Abstract
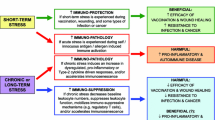
Interventions that reduce the magnitude of psychobiological responses are justified, at least in part, by the notion that exaggerated responses to stress can have detrimental effects on health. The biological processes underlying the association between stress and coronary heart disease (CHD) are thought to involve haemodynamic, neuroendocrine, inflammatory and haemostatic pathways. One of the many recognised benefits of exercise is in buffering physiological responses to psychosocial stressors, which is thought to be partly mediated by sympatho-inhibitory mechanisms, although other potentially important psychobiological processes such as inflammatory, neuroendocrine and haemostatic pathways have gained little attention. Thus, the present review focuses on the role of exercise in buffering psychobiological processes, particularly in relation to pathways that are directly relevant for reducing CHD risk. There are inconsistencies in the literature regarding the effects of exercise on cardiovascular responses to stressors, which may in part be accounted for by differences in experimental design, characteristics of participants, inadequate assessment of physical fitness, and the confounding effects of acute exercise. However, new emerging evidence suggests that exercise promotes an anti-inflammatory environment and increases tissue sensitivity to glucocorticoids, which may have implications for the effects of exercise on stress-induced inflammatory pathways. Future work should focus on the efficacy of exercise for promoting anti-inflammatory pathways in relation to psychosocial stress.


Similar content being viewed by others
References
Rozanski A, Blumenthal JA, Davidson KW, et al. The epidemiology, pathophysiology, and management of psychosocial risk factors in cardiac practice: the emerging field of behavioral cardiology. J Am Coll Cardiol 2005; 45: 637–51
Hemingway H, Marmot M. Evidence based cardiology: psychosocial factors in the aetiology and prognosis of coronary heart disease: systematic review of prospective cohort studies. BMJ 1999; 318: 1460–7
Burg MM, Barefoot J, Berkman L, et al. Low perceived social support and post-myocardial infarction prognosis in the enhancing recovery in coronary heart disease clinical trial: the effects of treatment. Psychosom Med 2005; 67: 879–88
Blumenthal JA, Sherwood A, Babyak MA, et al. Effects of exercise and stress management training on markers of cardiovascular risk in patients with ischemic heart disease: a randomized controlled trial. JAMA 2005; 293: 1626–34
Paffenbarger RS, Hyde RT, Wing AL, et al. The association of changes in physical-activity level and other lifestyle characteristics with mortality among men. N Engl J Med 1993; 328: 538–45
Blair SN, Kampert JB, Kohl HW, et al. Influences of cardiorespiratory fitness and other precursors on cardiovascular disease and all-cause mortality in men and women. JAMA 1996; 276: 205–10
Ross R. Atherosclerosis: an inflammatory disease. N Engl J Med 1999; 340: 115–26
Brydon L, Magid K, Steptoe A. Platelets, coronary heart disease, and stress. Brain Behav Immun 2005; 20: 113–9. Epub 2005 Sep 23
Steptoe A, Brydon L. Psychoneuroimmunology and coronary heart disease. In: Vedhara K, Irwin MR, editors. Human psychoneuroimmunology. Oxford: Oxford University Press, 2005: 107–35
von Kanel R, Mills PJ, Fainman C, et al. Effects of psychological stress and psychiatric disorders on blood coagulation and fibrinolysis: a biobehavioral pathway to coronary artery disease? Psychosom Med 2001; 63: 531–44
Kaplan JR, Manuck SB, Clarkson TB, et al. Social stress and atherosclerosis in normocholesterolemic monkeys. Science 1983; 220: 733–5
Strawn WB, Bondjers G, Kaplan JR, et al. Endothelial dysfunction in response to psychosocial stress in monkeys. Circ Res 1991; 68: 1270–9
Kuper H, Singh-Manoux A, Siegrist J, et al. When reciprocity fails: effort-reward imbalance in relation to coronary heart disease and health functioning within the Whitehall II study. Occup Environ Med 2002; 59: 777–84
Bosma H, Peter R, Siegrist J, et al. Two alternative job stress models and the risk of coronary heart disease. Am J Public Health 1998; 88: 68–74
Kivimaki M, Leino-Arjas P, Luukkonen R, et al. Work stress and risk of cardiovascular mortality: prospective cohort study of industrial employees. BMJ 2002; 325: 857
Gallo LC, Troxel WM, Kuller LH, et al. Marital status, marital quality, and atherosclerotic burden in postmenopausal women. Psychosom Med 2003; 65: 952–62
Schulz R, Beach SR. Caregiving as a risk factor for mortality: the Caregiver Health Effects Study. JAMA 1999; 282: 2215–9
Ford DE, Mead LA, Chang PP, et al. Depression is a risk factor for coronary artery disease in men: the precursors study. Arch Intern Med 1998; 158: 1422–6
Barefoot JC, Schroll M. Symptoms of depression, acute myocardial infarction, and total mortality in a community sample. Circulation 1996; 93: 1976–80
Jennings JR, Kamarck TW, Everson-Rose SA, et al. Exaggerated blood pressure responses during mental stress are prospectively related to enhanced carotid atherosclerosis in middle-aged Finnish men. Circulation 2004; 110: 2198–203
Gianaros PJ, Salomon K, Zhou F, et al. A greater reduction in high-frequency heart rate variability to a psychological stressor is associated with subclinical coronary and aortic calcification in postmenopausal women. Psychosom Med 2005; 67: 553–60
Ghiadoni L, Donald AE, Cropley M, et al. Mental stress induces transient endothelial dysfunction in humans. Circulation 2000; 102: 2473–8
Steptoe A, Willemsen G, Owen N, et al. Acute mental stress elicits delayed increases in circulating inflammatory cytokine levels. Clin Sci 2001; 101: 185–92
Brydon L, Edwards S, Jia H, et al. Psychological stress activates interleukin-1beta gene expression in human mononuclear cells. Brain Behav Immun 2005; 19: 540–6
Steptoe A, Magid K, Edwards S, et al. The influence of psychological stress and socioeconomic status on platelet activation in men. Atherosclerosis 2003; 168: 57–63
Steptoe A, Kunz-Ebrecht S, Rumley A, et al. Prolonged elevations in haemostatic and rheological responses following psychological stress in low socioeconomic status men and women. Thromb Haemost 2003; 89: 83–90
von Kanel R, Dimsdale JE, Ziegler MG, et al. Effect of acute psychological stress on the hypercoagulable state in subjects (spousal caregivers of patients with Alzheimer’s disease) with coronary or cerebrovascular disease and/or systemic hypertension. Am J Cardiol 2001; 87: 1405–8
Brydon L, Edwards S, Mohamed-Ali V, et al. Socioeconomic status and stress-induced increases in interleukin-6. Brain Behav Immun 2004; 18: 281–90
Hamer M, Williams E, Vuonovirta R, et al.. The effects of effort: reward imbalance on inflammatory and cardiovascular responses to mental stress. Psychosom Med 2006 May-Jun; 68 (3): 408–13
Light KC, Kothandapani RV, Allen MT. Enhanced cardiovascular and catecholamine responses in women with depressive symptoms. Int J Psychophysiol 1998; 28: 157–66
Matthews SC, Nelesen RA, Dimsdale JE. Depressive symptoms are associated with increased systemic vascular resistance to stress. Psychosom Med 2005; 67: 509–13
Miller GE, Rohleder N, Stetler C, et al. Clinical depression and regulation of the inflammatory response during acute stress. Psychosom Med 2005; 67: 679–87
von Kanel R, Dimsdale JE, Adler KA, et al. Effects of depressive symptoms and anxiety on hemostatic responses to acute mental stress and recovery in the elderly. Psychiatry Res 2004; 126: 253–64
Hull EM, Young SH, Ziegler MG. Aerobic fitness affects cardiovascular and catecholamine responses to stressors. Psychophysiology 1984; 21: 353–60
Salmon P. Effects of physical exercise on anxiety, depression, and sensitivity to stress: a unifying theory. Clin Psychol Rev 2001; 21: 33–61
Sothmann MS, Buckworth J, Claytor RP, et al. Exercise training and the cross-stressor adaptation hypothesis. Exerc Sport Sci Rev 1996; 24: 267–87
Jern S, Bergbrant A, Bjorntorp P, et al. Relation of central hemodynamics to obesity and body fat distribution. Hypertension 1992; 19: 520–7
Sung BH, Wilson MF, Izzo JL, et al. Moderately obese, insulinresistant women exhibit abnormal vascular reactivity to stress. Hypertension 1997; 30: 848–53
Davis MC, Twamley EW, Hamilton NA, et al. Body fat distribution and hemodynamic stress responses in premenopausal obese women: a preliminary study. Health Psychol 1999; 18: 625–33
Waldstein SR, Burns HO, Toth MJ, et al. Cardiovascular reactivity and central adiposity in older blacks. Health Psychol 1999; 18: 221–8
Rosmond R, Dallman MF, Bjorntorp P. Stress-related cortisol secretion in men: relationships with abdominal obesity and endocrine, metabolic and hemodynamic abnormalities. J Clin Endocrinol Metab 1998; 83: 1853–9
Ribeiro MM, Silva AG, Santos NS, et al. Diet and exercise training restore blood pressure and vasodilatory responses during physiological maneuvers in obese children. Circulation 2005; 111: 1915–23
Claytor RP. Stress reactivity: hemodynamic adjustments in trained and untrained humans. Med Sci Sports Exerc 1991; 23: 873–81
Sothmann MS, Hart BA, Horn TS. Plasma catecholamine response to acute psychological stress in humans: relation to aerobic fitness and exercise training. Med Sci Sports Exerc 1991; 23: 860–7
De Geus EJC, Van Doornen LJP. The effects of fitness training on the physiological stress response. Work Stress 1993; 7: 141–59
Crews DJ, Landers DM. A meta-analytic review of aerobic fitness and reactivity to psychosocial stressors. Med Sci Sports Exerc 1987; 19: S114–20
Dishman RK, Jackson EM. Cardiorespiratory fitness and laboratory stress: a meta-regression analysis. Psychophysiology 2006; 43: 57–72
Hamer M, Taylor A, Steptoe A. The effect of acute aerobic exercise on stress related blood pressure responses: a systematic review and meta-analysis. Biol Psychol 2006; 71: 183–90
Hamer M, Boutcher Y, Boutcher SH. Cardiovascular and renal responses to mental challenge in highly and moderately active males with a family history of hypertension. J Hum Hypertens 2002; 16: 319–26
King AC, Baumann K, O’Sullivan P, et al. Effects of moderateintensity exercise on physiological, behavioral, and emotional responses to family caregiving: a randomized controlled trial. J Gerontol A Biol Sci Med Sci 2002; 57: M26–36
Sinyor D, Schwartz SG, Peronnet F, et al. Aerobic fitness level and reactivity to psychosocial stress: physiological, biochemical, and subjective measures. Psychosom Med 1983; 45: 205–17
van Doornen LJ, de Geus EJ. Aerobic fitness and the cardiovascular response to stress. Psychophysiology 1989; 26: 17–28
Boutcher SH, Nurhayati Y, McLaren PF. Cardiovascular response of trained and untrained old men to mental challenge. Med Sci Sports Exerc 2001; 33: 659–64
Boutcher SH, Nugent FW, McLaren PF, et al. Heart period variability of trained and untrained men at rest and during mental challenge. Psychophysiology 1998; 35: 16–22
Sherwood A, Light KC, Blumenthal JA. Effects of aerobic exercise training on hemodynamic responses during psychosocial stress in normotensive and borderline hypertensive type A men: a preliminary report. Psychosom Med 1989; 51: 123–36
Brooke ST, Long BC. Efficiency of coping with a real-life stressor: a multimodal comparison of aerobic fitness. Psychophysiology 1987; 24: 173–80
Halliwill JR. Mechanisms and clinical implications of post-exercise hypotension in humans. Exerc Sport Sci Rev 2001; 29: 65–70
West SG, Brownley KA, Light KC. Post-exercise vasodilatation reduces diastolic blood pressure responses to stress. Ann Behav Med 1998; 20: 77–83
Brownley KA, Hinderlitter AL, West SG, et al. Sympathoadrenergic mechanisms in reduced hemodynamic stress responses after exercise. Med Sci Sport Exerc 2003; 35: 978–86
Walther C, Gielen S, Hambrecht R. The effect of exercise training on endothelial function in cardiovascular disease in humans. Exerc Sport Sci Rev 2004; 32: 129–34
Blumenthal JA, Jiang W, Babyak MA, et al. Stress management and exercise training in cardiac patients with myocardial ischemia: effects on prognosis and evaluation of mechanisms. Arch Intern Med 1997; 157: 2213–23
Blumenthal JA, Babyak M, Wei J, et al. Usefulness of ps ychosocial treatment of mental stress-induced myocardial ischemia in men. Am J Cardiol 2002; 89: 164–8
Barnes PJ, Karin M. Nuclear factor-kappaB: a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med 1997; 336: 1066–71
Bierhaus A, Wolf J, Andrassy M, et al. A mechanism converting psychosocial stress into mononuclear cell activation. Proc Natl Acad Sci U S A 2003; 100: 1920–5
Radak Z, Chung HY, Naito H, et al. Age-associated increase in oxidative stress and nuclear factor kappaB activation are attenuated in rat liver by regular exercise. FASEB J 2004; 18: 749–50
Steensberg A, Fischer CP, Keller C, et al. IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am J Physiol Endocrinol Metab 2003; 285: E433–7
Smith JK, Dykes R, Douglas JE, et al. Long-term exercise and atherogenic activity of blood mononuclear cells in persons at risk of developing ischemic heart disease. JAMA 1999; 281: 1722–7
Stewart LK, Flynn MG, Campbell WW, et al. Influence of exercise training and age on CD14+ cell-surface expression of toll-like receptor 2 and 4. Brain Behav Immun 2005; 19: 389–97
Kasapis C, Thompson PD. The effects of physical activity on serum C-reactive protein and inflammatory markers: a systematic review. J Am Coll Cardiol 2005; 45: 1563–9
Kiang JG, Tsokos GC. Heat shock protein 70 kDa: molecular biology, biochemistry, and physiology. Pharmacol Ther 1998; 80: 183–201
Campisi J, Leem TH, Greenwood BN, et al. Habitual physical activity facilitates stress-induced HSP72 induction in brain, peripheral, and immune tissues. Am J Physiol Regul Integr Comp Physiol 2003; 284: R520–30
Rohleder N, Schommer NC, Hellhammer DH, et al. Sex differences in glucocorticoid sensitivity of proinflammatory cytokine production after psychosocial stress. Psychosom Med 2001; 63: 966–72
Rohleder N, Joksimovic L, Wolf JM, et al. Hypocortisolism and increased glucocorticoid sensitivity of pro-Inflammatory cytokine production in Bosnian war refugees with posttraumatic stress disorder. Biol Psychiatry 2004; 55: 745–51
Duclos M, Gouarne C, Bonnemaison D. Acute and chronic effects of exercise on tissue sensitivity to glucocorticoids. J Appl Physiol 2003; 94: 869–75
Woiciechowsky C, Schoning B, Daberkow N, et al. Brain-IL-1beta induces local inflammation but systemic anti-inflammatory response through stimulation of both hypothalamicpituitary-adrenal axis and sympathetic nervous system. Brain Res 1999; 816: 563–71
Mastorakos G, Chrousos GP, Weber JS. Recombinant interleukin-6 activates the hypothalamic-pituitary-adrenal axis in humans. J Clin Endocrinol Metab 1993; 77: 1690–4
Droste SK, Gesing A, Ulbricht S, et al. Effects of long-term voluntary exercise on the mouse hypothalamic-pituitary-adrenocortical axis. Endocrinology 2003; 144: 3012–23
Traustadottir T, Bosch PR, Matt KS. The HPA axis response to stress in women: effects of aging and fitness. Psychoneuroendocrinology 2005; 30: 392–402
Traustadottir T, Bosch PR, Cantu T, et al. Hypothalamic-pituitary-adrenal axis response and recovery from high-intensity exercise in women: effects of aging and fitness. J Clin Endocrinol Metab 2004; 89: 3248–54
Anisman H, Ravindran AV, Griffiths J, et al. Endocrine and cytokine correlates of major depression and dysthymia with typical or atypical features. Mol Psychiatry 1999; 4: 182–8
Suarez EC, Krishnan RR, Lewis JG. The relation of severity of depressive symptoms to monocyte-associated proinflammatory cytokines and chemokines in apparently healthy men. Psychosom Med 2003; 65: 362–8
Suarez EC, Lewis JG, Krishnan RR, et al. Enhanced expression of cytokines and chemokines by blood monocytes to in vitro lipopolysaccharide stimulation are associated with hostility and severity of depressive symptoms in healthy women. Psychoneuroendocrinology 2004; 29: 1119–28
De Kloet ER. Hormones and the stressed brain. Ann N Y Acad Sci 2004; 1018: 1–15
Lawlor DA, Hopker SW. The effectiveness of exercise as an intervention in the management of depression: systematic review and meta-regression analysis of randomised controlled trials. BMJ 2001; 322: 763–7
Tracey KJ. The inflammatory reflex. Nature 2002; 420: 853–9
El-Sayed MS, Ali N, El-Sayed Ali Z. Aggregation and activation of blood platelets in exercise and training. Sports Med 2005; 35: 11–22
Wang JS, Li YS, Chen JC, et al. Effects of exercise training and deconditioning on platelet aggregation induced by alternating shear stress in men. Arterioscler Thromb Vasc Biol 2005; 25: 454–60
Whincup PH, Danesh J, Walker M, et al. von Willebrand factor and coronary heart disease: prospective study and meta-analysis. Eur Heart J 2002; 23: 1764–70
Hong S, Farag NH, Nelesen RA, et al. Effects of regular exercise on lymphocyte subsets and CD62L after psychological vs. physical stress. J Psychosom Res 2004; 56: 363–7
Critchley HD, Corfield DR, Chandler MP, et al. Cerebral correlates of autonomic cardiovascular arousal: a functional neuroimaging investigation in humans. J Physiol 2000; 523: 259–70
Matthews SC, Paulus MP, Simmons AN, et al. Functional subdivisions within anterior cingulate cortex and their relationship to autonomic nervous system function. Neuroimage 2004; 22: 1151–6
Gianaros PJ, May JC, Siegle GJ, et al. Is there a functional neural correlate of individual differences in cardiovascular reactivity? Psychosom Med 2005; 67: 31–9
Williamson JW, McColl R, Mathews D. Changes in regional blood flow distribution during post-exercise hypotension in humans. J Appl Physiol 2004; 96: 719–24
Dishman RK. Brain monoamines, exercise, and behavioral stress: animal models. Med Sci Sports Exerc 1997; 29: 63–74
Steptoe A, Wardle J, Marmot M. Positive affect and health-related neuroendocrine, cardiovascular, and inflammatory processes. Proc Natl Acad Sci U S A 2005; 102: 6508–12
Biddle SJH. Emotion, mood and physical activity. In: Biddle SJH, Fox KR, Boutcher SH, editors. Physical activity and psychological well-being. London: Routledge, 2000: 63–88
Bonanno GA, Papa A, Lalande K, et al. The importance of being flexible: the ability to both enhance and suppress emotional expression predicts long-term adjustment. Psychol Sci 2004; 15: 482–7
McCubbin JA, Surwit RS, Williams RB. Endogenous opiate peptides, stress reactivity, and risk for hypertension. Hypertension 1995; 7: 808–11
McCubbin JA, Cheung R, Montgomery TB, et al. Aerobic fitness and opioidergic inhibition of cardiovascular stress reactivity. Psychophysiology 1992; 29: 687–97
Acknowledgements
Dr Hamer is supported by a grant from the British Heart Foundation (UK). The author has no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hamer, M. Exercise and Psychobiological Processes. Sports Med 36, 829–838 (2006). https://doi.org/10.2165/00007256-200636100-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00007256-200636100-00002