniae were observed in the sequence type 512 (ST512) multi-drug resistance (MDR) clone, with some recent reports of ST101 and ST307 as well (Carattoli et al., 2021).
Currently, ceftazidime/avibactam (CZA), a combination of a third-generation, broad-spectrum cephalosporin and a β-lactamase inhibitor that inactivates ESBLs (TEM, SHV, CTX-M), AmpC and carbapenemases (KPC-2, KPC-3, OXA-10 and OXA-48), but not metallo-enzymes (Shirley, 2018), is one of the best therapeutic options available for infections caused by such isolates. However, emergence of CZA resistance in carbapenemase-producing Enterobacterales (CPE) has been reported in patients (Shields et al., 2017; Giddins et al., 2018). Several mechanisms of CZA resistance have been linked to specific mutations in the blaKPC gene (Shields et al., 2017; Giddins et al., 2018) and to permeability defects (porin deficiency), often combined with increased expression of KPC or ESBL determinants (Humphries and Hemarajata, 2017).
From a clinical standpoint, it is important to understand the cross-resistance pattern of these microorganisms in order to optimize drug selection and guide treatment strategies. In this sense, it is mandatory to characterize the genes involved in phenotypic and clinical resistance by means of rapid tests.
In this study, we described the characterization of CZA-resistant KPC-producing K. pneumoniae strains isolated in three different hospitals. The great majority of these strains retained activity to meropenem/vaborbactam, while all strains were susceptible to cefiderocol. Three different ST-types were identified, and whole genome sequencing (WGS) analysis revealed differences in CZA resistance mechanisms resulting from the complex resistome and virulome profiles of these isolates.
Materials and methods
Sample
The present study was conducted at three hospitals located in Catania (A.O.U. Policlinico “G. Rodolico - San Marco” and “A.O. Cannizzaro”) and Catanzaro (“Mater Domini” University Hospital of the “Magna Graecia” University) in the period between May 2020 and October 2021. Included were all CZA-resistant KPC-producing K. pneumoniae isolates collected from each patient. For a same patient, more than one strain was included if isolated from two different clinical samples collected at least two weeks apart from each other. Identification and antimicrobial susceptibility had been previously performed by the Vitek 2 system (bioMerieux, Marcy l’Etoile, France) at the above mentioned hospitals and re-confirmed by standard methods (EUCAST, 2022).
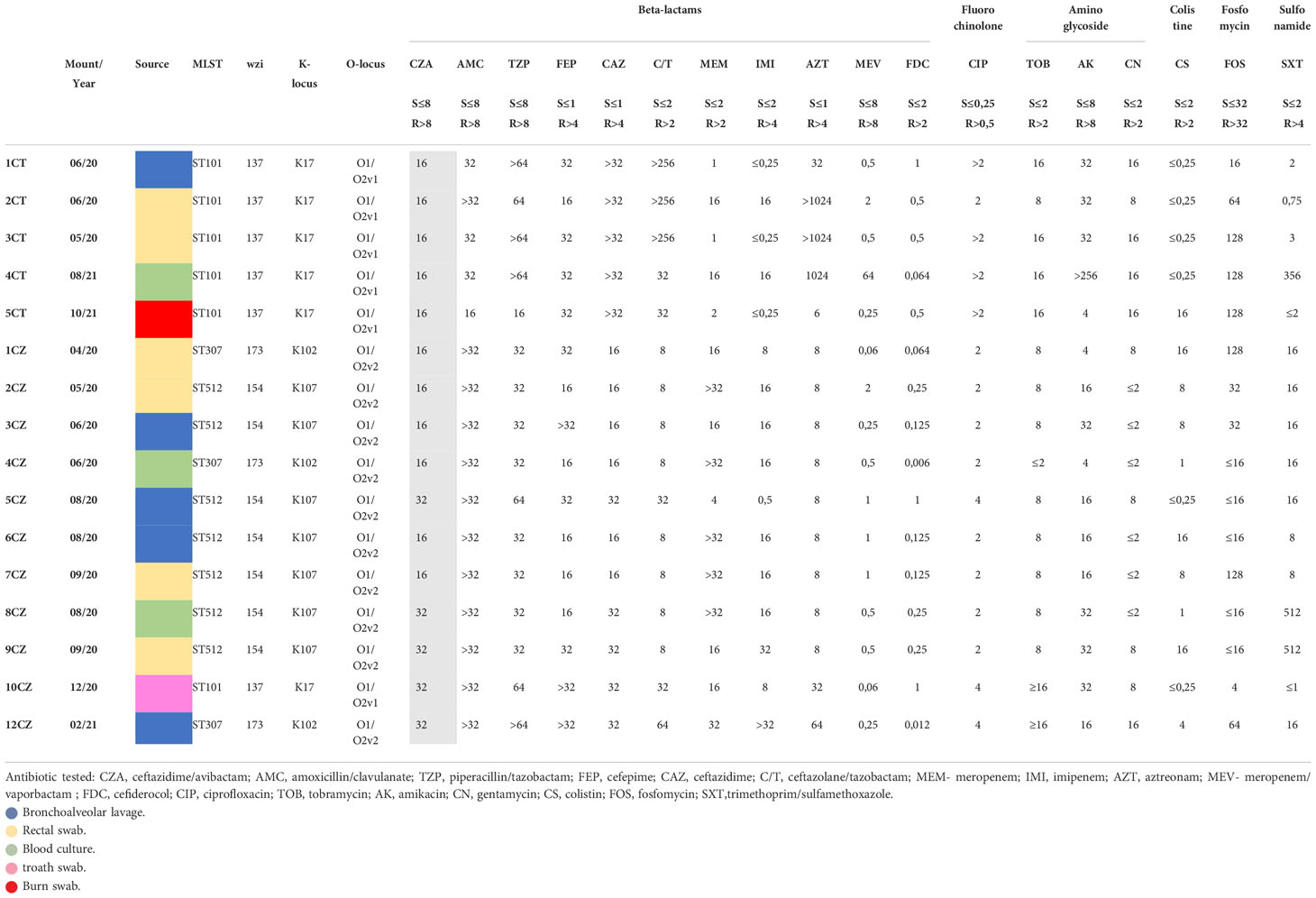
In addition, minimum inhibitory concentrations (MICs) were performed at the microbiology laboratories involved, using broth microdilution as described by the CLSI guidelines (Clinical and Laboratory Standards Institute, 2015). The following antibiotics were tested: ceftazidime/avibactam (CZA), amoxicillin/clavulanate (AMC), piperacillin/tazobactam (TZP), cefepime (FEP), ceftazidime (CAZ), ceftazolane/tazobactam (C/T), meropenem (MEM), imipenem (IMI), aztreonam (AZT), meropenem/vaporbactam (MEV), cefiderocol (FDC), ciprofloxacin (CIP), tobramycin (TOB), amikacin (AK), gentamycin (CN), colistin (CS), fosfomycin (FOS), trimethoprim/sulfamethoxazole (SXT). Breakpoints of antibiotics for the interpretative criteria for clinical isolates were used according to the EUCAST v 12.0 (EUCAST, 2022). E. coli ATCC 25922 was used as the quality control strain. Sample characteristics, date of isolation, patient demographics and therpy are reported in Table S1.
DNA extraction
DNA extraction was carried out following the manufacturer’s instructions provided by QIAGEN QIAamp® DNA Mini Kit (Ref. 51304, QIAGEN, 40724 Hilden, Germany).
DNA was quantified using both the Eppendorf BioPhotometer® D30 and the fluorimeter Qubit dsDNA BR Assay Kit to evaluate purity and quantity of the initial sample, respectively (Ref. 32850, Invitrogen, 92008 Carlsbad, CA, USA). The results of quantification are shown in Table S2.
NGS Sequencing
A concentration of 100 ng of each sample was used for NGS sequencing. This was performed at the Molecular Biology laboratory of University of Catania on a Illumina MiSeq platform according to the manufacturer’s instructions provided in the Illumina DNA Prep – (M) Tagmentation for Illumina® (Ref. 20018707, Illumina, Inc., 92122, San Diego, CA, USA). Indexes were provided with Nextera™ DNA CD Indexes Illumina® (24 Indexes, 24 Samples) (Ref. 20019105, Illumina, Inc., 92122, San Diego, CA, USA).
Libraries were quantified and their quality evaluated using both the fluorometric Qubit dsDNA HS Assay Kit (Ref. Q32851, Invitrogen, Carlsbad, CA 92008, USA) and the Agilent® High Sensitivity DNA Kit (Ref. 5067-4626).
Denature and dilute libraries were performed following the “Denature and Dilute Libraries Guide” protocol provided by Illumina®, choosing 8,5 pM as the loading concentration. Finally, sequencing was performed using the MiSeq Reagent Kits v3 (Ref. 15043895, Illumina, Inc., 92122, San Diego, CA, USA). The Sample Sheet was created using the Local Run Manager v3 software, and following the instructions in the Local Run Manager v3 Software Guide provided by Illumina (Software Guide provided by Illumina (Local run manager generate FASTQ analysis module workflow guide, 2018)).
Data analysis
Data were analyzed using the QIAGEN CLC Genomics Workbench software and following the User Manual for the CLC Microbial Genomics Module v22.0, released on January 4, 2022 (QIAGEN, Aarhus, 8000 Denmark), that uses the CARD database to assign resistance, virulence and MLST genes (https://card.mcmaster.ca/).
Bioinformatic analysis
Sixteen paired-end bacterial raw reads were firstly trimmed with TrimGalore (v0.5.0) (Martin, 2011; Krueger, 2022) to remove the adapter sequence. After that, bacterial genome was assembled de novo using Unicycler (v0.4.8) (Wick et al., 2017) with the Illumina only assembly modality. Known virulence factors, resistance genes and capsule loci were identified using Kleborate (v2.2.0) and the Kaptive command (Wyres et al., 2016; Lam et al., 2021). Prokka (v1.13) (Seemann, 2014) was used for bacterial annotation. Moreover, Unicycler output assemblies were aligned with several protein sequences in order to identify punctual mutations.
Thereafter, files were sorted by SAM tools, variants were called and consensus sequences were generated using BCTftools (Li, 2011; Danecek et al., 2021).
Statistical analysis
In order to establish a potential correlation between beta-lactams MIC values and related genes, Pearson’s correlation analyses were performed for the 16 isolates. This correlation matrix is a statistical tool that measures the linear correlation between two variables, X and Y. It has a value between +1 and −1, where +1 indicates total positive linear correlation, 0 no linear correlation, and −1 total negative linear correlation. The correlation coefficient ranges from −1 to 1, where 1 implies that a linear equation perfectly describes the relationship between X and Y, with all data points lying on a line for which Y increases as X increases. In addition to this, R-Squared was also calculated. This measures the reliability of the linear relationship between the variables included in the model. Its value is between 0 (fully correlated variables) and 1 (unrelated variables).
For all statistical comparisons, a significance level of p <0.05 was considered to show differences between the groups. Data analysis was performed using Prims Version 9.4.0, June 3, 2022.
Results
Clone characteristics
A total of 16 CZA-resistant KPC-producing K. pneumoniae strains were isolated from clinical samples collected from 13 patients and included in the study (Table S1, Supplementary Material 1). They were MDR and resistant to CZA, with MICs one or two dilutions higher than the clinical breakpoint.
Isolates 1/2CT, 6/7CZ, 8/9CZ were collected from the same patients. In 1/2CT and 6/7CZ patients have not been treated with CZA during the previous 12 months (Table S1). All strains were also resistant to ciprofloxacin, extended-spectrum cephalosporins and aztreonam, 13/16 were resistant to meropenem, while 15/16 (MIC range of 4->32 mg/L) were susceptible to meropenem/vaborbactam (MIC range 0.006-0.5 mg/L); all strains were fully susceptible to cefiderocol (MIC range 0.006-0.5 mg/L). With regard to colistin and fosfomycin, resistance was observed in 8/16 and 7/16 strains, respectively.
All strains were genomically identified as belonging to three different STs, namely ST101, 307 and 512, whereas the LPS O-antigen, composed of D-galactans, was encoded by genes that differentiate between two serotypes, i.e., O1/O2v1 and O1/O2v2, distinguished by rearrangements detected in the rfb region, and possessed three different capsular polysaccharides (K locus) K17, 102, 107 linked to the wzi gene sequence (Table 1).
Resistome analyses
A common core of resistance genes was detected in all isolates, namely: ampH (E. coli beta-lactamase), kpnE-F-G (K. pneumoniae efflux pump), oqxA-B (quinolone - efflux pump), lptD (LPS assembly protein – efflux pump), mdfA (E. coli quinolone - efflux pump), arnT and eptB (altering cell wall), and ompA (protein modulating permeability to antibiotics).
Furthermore, the 16 CZA-resistant strains revealed, together with blaKPC genes, the constant presence of different blaSHV alleles (blaSHV-28-45-55-100-106-187-205-212) and different blaOXA and blaTEM genes in their resistome profile. Regarding acquired resistome, different blaKPC variants were detected in all isolates, namely 3, 28, 31, 34; blaKPC-3 in 10/16 isolates, blaKPC-31 in 3/16, blaKPC-28, blaKPC-34 and blaKPC-50 in one isolate (Table 2).
The blaCTX-M-15 gene was detected in two isolates, while a plethora of other acquired determinants associated with resistance to beta-lactams (blaOXA-1-9-48 and blaTEM-181), aminoglycosides (aph, AAC, aad, ANT and armA), sulfonamides (sul1 and sul2), trimetroprim (dfrA12 and dfrA14), quinolones (qnrS1 and qnrB17), macrolides (mphA and mphE) and phenicoles (catB3 and catL) were also variably present. The fosA6 gene, associated with fosfomycin resistance, was detected in 10/16 strains, independently linked to the MIC values of this drug. In five strains out of 16, the armA gene for 16rRNA methylase was detected. Overall, the resistome was generally consistent with the resistance profile.
Remarkable differences were observed when comparing CZA, MEM and MEV susceptibility profiles with their resistome as described in Table 3.
CZA resistance correlated with the presence of blaKPC3 variants in 6 isolates (harboring the following variants: 31, 34, 28, 50), while in one strain (5CT), a blaKPC3 variant with a deletion of two amino acids (Glu and Leu) in the 167 and 168 (498del-6 nucleotide GAG/CTG) position was found. In strain 4CT, resistant amongst others to MER/VAB, the presence of a wild type blaKPC-3 together with blaOXA-48 and blaSHV variants was associated with a full resistance profile. The remaining strains harboring wild-type blaKPC3 showed a complex array of blaSHV variants, together with blaTEM-181; in two strains, namely 1CZ and 5CZ, blaCTX-M-15 was found.
Membrane permeability was also investigated: in 16/17 strains ompK35 harbored frameshift mutations while ompK36 showed variants (insertions and SNPs), only in CZ1 wild type proteins were detected. With regard to ompK37, all strains harbored a non-functional protein. Our isolates carried wild-type PBP3 genes.
A statistical correlation analysis of all beta-lactamase genes was performed using the Pearson coefficient (Figure S1), which showed a direct significant association between the presence of blaSHV187 with blaOXA-9 (0.75) and blaTEM-181, between blaSHV-28 and blaSHV-100 (0.51), blaSHV-105 and blaSHV-106 (0.71), between blaSHV-100 and blaKPC-28 (0.68), with the most significant association found for blaSHV-45 and blaSHV-55, blaSHV-106 and blaSHV-205, blaOXA-1 and blaCTX-M-15, and blaOXA-9 and blaTEM-181 (1).
Virulome analysis
The Virulence Factor Database was used to predict and compare virulence genes. All CZA-resistant isolates carried a large array of virulence determinants (Table 4).
As regards the genes involved in capsule production, we found that galF and the genes responsible for the upregulation of capsule production (rcsAB system) were present in the whole sample (rcsA alone in 93,75% (15/16)); rpmA2, that regulates the mucoid phenotype, in 25% (4/16) only.
Genes for LPS synthesis were variably present: glf in 100%, wzm in 93,75% (15/16), wbbN-O in 68,75% (11/16), kfoC in 62,5% (10/16), wzt in 37,5 (6/16), and wbbM in 31,25% (5/16) of samples. The efflux pump arcAB gene was present in the whole sample. Furthermore, a complete set of genes for type I (fimA-K) and type III (mrkA-B-C-D-F-H-I-J) fimbriae was found in all isolates.
Genes for siderophores were variably present in our sample. The Yersiniabactin genes were found as follows: ybtE-P-Q-T-U, fyuA, irp1-2 in 56,25% (9/16), ybtS-X in 50% (8/16) and ybtA in 37,5% (6/16) of samples, respectively. Aerobactine genes iucA-B-C were detected in 31,2% (5/16) of strains and iutA in 25% (4/16). Among enterobactine genes, all strains (100%) carried the entB-C-D-F, fepB-C-D-G, fes and ybdA loci; entA was found in 62,5% (10/16), entE in 56,25% (9/16) and fepA in 18,75% (3/16). The iroE gene encoding salmochelin was present in the whole sample.
The arcAB, tssF-G, sciN/tssJ type VI secretion system genes, which were part of the structural core, were present in 100% of the sample, followed by clpV/tssH, dotU/tssL, hcp/tssD, vasE/tssK, vipA/tssB, vipB/tssC, found in 93,75% (15/16), impA/tssA and KPHS_23120 in 62,5% (10/16), vgrG/tssI in 56,25% (9/16), icmF/tssM in 43,75 (7/16), and tli1 only in 6,25% (1/16).
Discussion
In this study, we reported the occurrence of CZA resistance in a group of KPC-producing K. pneumoniae isolates collected in three hospitals located in two southern Italian regions during the pandemic period. All isolates represented different genotypes and were further analyzed for their virulome as well as for the consistency of their susceptibility profiles and resistome. Tracking their dissemination and understanding their evolution is an important step towards monitoring and controlling these pathogens.
Several findings are particularly noteworthy.
First of all, CZA-resistant strains showed a comparable MEM resistance profile (13/16 isolates with a MIC range of 4->32 mg/L), whereas MEV exhibited excellent in vitro activity against 15/16 isolates (MIC range 0.006-0.5 mg/L); full susceptibility to cefiderocol (MIC range 0.006-0.5 mg/L) was also observed.
MDR susceptibility profile included resistance to aminoglycosides and fluoroquinolones; 9 strains were also resistant to colistin and 7/16 to fosfomycin, in the absence of strict correlation with the presence of the fosA gene, which was found in all ST512 and ST307 isolates, but not in ST101.
Remarkable differences were observed when analyzing consistency of the CZA, MEM and MEV phenotypic profiles with the corresponding resistome. CZA resistance correlated with the presence of blaKPC3 variants in 6 isolates, in which different alleles were found (namely blaKPC31-34-28-50). One strain (5CT) harbored a blaKPC-3 variant with a deletion of two amino acids (Glu and Leu) in the 167 and 168 (498del-6 nucleotide GAG/CTG) position, as previously described (Antinori et al., 2020). All other carried blaKPC-3 wild-type genes variably associated with a plethora of other resistance determinants; among these, one strain (4CT) carried blaKPC-3 with blaOXA-48 and blaSHV-187, associated to MEV resistance.
The remaining blaKPC-3 carriers carried different alleles for blaSHV, blaTEM and blaOXA. Strains 1 and 5CZ also carried blaCTX-M-15.
Even though non-carbapenemase beta-lactamases, such as ESBLs, have the capacity to hydrolyze carbapenemase (Paterson and Bonomo, 2005), in our study we were not able to determine their contribution, suggesting – as already observed (Paterson and Bonomo, 2005) – that their presence did not increase carbapenemase resistance among isolates that already harbored a carbapenemase gene.
Adding to this complex gene acquisition, all isolates have mutation in OmpK35, OmpK36 and OmpK37, related to deletion, insertion and frameshift.
In our study, we confirmed that, in KPC producing-K. pneumoniae strains, mutations in the porine gene ompK36 were present and, as previously observed by other Authors (Venditti et al., 2021) presumably correlate with high-level resistance to carbapenems (Wong et al., 2019; David et al., 2022). In addition to the indel found in the ompK36 gene, our isolates harbored frameshift mutations in ompK35 and ompK37, leading to nonfunctional proteins, associated, also in this case, with lower carbapenemase MICs, as already described by other Authors (Gaibani et al., 2020; Venditti et al., 2021).
Outer membrane protein alterations can also influence carbapenem activity in K. pneumoniae; in fact, loss of function of one or multiple porin channels can diminish their activity.
Even though we could not identify the role of each mutation in our study, our data support the role of several biologically plausible genotypes in the determination of resistance. Interestingly, in our isolates vaborbactam maintained its ability to enter the bacterial cell despite the loss of the two major proteins OmpK35 and OmpK37. It is already known that vaborbactam crosses the outer membrane of K. pneumoniae exploiting both OmpK35 and OmpK36, with the latter being the preferred one (Venditti et al., 2021). In our isolates, the mutations found in OmpK36 were unable to affect the entry ability of the inhibitor, thus reducing the MIC values of meropenem. Generally speaking, despite this complexity, MEV (15/16 isolates) and cefiderocol were still in the range of susceptibility in all our CZA resistant isolates, proving able to maintain reliable in vitro activity against these difficult-to-treat isolates. Taken together, these data showed that a new cephalosporin with a new mechanism of entry, and vaborbactam a new beta lactamase inhibitor (BLI), can inhibit these isolates.
Finally, the 16 isolates belonged to three different STs, namely ST101, ST307 and ST512, each associated with a specific capsular locus (17, 102 and 107) and an O-antigen. ST101, found only in Catania and in one strain from Catanzaro, was associated to the O1/O2v1 variant, while both ST307 and ST512 have somatic antigen variants, i.e., O1/O2v1 and O1/O2v2 (Artyszuk et al., 2020). Of interest in these isolates is the co-presence of the rmpA2 regulator and the iutA aerobactin in 4 strains from Catania, indicating a hypermucoviscosity phenotype (Paczosa and Mecsas, 2016; Wang et al., 2020). Furthermore, all ST101 isolates carried the wzi gene, encoding the surface protein involved in capsule attachment to the outer membrane (Gona et al., 2019).
Conclusion
In conclusion, CZA has definitely become an important first-line option for KPC-Kp infections. With this increased use, the expected resistance will continue to emerge, both through the spread of high-level epidemic clones and the dissemination of plasmids carrying mutant genes.
The results of this study provide further evidence of the plasticity and evolutionary potential of CAZ and MER-resistant clones of K. pneumoniae, showing evidence of multiple adaptation and raising concerns about the strong selective pressure acting on all drugs, including novel carbapenemase inhibitors.
The convergence of clinically relevant resistance and virulence determinants, including hypervirulent clones, may enable the parallel evolution of resistance and virulence, which is a worrisome event.
With the increasing number of MDR lineages harboring variants of resistance determinants, it becomes mandatory to detect and track their dissemination by using high throughput technologies.
Further studies are urgently needed to validate the efficacy of new drugs in case of CZA resistance, under the guide of advanced molecular resistance tests in conjunction with phenotypic results and eventually synergy analyses.
Data availability statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found at: http://www.ncbi.nlm.nih.gov/bioproject/PRJNA866305.
Author contributions
The first author named is lead and corresponding author. We describe contributions to the paper using the taxonomy provided above. Writing – Original Draft: DB, DAB, MM, GP, and SS. Writing – Review and Editing: GM, CT, and SS. Conceptualization: GM, CT, SS, and MM. Investigation: DAB and CC. Methodology: DB, ET, AR, NM, and AQ. Software: NiM and GP. Formal Analysis: NiM, DB, and DAB. Validation: ET, AR, NM, AQ, and GS. Visualization: DB, NM, MM, AQ, and GS. Project Administration, Funding Acquisition and Resouces: GM, CT, and SS. All authors contributed to the article and approved the submitted version.
Funding
The manuscript was partially supported by: a research project grant, PRIN2020 CODICO PROGETTO 202089LLEH_002 from the Ministry of Research (MIUR) Italy, and by the research project Grant of European Union - FSE-REACT-EU, PON Ricerca e Innovazione 2014-2020 DM1062/2021.
Acknowledgments
The authors would like to acknowledge the BRIT laboratory at the University of Catania (Italy) for the valuable technical assistance and the use of its facility. The authors wish to thank the PharmaTranslated (http://www.pharmatranslated.com/access data 27 July 2022) and in particular to Silvia Montanari. The authors would like to acknowledge: Dr Marianna Perez, Dr Carmelo Iacobello, Dr Ausilia Aprile, Dr Adriana Tempesta, Dr Luigia Gallo, Dr Carolina Di Cello, Dr Valentina Esposito, Dr Chiara Costa, Dr Francesca Serapide, Dr Paolo Fusco, Dr Rosaria Lionello, Dr Chiara Davoli, Dr Valentina La Gamba, Dr Vincenzo Scaglione, Prof Federico Longhini, Prof Eugenio Garofalo, Prof Andrea Bruni.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcimb.2022.1010979/full#supplementary-material
References
Antinori, E., Unali, I., Bertoncelli, A., Mazzariol, A. (2020). Klebsiella pneumoniae carbapenemase (KPC) producer resistant to ceftazidime-avibactam due to a deletion in the blaKPC3 gene. Clin. Microbiol. Infect. 26 (7), 946.e1–946.e3. doi: 10.1016/j.cmi.2020.02.007
Artyszuk, D., Izdebski, R., Maciejewska, A., Kaszowska, M., Herud, A., Szijártó, V., et al. (2020). The impact of insertion sequences on O-serotype phenotype and its O-Locus-Based prediction in Klebsiella pneumoniae O2 and O1. Int. J. Mol. Sci. 21 (18), E6572. doi: 10.3390/ijms21186572
Carattoli, A., Arcari, G., Bibbolino, G., Sacco, F., Tomolillo, D., Di Lella, F. M., et al. (2021). Evolutionary trajectories toward ceftazidime-avibactam resistance in Klebsiella pneumoniae clinical isolates. Antimicrob. Agents Chemother. 65 (10), e0057421. doi: 10.1128/AAC.00574-21
Danecek, P., Bonfield, J. K., Liddle, J., Marshall, J., Ohan, V., Pollard, M. O., et al. (2021). Twelve years of SAMtools and BCFtools. Gigascience 10 (2), giab008. doi: 10.1093/gigascience/giab008
David, S., Wong, J. L. C., Sanchez-Garrido, J., Kwong, H. S., Low, W. W., Morecchiato, F., et al. (2022). Widespread emergence of OmpK36 loop 3 insertions among multidrug-resistant clones of klebsiella pneumoniae. PloS Pathog. 18 (7), e1010334. doi: 10.1371/journal.ppat.1010334
EUCAST. (2022) Clinical breakpoints and dosing of antibiotics. Available at: https://www.eucast.org/clinical_breakpoints/ (Accessed 12 July 2022).
Gaibani, P., Re, M. C., Campoli, C., Viale, P. L., Ambretti, S. (2020). Bloodstream infection caused by KPC-producing klebsiella pneumoniae resistant to ceftazidime/avibactam: Epidemiology and genomic characterization. Clin. Microbiol. Infect. 26 (4), 516.e1–516.e4. doi: 10.1016/j.cmi.2019.11.011
Giani, T., Pini, B., Arena, F., Conte, V., Bracco, S., Migliavacca, R., et al. (2013). Epidemic diffusion of KPC carbapenemase-producing Klebsiella pneumoniae in Italy: results of the first countrywide survey, 15 may to 30 June 2011. Euro. Surveill. 18 (22), 20489. doi: 10.2807/ese.18.22.20489-en
Giddins, M. J., Macesic, N., Annavajhala, M. K., Stump, S., Khan, S., McConville, T. H., et al. (2018). Successive emergence of ceftazidime-avibactam resistance through distinct genomic adaptations in blaKPC-2-Harboring Klebsiella pneumoniae sequence type 307 isolates. Antimicrob. Agents Chemother. 62 (3), e02101–e02117. doi: 10.1128/AAC.02101-17
Gona, F., Barbera, F., Pasquariello, A. C., Grossi, P., Gridelli, B., Mezzatesta, M. L., et al. (2014). In vivo multiclonal transfer of bla(KPC-3) from Klebsiella pneumoniae to escherichia coli in surgery patients. Clin. Microbiol. Infect. 20 (10), O633–O635. doi: 10.1111/1469-0691.12577
Gona, F., Bongiorno, D., Aprile, A., Corazza, E., Pasqua, B., Scuderi, M. G., et al. (2019). Emergence of two novel sequence types (3366 and 3367) NDM-1- and OXA-48-co-producing k. pneumoniae in Italy. Eur. J. Clin. Microbiol. Infect. Dis. 38 (9), 1687–1691. doi: 10.1007/s10096-019-03597-w
Humphries, R. M., Hemarajata, P. (2017). Resistance to ceftazidime-avibactam in Klebsiella pneumoniae due to porin mutations and the increased expression of KPC-3. Antimicrob. Agents Chemother. 61 (6), e00537–e00517. doi: 10.1128/AAC.00537-17
Krueger, F. (2022) Trim galore. Available at: https://github.com/FelixKrueger/TrimGalore (Accessed 12 July 2022).
Lam, M. M. C., Wick, R. R., Watts, S. C., Cerdeira, L. T., Wyres, K. L., Holt, K. E. (2021). A genomic surveillance framework and genotyping tool for Klebsiella pneumoniae and its related species complex. Nat. Commun. 12 (1), 4188. doi: 10.1038/s41467-021-24448-3
Li, H. (2011). A statistical framework for SNP calling, mutation discovery, association mapping and population genetical parameter estimation from sequencing data. Bioinformatics 27 (21), 2987–2993. doi: 10.1093/bioinformatics/btr509
Martin, M. (2011). Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet.journal 17 (1), 10–12. doi: 10.14806/ej.17.1.200
Paczosa, M. K., Mecsas, J. (2016). Klebsiella pneumoniae: going on the offense with a strong defense. Microbiol. Mol. Biol. Rev. 80 (3), 629–661. doi: 10.1128/MMBR.00078-15
Patel, G., Bonomo, R. A. (2013). «Stormy waters ahead»: global emergence of carbapenemases. Front. Microbiol. 4, 48. doi: 10.3389/fmicb.2013.00048
Paterson, D. L., Bonomo, R. A. (2005). Extended-spectrum beta-lactamases: a clinical update. Clin. Microbiol. Rev. 18 (4), 657–686. doi: 10.1128/CMR.18.4.657-686.2005
Seemann, T. (2014). Prokka: rapid prokaryotic genome annotation. Bioinformatics 30 (14), 2068–2069. doi: 10.1093/bioinformatics/btu153
Shields, R. K., Chen, L., Cheng, S., Chavda, K. D., Press, E. G., Snyder, A., et al. (2017). Emergence of ceftazidime-avibactam resistance due to plasmid-borne blaKPC-3 mutations during treatment of carbapenem-resistant Klebsiella pneumoniae infections. Antimicrob. Agents Chemother. 61 (3), e02097–e02016. doi: 10.1128/AAC.02097-16
Shirley, M. (2018). Ceftazidime-avibactam: A review in the treatment of serious gram-negative bacterial infections. Drugs 78 (6), 675–692. doi: 10.1007/s40265-018-0902-x
Software Guide provided by Illumina. Local run manager generate FASTQ analysis module workflow guide (1000000003344), (2018). Vol. 13.
Venditti, C., Butera, O., Meledandri, M., Balice, M. P., Cocciolillo, G. C., Fontana, C., et al. (2021). Molecular analysis of clinical isolates of ceftazidime-avibactam-resistant klebsiella pneumoniae. Clin. Microbiol. Infect. 27 (7), 1040.e1–1040.e6. doi: 10.1016/j.cmi.2021.03.001
Wang, G., Zhao, G., Chao, X., Xie, L., Wang, H. (2020). The characteristic of virulence, biofilm and antibiotic resistance of klebsiella pneumoniae. Int. J. Environ. Res. Public Health 17 (17), E6278. doi: 10.3390/ijerph17176278
Wick, R. R., Judd, L. M., Gorrie, C. L., Holt, K. E. (2017). Unicycler: Resolving bacterial genome assemblies from short and long sequencing reads. PloS Comput. Biol. 13 (6), e1005595. doi: 10.1371/journal.pcbi.1005595
Wong, J. L. C., Romano, M., Kerry, L. E., Kwong, H. S., Low, W. W., Brett, S. J., et al. (2019). OmpK36-mediated carbapenem resistance attenuates ST258 klebsiella pneumoniae in vivo. Nat. Commun. 10, 3957. doi: 10.1038/s41467-019-11756-y
Keywords: next generation sequencing, KPC, OmpK proteins, resistome, virulome
Citation: Bongiorno D, Bivona DA, Cicino C, Trecarichi EM, Russo A, Marascio N, Mezzatesta ML, Musso N, Privitera GF, Quirino A, Scarlata GGM, Matera G, Torti C and Stefani S (2023) Omic insights into various ceftazidime-avibactam-resistant Klebsiella pneumoniae isolates from two southern Italian regions. Front. Cell. Infect. Microbiol. 12:1010979. doi: 10.3389/fcimb.2022.1010979
Received: 03 August 2022; Accepted: 12 September 2022;
Published: 05 January 2023.
Edited by:
Yichyi Lai, Chung Shan Medical University, TaiwanReviewed by:
Punyawee Dulyayangkul, Chulabhorn Research Institute, ThailandFupin Hu, Huashan Hospital, Fudan University, China
Copyright © 2023 Bongiorno, Bivona, Cicino, Trecarichi, Russo, Marascio, Mezzatesta, Musso, Privitera, Quirino, Scarlata, Matera, Torti and Stefani. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dafne Bongiorno, dafne.bongiorno@unict.it
 Dafne Bongiorno
Dafne Bongiorno Dalida A. Bivona
Dalida A. Bivona Claudia Cicino
Claudia Cicino Enrico M. Trecarichi
Enrico M. Trecarichi Alessandro Russo
Alessandro Russo Nadia Marascio
Nadia Marascio Maria Lina Mezzatesta
Maria Lina Mezzatesta Nicolò Musso
Nicolò Musso Grete F. Privitera1
Grete F. Privitera1  Giuseppe G. M. Scarlata
Giuseppe G. M. Scarlata Giovanni Matera
Giovanni Matera Carlo Torti
Carlo Torti