Abstract
Background
Host defence peptides (HDPs), including human β-defensins (hBDs) and psoriasin/S100A7, exert antimicrobial and immunoregulatory functions of the innate defense system. In addition to these functions, the search for cancer biomarkers has identified HDPs as playing a potential role in both tumor suppression and oncogenesis. Although HDPs are highly expressed in salivary glands, their role as molecules for potential diagnostic and therapeutic approaches has not yet been analyzed.
Objective
The aim of the present study was to investigate whether expression levels of putative pro- or antioncogenic hBDs, including hBD-1, -2, -3, and psoriasin/S 100A7, are altered in salivary gland tumor tissue as potential targets for molecular-based therapeutic approaches.
Methods
We analyzed the expression levels of hBD-1, -2, -3, and psoriasin/S100A7 by quantitative real-time polymerase chain reaction (qrt-PCR) and immunohistochemistry in a case control study by comparing salivary gland tumor samples relative to healthy control specimens from 58 patients. Expression level analysis of hBD-1, -2, -3, and psoriasin/S 100A7 by qrt-PCR was normalized to the endogenous 18S rRNA expression levels.
Results
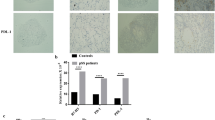
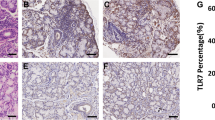
The results demonstrate the significant downregulation of hBD-1 (p<0.001), hBD-2 (p = 0.003), hBD-3 (p = 0.002), and psoriasin/S 100A7 (p = 0.003) mRNA in human salivary gland tumors compared with healthy control specimens. Protein expression levels of hBD-1, -2, -3, and psoriasin/S 100A7 in salivary gland tumor tissue were strongly reduced compared with healthy control specimens.
Conclusion
The data indicates a putative role of the innate defense system in salivary gland tumor formation. The identification of immunoregulatory molecules as diagnostic biomarkers or therapeutic targets could provide new approaches for molecular-based diagnostic and therapeutic support to treat salivary gland tumors as well as other malignancies. We suggest that HDPs should be taken into consideration for use in molecular-based therapeutic approaches.




Similar content being viewed by others
References
Diamond G, Zasloff M, Eck H, et al. Tracheal antimicrobial peptide, a cysteine-rich peptide from mammalian tracheal mucosa: peptide isolation and cloning of a cDNA. Proc Natl Acad Sci U S A 1991 May 1; 88 (9): 3952–6
Sun CQ, Arnold R, Fernandez-Golarz C, et al. Human beta-defensin-1, a potential chromosome 8p tumor suppressor: control of transcription and induction of apoptosis in renal cell carcinoma. Cancer Res 2006 Sep 1; 66 (17): 8542–9
Donald CD, Sun CQ, Lim SD, et al. Cancer-specific loss of beta-defensin 1 in renal and prostatic carcinomas. Lab Invest 2003 Apr; 83 (4): 501–5
Pantelis A, Wenghoefer M, Haas S, et al. Down regulation and nuclear localization of human beta-defensin-1 in pleomorphic adenomas of salivary glands. Oral Oncol 2009 Jun; 45 (6): 526–30
Stoeckelhuber M, Matthias C, Andratschke M, et al. Human ceruminous gland: ultrastructure and histochemical analysis of antimicrobial and cytoskeletal components. Anat Rec A Discov Mol Cell Evol Biol 2006 Aug; 288 (8): 877–84
Yamaguchi Y, Nagase T, Makita R, et al. Identification of multiple novel epididymis-specific beta-defensin isoforms in humans and mice. J Immunol 2002 Sep 1; 169 (5): 2516–23
Beckloff N, Diamond G. Computational analysis suggests beta-defensins are processed to mature peptides by signal peptidase. Protein Pept Lett 2008; 15 (5): 536–40
Droin N, Hendra JB, Ducoroy P, et al. Human defensins as cancer biomarkers and antitumour molecules. J Proteomics 2009 Aug 20; 72 (6): 918–27
Zhu BD, Feng Y, Huang N, et al. Mycobacterium bovis bacille Calmette-Guerin (BCG) enhances human beta-defensin-1 gene transcription in human pulmonary gland epithelial cells. Acta Pharmacol Sin 2003 Sep; 24 (9): 907–12
Abiko Y, Nishimura M, Kaku T. Defensins in saliva and the salivary glands. Med Electron Microsc 2003 Dec; 36 (4): 247–52
Abiko Y, Saitoh M. Salivary defensins and their importance in oral health and disease. Curr Pharm Des 2007; 13 (30): 3065–72
Sahasrabudhe KS, Kimball JR, Morton TH, et al. Expression of the antimicrobial peptide, human beta-defensin 1, in duct cells of minor salivary glands and detection in saliva. J Dent Res 2000 Sep; 79 (9): 1669–74
Froy O. Regulation of mammalian defensin expression by Toll-like receptordependent and independent signalling pathways. Cell Microbiol 2005 Oct; 7 (10): 1387–97
Winter J, Pantelis A, Reich R, et al. Human β-defensin-1, -2, and -3 exhibit opposite effects on oral squamous cell carcinoma cell proliferation. Cancer Invest 2011 Mar; 29 (3): 196–201
Boniotto M, Hazbon MH, Jordan WJ, et al. Novel hairpin-shaped primer assay to study the association of the −44 single-nucleotide polymorphism of the DEFB1 gene with early-onset periodontal disease. Clin Diagn Lab Immunol 2004 Jul; 11 (4): 766–9
Vatta S, Boniotto M, Bevilacqua E, et al. Human beta defensin 1 gene: six new variants. Hum Mutat 2000 Jun; 15 (6): 582–3
Schnapp D, Reid CJ, Harris A. Localization of expression of human beta defensin-1 in the pancreas and kidney. J Pathol 1998 Sep; 186 (1): 99–103
Bullard RS, Gibson W, Bose SK, et al. Functional analysis of the host defense peptide human beta defensin-1: new insight into its potential role in cancer. Mol Immunol 2008 Feb; 45 (3): 839–48
Shestakova T, Zhuravel E, Bolgova L, et al. Immunohistochemical analysis of beta-defensin-2 expression in human lung tumors. Exp Oncol 2010 Dec; 32 (4): 273–6
Kesting MR, Loeffelbein DJ, Hasler RJ, et al. Expression profile of human beta-defensin 3 in oral squamous cell carcinoma. Cancer Invest 2009 Jun; 27 (5): 575–81
Nishimura M, Abiko Y, Kusano K, et al. Localization of human beta-defensin 3 mRNA in normal oral epithelium, leukoplakia, and lichen planus: an in situ hybridization study. Med Electron Microsc 2003 Jun; 36 (2): 94–7
Mburu YK, Abe K, Ferris LK, et al. Human ta-defensin 3 promotes NF-kappaB-mediated CCR7 expression and anti-apoptotic signals in squamous cell carcinoma of the head and neck. Carcinogenesis 2010 Feb; 32 (2): 168–74
Madsen P, Rasmussen HH, Leffers H, et al. Molecular cloning, occurrence, and expression of a novel partially secreted protein “psoriasin” that is highly up-regulated in psoriatic skin. J Invest Dermatol 1991 Oct; 97 (4): 701–12
Celis JE, Rasmussen HH, Vorum H, et al. Bladder squamous cell carcinomas express psoriasin and externalize it to the urine. J Urol 1996 Jun; 155 (6): 2105–12
Al-Haddad S, Zhang Z, Leygue E, et al. Psoriasin (S100A7) expression and invasive breast cancer. Am J Pathol 1999 Dec; 155 (6): 2057–66
Moubayed N, Weichenthal M, Harder J, et al. Psoriasin (S100A7) is significantly up-regulated in human epithelial skin tumours. J Cancer Res Clin Oncol 2007 Apr; 133 (4): 253–61
Morgan MR, Jazayeri M, Ramsay AG, et al. Psoriasin (S100A7) associates with integrin beta6 subunit and is required for αvβ6-dependent carcinoma cell invasion. Oncogene 2011 Mar; 30 (12): 1422–35
Tripathi SC, Matta A, Kaur J, et al. Nuclear S100A7 is associated with poor prognosis in head and neck cancer. PLoS One 2010; 5 (8): e1 1939
Kesting MR, Sudhoff H, Hasler RJ, et al. Psoriasin (S100A7) up-regulation in oral squamous cell carcinoma and its relation to clinicopathologic features. Oral Oncol 2009 Aug; 45 (8): 731–6
Winter J, Pantelis A, Allam JP, et al. High α-defensin and S100A7 expression and missing DOC-1 down-regulation characterize irritation fibromas of the oral cavity and may counteract malignant transformation. J Craniofac Surg 2011 Jan; 22 (1): 100–4
Hocwald E, Korkmaz H, Yoo GH, et al. Prognostic factors in major salivary gland cancer. Laryngoscope 2001 Aug; 111 (8): 1434–9
Lima RA, Tavares MR, Dias FL, et al. Clinical prognostic factors in malignant parotid gland tumors. Otolaryngol Head Neck Surg 2005 Nov; 133 (5): 702–8
Cheung MC, Franzmann E, Sola JE, et al. A comprehensive analysis of parotid and salivary gland cancer: worse outcomes for male gender. J Surg Res 2011 Nov; 171 (1): 151–8
Surakanti SG, Agulnik M. Salivary gland malignancies: the role for chemotherapy and molecular targeted agents. Semin Oncol 2008 Jun; 35 (3): 309–19
Foschini MP, Marucci G, Eusebi V. Low-grade mucoepidermoid carcinoma of salivary glands: characteristic immunohistochemical profile and evidence of striated duct differentiation. Virchows Arch 2002 May; 440 (5): 536–42
Barnes L, Everson JW, Reichart P. World Health Organization classification of tumours, pathology and genetics of head and neck tumours. Lyon: JARC Press, 2005
Lundy FT, Orr DF, Gallagher JR, et al. Identification and overexpression of human neutrophil alpha-defensins (human neutrophil peptides 1, 2 and 3) in squamous cell carcinomas of the human tongue. Oral Oncol 2004 Feb; 40 (2): 139–44
Pazgier M, Hoover DM, Yang D, et al. Human β-defensins. Cell Mol Life Sci 2006 Jun; 63 (11): 1294–313
Joly S, Compton LM, Pujol C, et al. Loss of human beta-defensin 1, 2, and 3 expression in oral squamous cell carcinoma. Oral Microbiol Immunol 2009 Oct; 24 (5): 353–60
Roesch-Ely M, Nees M, Karsai S, et al. Proteomic analysis reveals successive aberrations in protein expression from healthy mucosa to invasive head and neck cancer. Oncogene 2007 Jan 4; 26 (1): 54–64
Escher N, Spies-Weisshart B, Kaatz M, et al. Identification of HNP3 as a tumour marker in CD4+ and CD4−lymphocytes of patients with cutaneous T-cell lymphoma. Eur J Cancer 2006 Jan; 42 (2): 249–55
Selsted ME, Ouellette AJ. Mammalian defensins in the antimicrobial immune response. Nat Immunol 2005 Jun; 6 (6): 551–7
Wenghoefer M, Pantelis A, Dommisch H, et al. Nuclear hBD-1 accumulation in malignant salivary gland tumours. BMC Cancer 2008; 8: 290
Wenghoefer M, Pantelis A, Dommisch H, et al. Decreased gene expression of human beta-defensin-1 in the development of squamous cell carcinoma of the oral cavity. Int J Oral Maxillofac Surg 2008 Jul; 37 (7): 660–3
Bose SK, Gibson W, Bullard RS, et al. PAX2 oncogene negatively regulates the expression of the host defense peptide human beta defensin-1 in prostate cancer. Mol Immunol 2009 Mar; 46 (6): 1140–8
Jaehne M, Roeser K, Jaekel T, et al. Clinical and immunohistologic typing of salivary duct carcinoma: a report of 50 cases. Cancer 2005 Jun 15; 103 (12): 2526–33
Zhuravel E, Shestakova T, Efanova O, et al. Human β-defensin-2 controls cell cycle in malignant epithelial cells: in vitro study. Exp Oncol 2011 Sep; 33 (3): 114–20
Sorensen OE, Thapa DR, Rosenthal A, et al. Differential regulation of β-defensin expression in human skin by microbial stimuli. J Immunol 2005 Apr 15; 174 (8): 4870–9
Sorensen OE, Thapa DR, Roupe KM, et al. Injury-induced innate immune response in human skin mediated by transactivation of the epidermal growth factor receptor. J Clin Invest 2006 Jul; 116 (7): 1878–85
Voss E, Wehkamp J, Wehkamp K, et al. NOD2/CARD15 mediates induction of the antimicrobial peptide human beta-defensin-2. J Biol Chem 2006 Jan 27; 281 (4): 2005–11
Kusmartsev S, Gabrilovich DI. Immature myeloid cells and cancer-associated immune suppression. Cancer Immunol Immunother 2002 Aug; 51 (6): 293–8
Wang J, Xi L, Hunt JL, et al. Expression pattern of chemokine receptor 6 (CCR6) and CCR7 in squamous cell carcinoma of the head and neck identifies a novel metastatic phenotype. Cancer Res 2004 Mar 1; 64 (5): 1861–6
Mburu YK, Abe K, Ferris LK, et al. Human β-defensin 3 promotes NF-κBmediated CCR7 expression and anti-apoptotic signals in squamous cell carcinoma of the head and neck. Carcinogenesis 2011 Feb; 32 (2): 168–74
Melle C, Ernst G, Schimmel B, et al. Discovery and identification of α-defensins as low abundant, tumor-derived serum markers in colorectal cancer. Gastroenterology 2005 Jul; 129 (1): 66–73
Albrethsen J, Moller CH, Olsen J, et al. Human neutrophil peptides 1, 2 and 3 are biochemical markers for metastatic colorectal cancer. Eur J Cancer 2006 Nov; 42 (17): 3057–64
Ghavami S, Asoodeh A, Klonisch T, et al. Brevinin-2R (1) semi-selectively kills cancer cells by a distinct mechanism, which involves the lysosomalmitochondrial death pathway. J Cell Mol Med 2008 Jun; 12 (3): 1005–22
Mader JS, Hoskin DW. Cationic antimicrobial peptides as novel cytotoxic agents for cancer treatment. Expert Opin Investig Drugs 2006 Aug; 15 (8): 933–46
Barker E, Reisfeld RA. A mechanism for neutrophil-mediated lysis of human neuroblastoma cells. Cancer Res 1993 Jan 15; 53 (2): 362–7
McKeown ST, Lundy FT, Nelson J, et al. The cytotoxic effects of human neutrophil peptide-1 (HNP1) and lactoferrin on oral squamous cell carcinoma (OSCC) in vitro. Oral Oncol 2006 Aug; 42 (7): 685–90
Hubert P, Herman L, Maillard C, et al. Defensins induce the recruitment of dendritic cells in cervical human papillomavirus-associated (pre)neoplastic lesions formed in vitro and transplanted in vivo. FASEB J 2007 Sep; 21 (11): 2765–75
Biragyn A. Defensins: non-antibiotic use for vaccine development. Curr Protein Pept Sci 2005 Feb; 6 (1): 53–60
Tani K, Murphy WJ, Chertov O, et al. Defensins act as potent adjuvants that promote cellular and humoral immune responses in mice to a lymphoma idiotype and carrier antigens. Int Immunol 2000 May; 12 (5): 691–700
Biragyn A, Ruffini PA, Leifer CA, et al. Toll-like receptor 4-dependent activation of dendritic cells by β-defensin 2. Science 2002 Nov 1; 298 (5595): 1025–9
Biragyn A, Coscia M, Nagashima K, et al. Murine b-defensin 2 promotes TLR-4/MyD88-mediated and NF-κB-dependent atypical death of APCs via activation of TNFR2. J Leukoc Biol 2008 Apr; 83 (4): 998–1008
Acknowledgments
1) No funding was provided for conduct of the study and/or preparation of the paper.
2) No funding organization or sponsor was involved in any of the following: design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript.
3) There is no conflict of interest for any of the authors.
4) All persons who made substantial contributions to the work meet the criteria for authorship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kesting, M.R., Stoeckelhuber, M., Kuppek, A. et al. Human β-Defensins and Psoriasin/S100A7 Expression in Salivary Glands. BioDrugs 26, 33–42 (2012). https://doi.org/10.2165/11597570-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11597570-000000000-00000