Abstract
Objective: To evaluate the acute bronchodilating effect of cumulative doses of formoterol administered via Turbuhaler™ or salmeterol administered via pressurised metered-dose inhaler (pMDI) in patients with mild acute exacerbations of chronic obstructive pulmonary disease (COPD) to determine if these drugs might be used in this condition.
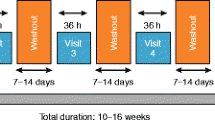
Design: This was a randomised, double-blind, double-dummy, crossover study taking place over two consecutive days.
Patients and setting: 30 patients attending our outpatient clinics with mild acute exacerbations of COPD (Anthonisen exacerbation type I or II), and who were willing to participate, were recruited.
Interventions: A dose-response curve to salmeterol pMDI (25 μg/inhalation) or formoterol Turbuhaler™ (12 μg/inhalation) was constructed using one inhalation, one inhalation, and two inhalations, i.e. a total cumulative dose of metered salmeterol 100μg or formoterol 48μg on two consecutive days. After baseline measurements, dose increments were given at 30-minute intervals with measurements being made 25 minutes after each dose. Oral bronchodilators were not permitted during the study. Short-acting inhaled β2-agonists were permitted soon after each test when required. All patients received treatment with an oral antibiotic (amoxicillin/clavulanic acid or levofloxacin) and an inhaled steroid (budesonide 400μg or fluticasone 250μg twice daily).
Main outcome measures: The highest forced vital capacity (FVC), inspiratory capacity (IC) and forced expiratory volume in one second (FEV1) values obtained from one or other of the curves were kept for analysis. We also measured oxygen saturation by pulse oximetry (SpO2) and heart rate (HR). The maximum FEV1 value during the dose-response curve to formoterol or salmeterol was chosen as the primary outcome variable to compare the two treatments.
Results: Both formoterol and salmeterol induced a large and significant (p < 0.001) dose-dependent increase in FEV1, FVC and IC. There was no significant difference between the FEV1 values (p = 0.081), the FVC values (p = 0.085) and the IC values (p = 0.111) after formoterol 48μg or salmeterol 100μg. Neither HR nor SpO2 changed significantly (p > 0.05).
Conclusions: Our results suggest that cumulative doses of long-acting β2-agonists may improve lung function in patients with mild acute exacerbations of COPD although we cannot advocate their use in the initial therapy of an acute exacerbation because much fundamental information is still lacking.




Similar content being viewed by others
Notes
1Use of tradenames is for product identification only and does not imply endorsement.
References
Ikeda A, Nishimura K, Izumi T. Pharmacological treatment in acute exacerbations of chronic obstructive pulmonary disease. Drugs Aging 1998; 12: 129–37
McCrory DC, Brown C, Gelfand SE, et al. Management of acute exacerbations of COPD. Chest 2001; 119: 1190–209
Karpel JP, Pesin J, Greenberg D, et al. A comparison of the effects of ipratropium bromide and metaproterenol sulfate in acute exacerbations of COPD. Chest 1990; 98: 835–9
Bernasconi M, Brandolese R, Poggi R, et al. Dose-response effects and time course of effects of inhaled fenoterol on respiratory mechanics and arterial oxygen tension in mechanically ventilated patients with chronic airflow obstruction. Intensive Care Med 1990; 16: 108–14
American Thoracic Society. Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 1995; 152(5 Suppl. Pt2): S77–121
Siafakas NM, Vermeire P, Pride NB, et al. Optimal assessment and management of chronic obstructive pulmonary disease (COPD). Eur Respir J 1995; 8: 1398–420
Rodriguez-Roisin R. Toward a consensus definition for COPD exacerbations. Chest 2000; 117 (5 Suppl. Pt 2): 398S–401S
The COPD Guidelines Group of the Standards of Care Committee of the BTS. BTS guidelines for the management of chronic obstructive pulmonary disease. Thorax 1997; 52 Suppl. 5: S1–28
American Thoracic Society. Standardization of spirometry: 1987 update. Am Rev Respir Dis 1987; 136: 1285–98
Kuhl DA, Agiri OA, Mauro LS. β-agonists in the treatment of acute exacerbation of chronic obstructive pulmonary disease. Ann Pharmacother 1994; 28: 1379–88
Pauwels RA, Buist AS, Calverley PM, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary. Am J Respir Crit Care Med 2001; 163: 1256–76
Celik G, Kayacan O, Beder S, et al. Formoterol and salmeterol in partially reversible chronic obstructive pulmonary disease: a crossover, placebo-controlled comparison of onset and duration of action. Respiration 1999; 66: 434–9
Cazzola M, Matera MG, Santangelo G, et al. Salmeterol and formoterol in partially reversible severe chronic obstructive pulmonary disease: a dose-response study. Respir Med 1995; 89: 357–62
Cazzola M, Spina D, Matera MG. The use of bronchodilators in stable chronic obstructive pulmonary disease. Pulm Pharmacol Ther 1997; 10: 129–44
Emerman CL, Cydulka RK. Effect of different albuterol dosing regimens in the treatment of acute exacerbation of chronic obstructive pulmonary disease. Ann Emerg Med 1997; 29: 474–8
Naline E, Zhang Y, Qian Y, et al. Relaxant effects and durations of action of formoterol and salmeterol on the isolated human bronchus. Eur Respir J 1994; 7: 914–20
Lulich KM, Goldie RG, Ryan G, et al. Adverse reactions to β2-agonistbronchodilators. Med Toxicol 1986; 1: 286–99
Cazzola M, Di Perna F, Noschese P, et al. Effects of a pretreatment with conventional doses of formoterol, salmeterol or oxitropium bromide on the dose-response curves to salbutamol in patients suffering from partially reversible COPD. Eur Respir J 1998; 11: 1337–41
Nelson HS, Berkowitz RB, Tinkelman DA, et al. Lack of subsensitivity to albuterol after treatment with salmeterol in patients with asthma. Am J Respir Crit Care Med 1999; 159: 1556–61
Backman R, Hellström PE. Fenoterol and ipratropium bromide for treatment of patients with chronic bronchitis. Curr Ther Res 1985; 38: 135–40
Emerman CL, Lukens TW, Effron D. Physician estimation of FEV1 in acute exacerbation of COPD. Chest 1994; 105: 1709–12
Braunwald E. Heart disease: a textbook of cardiovascular medicine. 4th ed. Philadelphia: WB Saunders, 1992
Maisel AS, Michel MC. Beta-adrenergic receptors in congestive heart failure: present knowledge and future directions. Cardiology 1989; 76: 338–46
Johnson M, Butchers PR, Coleman RA, et al. The pharmacology of salmeterol. Life Sci 1993; 52: 2131–43
Maesen FP, Costongs R, Smeets SJ, et al. Formoterol as dry powder inhalation: a dose finding study in comparison with formoterol metered dose inhaler and placebo. Chest 1992; 101: 1376–81
Ullman A, Svedmyr N. Salmeterol, a new long acting inhaled beta 2 adrenoceptor agonist: comparison with salbutamol in adult asthmatic patients. Thorax 1988; 43: 674–8
Cazzola M, Imperatore F, Salzillo A, et al. Cardiac effects of formoterol and salmeterol in patients suffering from COPD with preexisting cardiac arrhythmias and hypoxemia. Chest 1998; 114: 411–5
Knudson RJ, Constantine HP. An effect of isoproterenol on ventilation-perfusion in asthmatic versus normal subjects. J Appl Physiol 1967; 22: 402–6
Ingram RH Jr, Krumpe PE, Duffel GM, et al. Ventilation-perfusion changes after aerosolized isoproterenol in asthma. Am Rev Respir Dis 1970; 101: 364–70
Wagner PD, Dantzker DR, Iacovoni VE, et al. Ventilation-perfusion inequality in asymptomatic asthma. Am Rev Respir Dis 1978; 118: 511–24
Khoukaz G, Gross NJ. Effects of salmeterol on arterial blood gases in patients with stable chronic obstructive pulmonary disease comparison with albuterol and ipratropium. Am J Respir Crit Care Med 1999; 160: 1028–30
Ries AL. Oximetry-know thy limits. Chest 1987; 91: 316
Acknowledgements
MC and GDA have received financial support for research and attending meetings and have received fees for speaking and consulting by GlaxoSmithKline Italy and AstraZeneca Italy. MGM and PN have received financial support for attending meetings by GlaxoSmithKline Italy and AstraZeneca Italy.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cazzola, M., Matera, M.G., D’Amato, M. et al. Long-Acting β2-Agonists in the Treatment of Acute Exacerbations of COPD. Clin. Drug Investig. 22, 369–376 (2002). https://doi.org/10.2165/00044011-200222060-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00044011-200222060-00004