Abstract
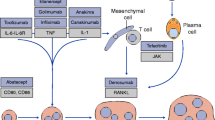
Rheumatoid arthritis (RA), characterized by progressive joint destruction, deformity, disability and impaired quality of life (QOL), is a prevalent autoimmune disease affecting 1% of adults in the US. The goal of therapy in patients with RA is to arrest the disease and to achieve remission by preventing or controlling joint damage, preventing loss of function and providing pain relief, thereby improving QOL. Non-biological disease-modifying antirheumatic drugs (DMARDs) have been the mainstay of early intervention in RA, of which methotrexate has been used most frequently. However, in the long term, patients treated with non-biological DMARDs (including methotrexate) may experience joint deterioration and subclinical inflammation even after clinical remission, emphasizing the need for alternative therapies. Several biological therapies, such as anti-tumour necrosis factor (TNF)-α agents, have been developed in the last decade and may be used either as monotherapy or in combination with non-biological DMARDs. Although anti-TNFα therapy is generally associated with an improvement in symptoms of RA, some patients may experience inadequate response to or may not tolerate these agents. The new biological agent abatacept, a recombinant protein consisting of the extracellular region of the human cytotoxic T-lymphocyte-associated antigen (CTLA)-4 receptor fused to the constant fragment (Fc) region of IgG1, binds to the CD80/CD86 molecules on antigen-presenting cells and modulates T-cell activation. Clinical trials have shown that abatacept is effective in reducing disease activity, structural joint damage and improving QOL in patients with RA who had inadequate response to prior methotrexate or anti-TNFα therapy. Pooled analysis of these trials showed that abatacept was also generally well tolerated in these patients. Thus, abatacept therapy may be an option for the treatment of RA in patients who have had an inadequate response to prior DMARD therapy.







Similar content being viewed by others
References
American College of Rheumatology Subcommittee on Rheumatoid Arthritis Guidelines. Guidelines for the management of rheumatoid arthritis: 2002 Update [see comment in Arthritis Rheum 2002 Nov; 46:3102-3; author reply 3103-4]. Arthritis Rheum 2002 Feb; 46: 328–46
Luqmani R, Hennell S, Estrach C, et al. British Society for Rheumatology and British Health Professionals in Rheumatology guideline for the management of rheumatoid arthritis (the first two years). Rheumatology (Oxford) 2006 Sep; 45: 1167–9
Scott DL. Pursuit of optimal outcomes in rheumatoid arthritis. Pharmacoeconomics 2004; 22: 13–26
Gordon P, West J, Jones H, et al. 10 year prospective followup of patients with rheumatoid arthritis 1986–96. J Rheumatol 2001; 28: 2409–15
Brown AK, Quinn MA, Karim Z, et al. Presence of significant synovitis in rheumatoid arthritis patients with disease-modifying antirheumatic drug-induced clinical remission: evidence from an imaging study may explain structural progression. Arthritis Rheum 2006; 54: 3761–73
Bristol-Myers Squibb Company Orencia® (abatacept): US prescribing information. New York: 2008
Bristol-Myers Squibb Company Orencia® (abatacept): summary of product characteristics. New York: 2008
Genentech Inc Rituxan® (rituximab): US prescribing information. South San Francisco (CA): 2008
Immunex Corporation Enbrel® (etanercept): US prescribing information. Seattle (WA): 2007
Wyeth Pharmaceuticals Enbrel® (etanercept): summary of product characteristics. Madison (WI): 2005
Abbott Laboratories Humira® (adalimumab): US prescribing information. Abbott Park (IL): 2008
Abbott Laboratories Humira® (adalimumab): summary of product characteristics. Abbott Park (IL): 2008
Amgen Ltd. Kineret® (anakinra): US prescribing information. Thousand Oaks (CA): 2001
Amgen Ltd. Kineret® (anakinra): summary of product characteristics. Thousand Oaks (CA): 2008
Roche Pharmaceuticals MabThera® (rituximab): summary of product characteristics. Basel: 2008
Centocor Inc. Remicade® (infliximab): US prescribing information. Horsham (PA): 2007
Centocor BV, Remicade® (infliximab): summary of product characteristics. Horsham (PA): 2008
Lipsky PE, Van der Heijde D, St Clair EW, et al. Infliximab and methotrexate in the treatment of rheumatoid arthritis. N Engl J Med 2000; 343: 1594–602
Cohen SB, Moreland LW, Cush JJ, et al. A multicentre, double blind, randomised, placebo controlled trial of anakinra (Kineret), a recombinant interleukin 1 receptor antagonist, in patients with rheumatoid arthritis treated with background methotrexate. Ann Rheum Dis 2004; 63: 1062–68
Keystone EC, Kavanaugh AF, Sharp JT, et al. Radio-graphic, clinical, and functional outcomes of treatment with adalimumab (a human anti-tumor necrosis factor monoclonal antibody) in patients with active rheumatoid arthritis receiving concomitant methotrexate therapy: a randomized, placebo-controlled, 52-week trial. Arthritis Rheum 2004; 50: 1400–11
Weinblatt ME, Kremer JM, Bankhurst AD, et al. A trial of etanercept, a recombinant tumor necrosis factor receptor:Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N Engl J Med 1999 Jan 28; 340: 253–9
Van Riel PL, Taggart AJ, Sany J, et al. Efficacy and safety of combination etanercept and methotrexate versus etanercept alone in patients with rheumatoid arthritis with an inadequate response to methotrexate: the ADORE study. Ann Rheum Dis 2006 Nov; 65: 1478–83
Klareskog L, van der Heijde D, de Jager JP, et al. Therapeutic effect of the combination of etanercept and methotrexate compared with each treatment alone in patients with rheumatoid arthritis: double-blind randomised controlled trial. Lancet 2004; 363: 675–81
Van der Heijde D, Klareskog L, Rodriguez-Valverde V, et al. Comparison of etanercept and methotrexate, alone and combined, in the treatment of rheumatoid arthritis: two-year clinical and radiographic results from the TEMPO study, a double-blind, randomized trial. Arthritis Rheum 2006 Apr; 54: 1063–74
Finckh A, Simard JF, Duryea J, et al. The effectiveness of anti-tumor necrosis factor therapy in preventing progressive radiographic joint damage in rheumatoid arthritis: a population-based study. Arthritis Rheum 2006; 54: 54–9
Goekoop-Ruiterman YPM, De Vries-Bouwstra JK, Allaart CF, et al. Clinical and radiographic outcomes of four different treatment strategies in patients with early rheumatoid arthritis (the BeSt study): a randomized, controlled trial. Arthritis Rheum 2005; 52: 3381–90
Weinblatt ME, Keystone EC, Furst DE, et al. Adalimumab, a fully human anti-tumor necrosis factor alpha monoclonal antibody, for the treatment of rheumatoid arthritis in patients taking concomitant methotrexate: the ARMADA trial. Arthritis Rheum 2003; 48: 35–45
Keane J, Gershon S, Wise RP, et al. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med 2001; 345: 1098–104
Wallis RS, Broder MS, Wong JY, et al. Granulomatous infectious diseases associated with tumor necrosis factor antagonists. Clin Infect Dis 2004; 38: 1261–5
Gomez-Reino JJ, Carmona L, Valverde VR, et al. Treatment of rheumatoid arthritis with tumor necrosis factor inhibitors may predispose to significant increase in tuberculosis risk: a multicenter active-surveillance report. Arthritis Rheum 2003; 48: 2122–7
Bongartz T, Sutton AJ, Sweeting MJ, et al. Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systematic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA 2006 May 17; 295: 2275–85
Ariza-Ariza R, Navarro-Sarabia F, Hernandez-Cruz B, et al. Dose escalation of the anti-TNF-alpha agents in patients with rheumatoid arthritis: a systematic review. Rheumatology (Oxford) 2007 Mar; 46: 529–32
Ollendorf DA, Massarotti E, Birbara C, et al. Frequency, predictors, and economic impact of upward dose adjustment of infliximab in managed care patients with rheumatoid arthritis. J Manage Care Pharm 2005 Jun; 11: 383–93
Lutt JR, Deodhar A. Rheumatoid arthritis: strategies in the management of patients showing an inadequate response to TNF-alpha antagonists. Drugs 2008; 68: 591–606
Hyrich KL, Lunt M, Watson KD, et al. Outcomes after switching from one anti-tumor necrosis factor alpha agent to a second anti-tumor necrosis factor alpha agent in patients with rheumatoid arthritis: results from a large UK national cohort study [see comment in Nat Clin Pract Rheumatol 2007 Aug; 3430-1]. Arthritis Rheum 2007 Jan; 56: 13–20
Solau-Gervais E, Laxenaire N, Cortet B, et al. Lack of efficacy of a third tumour necrosis factor alpha antagonist after failure of a soluble receptor and a monoclonal antibody. Rheumatology (Oxford) 2006 Sep; 45: 1121–4
Choy EH, Panayi GS. Cytokine pathways and joint inflammation in rheumatoid arthritis. N Engl J Med 2001 Mar 22; 344: 907–16
Van Boxel JA, Paget SA. Predominantly T-cell infiltrate in rheumatoid synovial membranes. N Engl J Med 1975 Sep 11; 293: 517–20
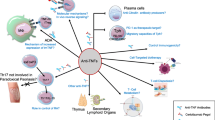
Teng GG, Turkiewicz AM, Moreland LW. Abatacept: A costimulatory inhibitor for treatment of rheumatoid arthritis. Exp Opin Biol Ther 2005; 5: 1245–54
Rozelle AL, Genovese MC. Efficacy results from pivotal clinical trials with abatacept. Clin Exp Rheum 2007 Sep–Oct; 25: S30–4
Reiser H, Stadecker MJ. Costimulatory B7 molecules in the pathogenesis of infectious and autoimmune diseases. N Engl J Med 1996 Oct 31; 335: 1369–77
Greene JL, Leytze GM, Emswiler J, et al. Covalent dimerization of CD28/CTLA-4 and oligomerization of CD80/CD86 regulate T cell costimulatory interactions. J Biol Chem 1996 Oct 25; 271: 26762–71
Brunner MC, Chambers CA, Chan FK, et al. CTLA-4-Mediated inhibition of early events of T cell proliferation. J Immunol 1999 May 15; 162: 5813–20
Finck BK, Linsley PS, Wofsy D. Treatment of murine lupus with CTLA4Ig. Science 1994 Aug 26; 265: 1225–7
Webb LM, Walmsley MJ, Feldmann M. Prevention and amelioration of collagen-induced arthritis by blockade of the CD28 co-stimulatory pathway: requirement for both B7-1 and B7-2. Eur J Immunol 1996 Oct; 26: 2320–8
Davis PM, Nadler SG, Stetsko DK, et al. Abatacept modulates human dendritic cell-stimulated T-cell proliferation and effector function independent of IDO induction. Clin Immunol 2008 Jan; 126: 38–47
Nadler S, Townsend R, Mikesell G, et al. Abatacept (CTLA4Ig; BMS-188667) significantly inhibits T-cell proliferation in vitro at clinically relevant concentrations [abstract no. THU0103]. Ann Rheum Dis 2004; 63: 142–3
Shimoyama Y, Nagafuchi H, Suzuki N, et al. Synovium infiltrating T cells induce excessive synovial cell function through CD28/B7 pathway in patients with rheumatoid arthritis. J Rheumatol 1999 Oct; 26: 2094–01
Moreland LW, Alten R, Van Den Bosch F, et al. Costimulatory blockade in patients with rheumatoid arthritis: a pilot, dose-finding, double-blind, placebo-controlled clinical trial evaluating CTLA-4Ig and LEA29Y eighty-five days after the first infusion. Arthritis Rheum 2002 Jun; 46: 1470–9
Kremer JM, Westhovens R, Leon M, et al. Treatment of rheumatoid arthritis by selective inhibition of T-cell activation with fusion protein CTLA4Ig. N Engl J Med 2003 Nov 13; 349: 1907–15
Kremer JM, Dougados M, Emery P, et al. Treatment of rheumatoid arthritis with the selective costimulation modulator abatacept: twelve-month results of a phase IIb, double-blind, randomized, placebo-controlled trial [see comment; erratum appears in Arthritis Rheum 2005; 52 (10): 3321]. Arthritis Rheum 2005 Aug; 52: 2263–71
Kremer JM, Genant HK, Moreland LW, et al. Effects of abatacept in patients with methotrexate-resistant active rheumatoid arthritis: a randomized trial [see comment; summary for patients in Ann Intern Med 2006; 144 (12): I18]. Ann Intern Med 2006 Jun 20; 144: 865–76
Genant HK, Peterfy CG, Westhovens R, et al. Abatacept inhibits progression of structural damage in rheumatoid arthritis: results from the long-term extension of the AIM trial. Ann Rheum Dis 2008 Aug; 67: 1084–9
Kremer JM, Genant HK, Moreland LW, et al. Results of a two-year followup study of patients with rheumatoid arthritis who received a combination of abatacept and methotrexate. Arthritis Rheum 2008 Apr; 58: 953–63
Genovese MC, Becker JC, Schiff M, et al. Abatacept for rheumatoid arthritis refractory to tumor necrosis factor alpha inhibition [erratum appears in N Engl J Med 2005 Nov 24; 353 (21): 2311]. N Engl J Med 2005 Sep 15; 353: 1114–23
Genovese MC, Schiff M, Luggen M, et al. Efficacy and safety of the selective co-stimulation modulator abatacept following 2 years of treatment in patients with rheumatoid arthritis and an inadequate response to anti-tumour necrosis factor therapy. Ann Rheum Dis 2008 Apr; 67: 547–54
Weinblatt M, Schiff M, Goldman A, et al. Selective costimulation modulation using abatacept in patients with active rheumatoid arthritis while receiving etanercept: a randomised clinical trial. Ann Rheum Dis 2007 Feb; 66: 228–34
Schiff M, Keiserman M, Codding C, et al. Efficacy and safety of abatacept or infliximab versus placebo in AT-TEST: a phase III, multi-centre, randomised, double-blind, placebo-controlled study in patients with rheumatoid arthritis and an inadequate response to methotrexate. Ann Rheum Dis 2008 Aug; 67: 1096–03
Michaud K, Bombardier C, Emery P. Quality of life in patients with rheumatoid arthritis: does abatacept make a difference? Clin Exp Rheum 2007; 25(Suppl. 46): S35–45
Dougados M, Russel A, Li T, et al. Abatacept provides sustained improvements in pain, fatigue and sleep quality through 2 years in the treatment of rheumatoid arthritis patients in the AIM and ATTAIN trials [abstract no. 502]. Arthritis Rheum 2006; 54: S245
Russell AS, Wallenstein GV, Li T, et al. Abatacept improves both the physical and mental health of patients with rheumatoid arthritis who have inadequate response to methotrexate treatment. Ann Rheum Dis 2007 Feb; 66: 189–94
Dougados M, Russel A, Li T, et al. Abatacept reduces fatigue and pain severity and sleep problems through 2 years in rheumatoid arthritis patients in the AIM and ATTAIN trials [abstract no. SAT0005]. Ann Rheum Dis 2007; 66: 429
Weinblatt M, Combe B, Covucci A, et al. Safety of the selective costimulation modulator abatacept in rheumatoid arthritis patients receiving background biologic and non-biologic disease-modifying antirheumatic drugs: a one-year randomized, placebo-controlled study. Arthritis Rheum 2006 Sep; 54: 2807–16
Sibilia J, Westhovens R. Safety of T-cell co-stimulation modulation with abatacept in patients with rheumatoid arthritis. Clin Exp Rheum 2007; 25: S46–56
St Clair EW, van der Heijde DM, Smolen JS, et al. Combination of infliximab and methotrexate therapy for early rheumatoid arthritis: a randomized, controlled trial. Arthritis Rheum 2004 Nov; 50: 3432–43
Moreland LW, Baumgartner SW, Schiff MH, et al. Treatment of rheumatoid arthritis with a recombinant human tumor necrosis factor receptor (p75)-Fc fusion protein [see comments in N Engl J Med 1997 Jul 17; 337: 195-7 and N Engl J Med 1997 Nov 20; 337: 1559-60; author reply 1560-1]. N Engl J Med 1997 Jul 17; 337: 141–7
Bathon JM, Martin RW, Fleischmann RM, et al. A comparison of etanercept and methotrexate in patients with early rheumatoid arthritis [see comment in ACP J Club 2001 Jul–Aug; 135: 2; errata appear in N Engl J Med 2001 Jan 4; 344: 76 and N Engl J Med 2001 Jan 18; 344: 240]. N Engl J Med 2000 Nov 30; 343: 1586–93
Acknowledgements
The authors wish to thank Dr Gabriele Forcina for his helpful scientific support. We thank Sohita Dhillon of Wolters Kluwer Health who provided medical writing services funded by Bristol-Myers Squibb Pharmaceuticals. The authors have no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Laganà, B., Vinciguerra, M. & D’Amelio, R. Modulation of T-Cell Co-Stimulation in Rheumatoid Arthritis. Clin. Drug Investig. 29, 185–202 (2009). https://doi.org/10.2165/00044011-200929030-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200929030-00005