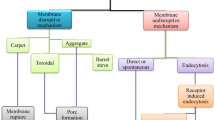
Abstract
Antimicrobial peptides (AMPs) are effector molecules of the innate immune system. A variety of AMPs have been isolated from species of all kingdoms and are classified based on their structure and amino acid motifs. AMPs have a broad antimicrobial spectrum and lyse microbial cells by interaction with biomembranes. Besides their direct antimicrobial function, they have multiple roles as mediators of inflammation with impact on epithelial and inflammatory cells influencing diverse processes such as cell proliferation, immune induction, wound healing, cytokine release, chemotaxis and protease-antiprotease balance. AMPs qualify as prototypes of innovative drugs that may be used as antimicrobials, anti-lipopolysaccharide drugs or modifiers of inflammation. Several strategies have been followed to identify lead candidates for drug development, to modify the peptides’ structures, and to produce sufficient amounts for pre-clinical and clinical studies. This review summarises the current knowledge about the basic and applied biology of AMPs.








Similar content being viewed by others
References
Zasloff M. Antimicrobial peptides of multicellular organisms. Nature 2002; 415(6870): 389–95
Tossi A, Sandri L, Giangaspero A. Amphipathic, alpha-helical antimicrobial peptides. Biopolymers 2000; 55(1): 4–30
Huttner KM, Bevins CL. Antimicrobial peptides as mediators of epithelial host defense. Pediatr Res 1999; 45(6): 785–94
Lehrer RI, Ganz T. Antimicrobial peptides in mammalian and insect host defence. Curr Opin Immunol 1999; 11(1): 23–7
Stein T, Vater J, Kraft V, et al. The multiple carrier model of nonribosomal peptide biosynthesis at modular multienzymatic templates. J Biol Chem 1996; 271(26): 15428–35
Beringer P. The clinical use of colistin in patients with cystic fibrosis. Curr Opin Pulm Med 2001; 7(6): 434–40
Evans ME, Feola DJ, Rapp RP. Polymyxin B sulfate and colistin: old antibiotics for emerging multiresistant gram-negative bacteria. Ann Pharmacother 1999; 33(9): 960–7
Stachelhaus T, Marahiel MA. Modular structure of genes encoding multifunctional peptide synthetases required for nonribosomal peptide synthesis. FEMS Microbiol Lett 1995; 125(1): 3–14
Zhang J, Demain AL. ACV synthetase. Crit Rev Biotechnol 1992; 12(3): 245–60
Bals R, Wattler S, Nehls M, et al. Mouse beta-defensin 3 is a regulated antimicrobial peptide expressed in mucousal organs. Infect Immun 1999; 67: 3542–7
Harder J, Meyer-Hoffert U, Teran LM, et al. Mucoid pseudomonas aeruginosa, TNF-alpha, and IL-1beta, but not IL-6, induce human beta-defensin-2 in respiratory epithelia. Am J Respir Cell Mol Biol 2000; 22(6): 714–21
O’Neil DA, Porter EM, Elewaut D, et al. Expression and regulation of the human beta-defensins hBD-1 and hBD-2 in intestinal epithelium. J Immunol 1999; 163(12): 6718–24
Tsutsumi-Ishii Y, Nagaoka I. NF-kappa B-mediated transcriptional regulation of human beta-defensin-2 gene following lipopolysaccharide stimulation. J Leukoc Biol 2002; 71(1): 154–62
Krisanaprakornkit S, Kimball JR, Dale BA. Regulation of human beta-defensin-2 in gingival epithelial cells: the involvement of mitogen-activated protein kinase pathways, but not the NF-kappaB transcription factor family. J Immunol 2002; 168(1): 316–24
Takahashi A, Wada A, Ogushi K, et al. Production of beta-defensin-2 by human colonie epithelial cells induced by Salmonella enteritidis flagella filament structural protein. FEBS Lett 2001; 508(3): 484–8
Garcia JR, Jaumann F, Schulz S, etal. Identification of a novel, multifunctional beta-defensin (human beta-defensin 3) with specific antimicrobial activity: its interaction with plasma membranes of Xenopus oocytes and the induction of macrophage chemoattraction. Cell Tissue Res 2001; 306(2): 257–64
Hiratsuka T, Nakazato M, Date Y, etal. Identification of human beta-defensin-2 in respiratory tract and plasma and its increase in bacterial pneumonia. Biochem Biophys Res Commun 1998; 249(3): 943–7
Bals R, Weiner DJ, Meegalla RL, et al. Salt-independent abnormality of antimicrobial activity in cystic fibrosis airway surface fluid. Am J Respir Cell Mol Biol 2001; 25(1): 21–5
Dorschner RA, Pestonjamasp VK, Tamakuwala S, et al. Cutaneous injury induces the release of cathelicidin anti-microbial peptides active against group A Streptococcus. J Invest Dermatol 2001; 117(1): 91–7
Birchler T, Seibl R, Buchner K, et al. Human Toll-like receptor 2 mediates induction of the antimicrobial peptide human beta-defensin 2 in response to bacterial lipoprotein. Eur J Immunol 2001; 31(11): 3131–7
Meister M, Lemaitre B, Hoffmann JA. Antimicrobial peptide defense in Drosophila. Bioessays 1997; 19(11): 1019–26
Imler JL, Hoffmann JA. Signaling mechanisms in the antimicrobial host defense of Drosophila. Curr Opin Microbiol 2000; 3(1): 16–22
Tzou P, Ohresser S, Ferrandon D, et al. Tissue-specific inducible expression of antimicrobial peptide genes in Drosophila surface epithelia. Immunity 2000; 13(5): 737–48
Lemaitre B, Reichhart JM, Hoffmann JA. Drosophila host defense: differential induction of antimicrobial peptide genes after infection by various classes of microorganisms. Proc Natl Acad Sci U S A 1997; 94(26): 14614–9
Levashina EA, Ohresser S, Lemaitre B, et al. Two distinct pathways can control expression of the gene encoding the Drosophila antimicrobial peptide metchnikowin. J Mol Biol 1998; 278(3): 515–27
Manfruelli P, Reichhart JM, Steward R, et al. A mosaic analysis in Drosophila fat body cells of the control of antimicrobial peptide genes by the Rel proteins Dorsal and DIF. EMBO J 1999; 18(12): 3380–91
Tauszig S, Jouanguy E, Hoffmann JA, et al. Toll-related receptors and the control of antimicrobial peptide expression in Drosophila. Proc Natl Acad Sci U S A 2000; 97(19): 10520–5
Onfelt Tingvall T, Roos E, Engstrom Y. The imd gene is required for local Cecropin expression in Drosophila barrier epithelia. EMBO Rep 2001; 2(3): 239–43
van’t Hof W, Veerman EC, Heimerhorst EJ, et al. Antimicrobial peptides: properties and applicability. Biol Chem 2001; 382(4): 597–619
Hancock RE. Peptide antibiotics. Lancet 1997; 349(9049): 418–22
Andreu D, Rivas L. Animal antimicrobial peptides: an overview. Biopolymers 1998; 47(6): 415–33
Brogden KA, Ackermann MR, McCray Jr PB, et al. Differences in the concentrations of small, anionic, antimicrobial peptides in bronchoalveolar lavage fluid and in respiratory epithelia of patients with and without cystic fibrosis. Infect Immun 1999; 67(8): 4256–9
Lai R, Liu H, Hui Lee W, et al. An anionic antimicrobial peptide from toad Bombina maxima. Biochem Biophys Res Commun 2002; 295(4): 796–9
Orlov DS, Nguyen T, Lehrer RI. Potassium release, a useful tool for studying antimicrobial peptides. J Microbiol Methods 2002; 49(3): 325–8
Singh P, Jia H, Wiles K, et al. Production of beta-defensins by human airway epithelia. Proc Natl Acad Sci U S A 1998; 95: 14961–6
Steinberg DA, Lehrer RI. Designer assays for antimicrobial peptides. In: Shafer WM, editor. Antimicrobial peptide protocols. Totowa (NJ): Humana Press, 1997: 169–86
Steinberg DA, Hurst MA, Fujii CA, et al. Protegrin-1: a broad-spectrum, rapidly microbicidal peptide with in vivo activity. Antimicrob Agents Chemother 1997; 41(8): 1738–42
Bals R, Lang C, Weiner D, et al. Rhesus monkey (Macaca mulatta) mucosal antimicrobial peptides are close homologues of human molecules. Clin Diagn Lab Immunol 2001; 8(2): 370–5
Li P, Chan HC, He B, et al. An antimicrobial peptide gene found in the male reproductive system of rats. Science 2001; 291(5509): 1783–5
Tossi A, Scocchi M, Zanetti M, et al. An approach combining rapid cDNA amplification and chemical synthesis for the identification of novel, cathelicidin-derived, antimicrobial peptides. Methods Mol Biol 1997; 78: 133–50
Conejo Garcia J-R, Jaumann F, Schulz S, et al. Human b-defensin 3 is an inducible antimicrobial peptide expressed in epithelial and non-epithelial tissues. Cell Tissue Res 2001; 306: 257–64
Garcia JR, Krause A, Schulz S, et al. Human beta-defensin 4: a novel inducible peptide with a specific salt-sensitive spectrum of antimicrobial activity. FASEB J 2001; 15(10): 1819–21
Jia HP, Schutte BC, Schudy A, et al. Discovery of new human beta-defensins using a genomics-based approach. Gene 2001; 263(1–2): 211–8
Schutte BC, Mitros JP, Bartlett JA, et al. Discovery of five conserved beta-defensin gene clusters using a computational search strategy. Proc Natl Acad Sci U S A 2002; 99(4): 2129–33
Lehrer R, Ganz T, Selsted M. Defesins: endogenous antibiotic peptides of animal cells. Cell 1991; 64: 229–30
Garcia-Olmedo F, Molina A, Alamillo JM, et al. Plant defense peptides. Biopolymers 1998; 47(6): 479–91
Ganz T, Selsted ME, Szklarek D, et al. Defensins: natural peptide antibiotics of human neutrophils. J Clin Invest 1985; 76(4): 1427–35
Selsted ME, Harwig SS, Ganz T, et al. Primary structures of three human neutrophil defensins. J Clin Invest 1985; 76(4): 1436–9
Linzmeier R, Ho CH, Hoang BV, et al. A 450-kb contig of defensin genes on human chromosome 8p23. Gene 1999; 233(1–2): 205–11
Wilson CL, Ouellette AJ, Satchell DP, et al. Regulation of intestinal alpha-defensin activation by the metalloproteinase matrilysin in innate host defense. Science 1999; 286(5437): 113–7
Diamond G, Zasloff M, Eck H, et al. Tracheal antimicrobial peptide, a cysteine-rich peptide from mammalian tracheal mucosa: peptide isolation and cloning of a cDNA. Proc Natl Acad Sci USA 1991; 88: 3952–6
Bensch K, Raida M, Magert H-J, et al. hBD-1: a novel β-defensin from human plasma. FEBS Lett 1995; 368: 331–5
McCray Jr P, Bentley L. Human airway epithelia express a beta-defensin. Am J Respir Cell Mol Biol 1997; 16(3): 343–9
Goldman MJ, Anderson GM, Stolzenberg ED, et al. Human beta-defensin-1 is a salt-sensitive antibiotic in lung that is inactivated in cystic fibrosis. Cell 1997; 88(4): 553–60
Valore EV, Park CH, Quayle AJ, et al. Human beta-defensin-1: an antimicrobial peptide of urogenital tissues. J Clin Invest 1998; 101(8): 1633–42
Harder J, Bartels J, Christophers E, et al. A peptide antibiotic from human skin [letter]. Nature 1997; 387: 861
Bals R, Wang X, Wu Z, et al. Human beta-defensin 2 is a salt-sensitive peptide antibiotic expressed in human lung. J Clin Invest 1998; 102: 874–80
Harder J, Bartels J, Christophers E, et al. Isolation and characterization of human beta-defensin-3, a novel human inducible peptide antibiotic. J Biol Chem 2001; 276(8): 5707–13
Tang Y-Q, Yaun J, Osapay G, et al. A cyclic antimicrobial peptide produced in primate leukocytes by the ligation of two truncated alpha-defensins. Science 1999; 286: 498–502
Leonova L, Kokryakov VN, Aleshina G, et al. Circular minidefensins and posttranslational generation of molecular diversity. J Leukoc Biol 2001; 70(3): 461–4
Zanetti M, Gennaro R, Romeo D. Cathelicidins: a novel protein family with a common proregion and a variable C-terminal antimicrobial domain. FEBS Lett 1995; 374: 1–5
Lehrer RI, Ganz T. Cathelicidins: a family of endogenous antimicrobial peptides. Curr Opin Hematol 2002; 9(1): 18–22
Gudmundsson GH, Agerberth B, Odeberg J, et al. The human gene FALL39 and processing of the cathelin precursor to the antibacterial peptide LL-37 in granulocytes. Eur J Biochem 1996; 238(2): 325–32
Cowland J, Johnsen A, Borregaard N. hCAP-18, a cathelin/probactenecin-like protein of human neutrophil specific granules. FEBS Lett 1995; 368(1): 173–6
Larrick J, Hirata M, Balint R, et al. Human CAP18: a novel antimicrobial lipopolysaccharide-binding protein. Infect Immun 1995; 63: 1291–7
Agerberth B, Gunne H, Odeberg J, et al. FALL-39, a putative human peptide antibiotic, is cysteine-free and expressed in bane marrow and testis. Proc Natl Acad Sci USA 1995; 92: 195–9
Bals R, Wang X, Zasloff M, et al. The peptide antibiotic LL-37/hCAP-18 is expressed in epithelia of the human lung where it has broad antimicrobial activity at the airway surface. Proc Natl Acad Sci U S A 1998; 95(16): 9541–6
Agerberth B, Grunewald J, Castanos-Velez E, et al. Antibacterial components in bronchoalveolar lavage fluid from healthy individuals and sarcoidosis patients. Am J Respir Crit Care Med 1999; 160(1): 283–90
Sorensen OE, Follin P, Johnsen AH, et al. Human cathelicidin, hCAP-18, is processed to the antimicrobial peptide LL-37 by extracellular cleavage with proteinase 3. Blood 2001; 97(12): 3951–9
Zhao C, Nguyen T, Boo LM, et al. RL-37, an alpha-helical antimicrobial peptide of the rhesus monkey. Antimicrob Agents Chemother 2001; 45(10): 2695–702
Nizet V, Ohtake T, Lauth X, et al. Innate antimicrobial peptide protects the skin from invasive bacterial infection. Nature 2001; 414(6862): 454–7
Krensky AM. Granulysin: a novel antimicrobial peptide of cytolytic T lymphocytes and natural killer cells. Biochem Pharmacol 2000; 59(4): 317–20
Ochoa MT, Stenger S, Sieling PA, et al. T-cell release of granulysin contributes to host defense in leprosy. Nat Med 2001; 7(2): 174–9
Stenger S, Hanson DA, Teitelbaum R, et al. An antimicrobial activity of cytolytic T cells mediated by granulysin. Science 1998; 282(5386): 121–5
Daw MA, Falkiner FR. Bacteriocins: nature, function and structure. Micron 1996; 27(6): 467–79
Guder A, Wiedemann I, Sahl HG. Posttranslationally modified bacteriocins: the lantibiotics. Biopolymers 2000; 55(1): 62–73
McAuliffe O, Ross RP, Hill C. Lantibiotics: structure, biosynthesis and mode of action. FEMS Microbiol Rev 2001; 25(3): 285–308
Delves-Broughton J, Blackburn P, Evans RJ, et al. Applications of the bacteriocin, nisin. Antonie Van Leeuwenhoek 1996; 69(2): 193–202
Breukink E, Wiedemann I, van Kraaij C, et al. Use of the cell wall precursor lipid II by a pore-forming peptide antibiotic. Science 1999; 286(5448): 2361–4
Wiedemann I, Breukink E, van Kraaij C, et al. Specific binding of nisin to the peptidoglycan precursor lipid II combines pore formation and inhibition of cell wall biosynthesis for potent antibiotic activity. J Biol Chem 2001; 276(3): 1772–9
Schittek B, Hipfel R, Sauer B, et al. Dermcidin: a novel human antibiotic peptide secreted by sweat glands. Nat Immunol 2001; 2(12): 1133–7
Hoffmann JA, Reichhart JM. Drosophila innate immunity: an evolutionary perspective. Nat Immunol 2002; 3(2): 121–6
Ladokhin AS, Selsted ME, White SH. Bilayer interactions of indolicidin, a small antimicrobial peptide rich in tryptophan, proline, and basic amino acids. Biophys J 1997; 72 (2 Pt 1): 794–805
Matsuzaki K, Sugishita K, Miyajima K. Interactions of an antimicrobial peptide, magainin 2, with lipopolysaccharide-containing liposomes as a model for outer membranes of gram-negative bacteria. FEBS Lett 1999; 449(2–3): 221–4
Epand RF, Epand RM, Monaco V, et al. The antimicrobial peptide trichogin and its interaction with phospholipid membranes. Eur J Biochem 1999; 266(3): 1021–8
Rozek A, Friedrich CL, Hancock RE. Structure of the bovine antimicrobial peptide indolicidin bound to dodecyl-phosphocholine and sodium dodecyl sulfate micelles. Biochemistry 2000; 39(51): 15765–74
Falla TJ, Karunaratne DN, Hancock RE. Mode of action of the antimicrobial peptide indolicidin. J Biol Chem 1996; 271(32): 19298–303
Sokolov Y, Mirzabekov T, Martin DW, et al. Membrane channel formation by antimicrobial protegrins. Biochim Biophys Acta 1999; 1420(1–2): 23–9
Boheim G. Statistical analysis of alamethicin channels in black lipid membranes. J Membr Biol 1974; 19(3): 277–303
Wu M, Maier E, Benz R, et al. Mechanism of interaction of different classes of cationic antimicrobial peptides with planar bilayers and with the cytoplasmic membrane of Escherichia coli. Biochemistry 1999; 38(22): 7235–42
Heller WT, Waring AJ, Lehrer RI, et al. Membrane thinning effect of the beta-sheet antimicrobial protegrin. Biochemistry 2000; 39(1): 139–45
He K, Ludtke SJ, Heller WT, et al. Mechanism of alamethicin insertion into lipid bilayers. Biophys J 1996; 71(5): 2669–79
Skerlavaj B, Romeo D, Gennaro R. Rapid membrane per-meabilization and inhibition of vital functions of gram-negative bacteria by bactenecins. Infect Immun 1990; 58(11): 3724–30
Cabiaux V, Agerberth B, Johansson J, et al. Secondary structure and membrane interaction of PR-39, a Pro+Arg-rich antibacterial peptide. Eur J Biochem 1994; 224(3): 1019–27
Moser C, Weiner DJ, Lysenko E, et al. β-Defensin 1 contributes to pulmonary innate immunity in mice. Infect Immun 2002; 70(6): 3068–72
Lemaitre B, Nicolas E, Michaut L, et al. The dorsoventral regulatory gene cassette spätzle/Toil/cactus controls the potent antifungal response in Drosophila adults. Cell 1996; 86: 973–83
Tzou P, Reichhart JM, Lemaitre B. Constitutive expression of a single antimicrobial peptide can restore wild-type resistance to infection in immunodeficient Drosophila mutants. Proc Natl Acad Sci U S A 2002; 99(4): 2152–7
Bals R, Weiner D, Moscioni A, et al. Augmentation of innate host defense by expression of a cathelicidin antimicrobial peptide. Infect Immun 1999; 67: 6084–9
Bals R, Weiner DJ, Meegalla RL, et al. Transfer of a cathelicidin peptide antibiotic gene restores bacterial killing in a cystic fibrosis xenograft model. J Clin Invest 1998; 103: 1113–7
David SA. Towards a rational development of anti-endotoxin agents: novel approaches to sequestration of bacterial endotoxins with small molecules. J Mol Recognit 2001; 14(6): 370–87
Kirikae T, Hirata M, Yamasu H, et al. Protective effects of a human 18-kilodalton cationic antimicrobial protein (CAP18)-derived peptide against murine endotoxemia. Infect Immun 1998; 66(5): 1861–8
Peschel A. How do bacteria resist human antimicrobial peptides? Trends Microbiol 2002; 10(4): 179–86
Shafer WM, Qu X, Waring AJ, et al. Modulation of Neisseria gonorrhoeae susceptibility to vertebrate antibacterial peptides due to a member of the resistance/nodulation/division efflux pump family. Proc Nat Acad Sci U S A 1998; 95(4): 1829–33
Lysenko ES, Gould J, Bals R, et al. Bacterial phosphorylcholine decreases susceptibility to the antimicrobial peptide LL-37/hCAP18 expressed in the upper respiratory tract. Infect Immun 2000; 68(3): 1664–71
Peschel A, Otto M, Jack R, et al. Inactivation of the dlt operon in Staphylococcus aureus confers sensitivity to defensins, protegrins, and other antimicrobial peptides. J Biol Chem 1999; 274: 8405–10
Ernst RK, Yi EC, Guo L, et al. Specific lipopolysaccharide found in cystic fibrosis airway Pseudomonas aeruginosa. Science 1999; 286(5444): 1561–5
Islam D, Bandholtz L, Nilsson J, et al. Downregulation of bactericidal peptides in enteric infections: a novel immune escape mechanism with bacterial DNA as a potential regulator. Nat Med 2001;7(2): 180–5
Soong L, Ganz T, Ellison A, et al. Purification and characterization of defensins from cystic fibrosis sputum. Inflamm Res 1997; 46: 98–102
Panyutich AV, Panyutich EA, Krapivin VA, et al. Plasma defensin concentrations are elevated in patients with septicemia or bacterial meningitis. J Lab Clin Med 1993; 122(2): 202–7
Van Wetering S, Mannesse-Lazeroms SP, Van Sterkenburg MA, et al. Effect of defensins on interleukin-8 synthesis in airway epithelial cells. Am J Physiol 1997; 272 (5 Pt 1): L888–96
Van Wetering S, Mannesse-Lazeroms SP, Dijkman JH, et al. Effect of neutrophil serine proteinases and defensins on lung epithelial cells: modulation of cytotoxicity and IL-8 production. J Leukoc Biol 1997; 62(2): 217–26
Panyutich AV, Hiemstra PS, van Wetering S, et al. Human neutrophil defensin and serpins form complexes and inactivate each other. Am J Respir Cell Mol Biol 1995; 12(3): 351–7
Chertov O, Michiel DF, Xu L, et al. Identification of defensin-1, defensin-2, and CAP37/azurocidin as T-cell chemoattractant proteins released from interleukin-8-stimulated neutrophils. J Biol Chem 1996; 271(6): 2935–40
Yang D, Chen Q, Chertov O, et al. Human neutrophil defensins selectively chemoattract naive T and immature dendritic cells. J Leukoc Biol 2000; 68(1): 9–14
Yang D, Chen Q, Schmidt AP, et al. LL-37, the neutrophil granule- and epithelial cell-derived cathelicidin, utilizes formyl peptide receptor-like 1 (FPRL1) as a receptor to chemoattract human peripheral blood neutrophils, monocytes, and T-cells. J Exp Med 2000; 192(7): 1069–74
Lillard Jr JW, Boyaka PN, Chertov O, et al. Mechanisms for induction of acquired host immunity by neutrophil peptide defensins. Proc Natl Acad Sci U S A 1999; 96(2): 651–6
Zhu QZ, Hu J, Mulay S, et al. Isolation and structure of corticostatin peptides from rabbit fetal and adult lung. Proc Natl Acad Sci U S A 1988; 85(2): 592–6
Yang D, Chertov O, Bykovskaia S, et al. Beta-defensins: linking innate and adaptive immunity through dendritic and T cell CCR6. Science 1999; 286: 525–8
Niyonsaba F, Someya A, Hirata M, et al. Evaluation of the effects of peptide antibiotics human beta-defensins-l/-2 and LL-37 on histamine release and prostaglandin D (2) production from mast cells. Eur J Immunol 2001; 31(4): 1066–75
Wang Y, Agerberth B, Lothgren A, et al. Apolipoprotein A-I binds and inhibits the human antibacterial/cytotoxic peptide LL-37. J Biol Chem 1998; 273(50): 33115–8
Gallo R, Ono M, Povsic T, et al. Syndecans, cell surface heparan sulfate proteoglycans, are induces by a proline-rich antimicrobial peptide from wounds. Proc Natl Acad Sci U S A 1994; 91: 11035–9
Chan YR, Gallo RL. PR-39, a syndecan-inducing antimicrobial peptide, binds and affects pl30 (Cas). J Biol Chem 1998; 273(44): 28978–85
Li J, Post M, Volk R, et al. PR39, a peptide regulator of angiogenesis. Nat Med 2000; 6(1): 49–55
Gao Y, Lecker S, Post MJ, et al. Inhibition of ubiquitin-proteasome pathway-mediated I kappa B alpha degradation by a naturally occurring antibacterial peptide. J Clin Invest 2000; 106(3): 439–48
Hoffmeyer MR, Scalia R, Ross CR, et al. PR-39, a potent neutrophil inhibitor, attenuates myocardial ischemia-reperfusion injury in mice. Am J Physiol Heart Circ Physiol 2000; 279(6): H2824–8
Ikeda Y, Young LH, Scalia R, et al. PR-39, a proline/argininerich antimicrobial peptide, exerts cardioprotective effects in myocardial ischemia-reperfusion. Cardiovasc Res 2001; 49(1): 69–77
Cole AM, Ganz T, Liese AM, et al. Cutting edge: IFN-inducible ELR-CXC chemokines display defensin-like antimicrobial activity. J Immunol 2001; 167(2): 623–7
Giangaspero A, Sandri L, Tossi A. Amphipathic alpha helical antimicrobial peptides. Eur J Biochem 2001; 268(21): 5589–600
Situ H, Balasubramanian SV, Bobek LA. Role of alpha-helical conformation of histatin-5 in candidacidal activity examined by proline variants. Biochim Biophys Acta 2000; 1475(3): 377–82
Wieprecht T, Dathe M, Schumann M, et al. Conformational and functional study of magainin 2 in model membrane environments using the new approach of systematic double-D-amino acid replacement. Biochemistry 1996; 35(33): 10844–53
Wieprecht T, Dathe M, Beyermann M, et al. Peptide hydrophobicity controls the activity and selectivity of magainin 2 amide in interaction with membranes. Biochemistry 1997; 36(20): 6124–32
Dathe M, Wieprecht T. Structural features of helical antimicrobial peptides: their potential to modulate activity on model membranes and biological cells. Biochim Biophys Acta 1999; 1462(1-2): 71–87
Wieprecht T, Dathe M, Epand RM, et al. Influence of the angle subtended by the positively charged helix face on the membrane activity of amphipathic, antibacterial peptides. Biochemistry 1997; 36(42): 12869–80
Dathe M, Wieprecht T, Nikolenko H, et al. Hydrophobicity, hydrophobic moment and angle subtended by charged residues modulate antibacterial and haemolytic activity of amphipathic helical peptides. FEBS Lett 1997; 403(2): 208–12
Oh JE, Hong SY, Lee KH. Structure-activity relationship study: short antimicrobial peptides. J Pept Res 1999; 53(1): 41–6
Oh JE, Lee KH. Synthesis of novel unnatural amino acid as a building block and its incorporation into an antimicrobial peptide. Bioorg Med Chem 1999; 7(12): 2985–90
Lee KH, Oh JE. Design and synthesis of novel antimicrobial pseudopeptides with selective membrane-perturbation activity. Bioorg Med Chem 2000; 8(4): 833–9
Blondelle SE, Takahashi E, Houghten RA, et al. Rapid identification of compounds with enhanced antimicrobial activity by using conformationally defined combinatorial libraries. Biochem J 1996; 313 (Pt 1): 141–7
Hong SY, Oh JE, Kwon M, et al. Identification and characterization of novel antimicrobial decapeptides generated by combinatorial chemistry. Antimicrob Agents Chemother 1998; 42(10): 2534–41
Lee KH. Development of short antimicrobial peptides derived from host defense peptides or by combinatorial libraries. Curr Pharm Des 2002; 8(9): 795–813
Yu Q, Lehrer RI, Tarn JP. Engineered salt-insensitive alpha-defensins with end-to-end circularized structures. J Biol Chem 2000; 275(6): 3943–9
Fernandez-Lopez S, Kim HS, Choi EC, et al. Antibacterial agents based on the cyclic D,L-alpha-peptide architecture. Nature 2001; 412(6845): 452–5
De Bolle MF, Osborn RW, Goderis U, et al. Antimicrobial peptides from Mirabilis jalapa and Amaranthus caudatus: expression, processing, localization and biological activity in transgenic tobacco. Plant Mol Biol 1996; 31(5): 993–1008
Okamoto M, Mitsuhara I, Ohshima M, et al. Enhanced expression of an antimicrobial peptide sarcotoxin IA by GUS fusion in transgenic tobacco plants. Plant Cell Physiol 1998; 39(1): 57–63
Cary JW, Rajasekaran K, Jaynes JM, et al. Transgenic expression of a gene encoding a synthetic antimicrobial peptide results in inhibition of fungal growth in vitro and in planta. Plant Science 2000; 154(2): 171–81
Piers KL, Brown MH, Hancock RE. Recombinant DNA procedures for producing small antimicrobial cationic peptides in bacteria. Gene 1993; 134(1): 7–13
Harrison SJ, McManus AM, Marcus JP, et al. Purification and characterization of a plant antimicrobial peptide expressed in Escherichia coli. Protein Expr Purif 1999; 15(2): 171–7
Yarus S, Rosen JM, Cole AM, et al. Production of active bovine tracheal antimicrobial peptide in milk of transgenic mice. Proc Natl Acad Sci U S A 1996; 93(24): 14118–21
O’Connell BC, Xu T, Walsh TJ, et al. Transfer of a gene encoding the anticandidal protein histatin 3 to salivary glands. Hum Gene Ther 1996; 7(18): 2255–61
Fehlbaum P, Rao M, Zasloff M, et al. An essential amino acid induces epithelial beta-defensin expression. Proc Natl Acad Sci U S A 2000; 97(23): 12723–8
Loury D, Embree JR, Steinberg DA, et al. Effect of local application of the antimicrobial peptide IB-367 on the incidence and severity of oral mucositis in hamsters. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1999; 87(5): 544–51
Gough M, Hancock RE, Kelly NM. Antiendotoxin activity of cationic peptide antimicrobial agents. Infect Immun 1996; 64(12): 4922–7
Ahmad I, Perkins WR, Lupan DM, et al. Liposomal entrapment of the neutrophil-derived peptide indolicidin endows it with in vivo antifungal activity. Biochim Biophys Acta 1995; 1237(2): 109–14
Biragyn A, Surenhu M, Yang D, et al. Mediators of innate immunity that target immature, but not mature, dendritic cells induce antitumor immunity when genetically fused with nonimmunogenic tumor antigens. J Immunol 2001; 167(11): 6644–53
Nibbering PH, Welling MM, van den Broek PJ, et al. Radiolabelled antimicrobial peptides for imaging of infections: a review. Nucl Med Commun 1998; 19(12): 1117–21
Welling MM, Paulusma-Annema A, Balter HS, et al. Technetium-99m labelled antimicrobial peptides discriminate between bacterial infections and sterile inflammations. Eur J Nucl Med 2000; 27(3): 292–301
Welling MM, Lupetti A, Balter HS, et al. 99mTc-labeled antimicrobial peptides for detection of bacterial and Candida al-bicans infections. J Nucl Med 2001; 42(5): 788–94
Jacob L, Zasloff M. Potential therapeutic applications of mag-ainins and other antimicrobial agents of animal origin. Ciba Found Symp 1994; 186: 197–216
Acknowledgements
Studies in the Dr. Bals’ laboratory related to innate immunity of the respiratory tract are supported by grants of the Deutsche Forschungsgemeinschaft (Ba 1641/1, Ba 1641/3-1) and the Mukoviszidose e.V. We thank Dr. D. J. Weiner (University of Pennsylvania, PA, USA) for helpful discussions. The authors declare no conflicts of interest relevant to the contents of this manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koczulla, A.R., Bals, R. Antimicrobial Peptides. Drugs 63, 389–406 (2003). https://doi.org/10.2165/00003495-200363040-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200363040-00005