Abstract
-
▴ Gemtuzumab ozogamicin is a humanised monoclonal IgG4 antibody, linked to a cytotoxic calicheamicin derivative. It effects cell necrosis by specifically targeting the CD33 antigen which is expressed on the surface of leukaemic cell blasts in more than 90% of patients with acute myeloid leukaemia (AML), but is not present on normal stem cells.
-
▴ Therapy with gemtuzumab ozogamicin (2 doses of 9 mg/m2) in 3 noncomparative studies produced complete remission in 16% of adult patients with AML in first relapse, and complete remission with incomplete platelet recovery in an additional 13% of patients. Rates of remission did not differ between those aged less than 60 years and older than 60 years.
-
▴ Many patients were able to receive both doses of gemtuzumab ozogamicin therapy as outpatients. Survival duration was similar between those treated as outpatients and those requiring hospitalisation.
-
▴ About one-third of 11 children and adolescents treated with 2 doses of 9 mg/m2 gemtuzumab ozogamicin in a phase I study showed >5% bone marrow blasts after completion of therapy.
-
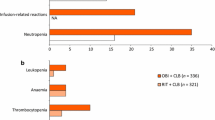
▴ The most commonly encountered adverse events in clinical trials with gemtuzumab ozogamicin were myelosuppression, increased levels of hepatic enzymes, infection, fever, bleeding, chills, nausea and vomiting and dyspnoea. No treatment-related renal failure or alopecia was reported.


Similar content being viewed by others
References
Liesner RJ, Goldstone AH. ABC of clinical haematology: the acute leukaemias. BMJ 1997; 314: 733–6
Lowenberg B, Downing JR, Burnett A. Acute myeloid leukemia. N Engl J Med 1999; 341: 1051–62
Appelbaum FR. Antibody-targeted therapy for myeloid leukemia. Semin Hematol 1999 Oct; 36 (4 Suppl. 6): 2–8
Hamann PR, Hinman LM, Hollander IJ, et al. Anti-CD33 calicheamicin hybrid conjugates for the treatment of acute myelogenous leukemia [abstract]. 87th Annual Meeting of the American Association for Cancer Research; 1996 Apr 20; Washington DC, 471
Hamann P. Gemtuzumab zogamicin, an anti-CD33 calicheamicin-antibody conjugate for treatment of AML [abstract]. 219th ACS National Meeting; 2000 Mar 26–30; San Francisco (CA)
Dinndorf PA, Andrews RG, Benjamin D, et al. Expression of normal myeloid-associated antigens by acute leukemia cells. Blood 1986; 67: 1048–53
Griffin JD, Linch D, Sabbath K, et al. A monoclonal antibody reactive with normal and leukemic human myeloid progenitor cells. Leuk Res 1984; 8: 521–34
Andrews RG, Singer JW, Bernstein ID. Precursors of colony-forming cells in humans can be distinguished from colonyforming cells by expression of the CD33 and CD34 antigens and light scatter properties. J Exp Med 1989; 169: 1721–31
Nicolaou KC, Pitsinos EN, Theodorakis EA, et al. Synthetic calicheamicin mimics with novel initiation mechanisms: DNA cleavage, cytotoxicity, and apoptosis. Chem Biol 1994; 1: 57–66
van der Velden VHJ, te Marvelde JG, Hoogeveen PG, et al. Targeting of the cd33-calicheamicin immunoconjugate Mylotarg (CMA-676) in acute myeloid leukemia: in vivo and in vitro saturation and internalization by leukemic and normal myeloid cells. Blood 2001; 97: 3197–204
Sievers EL, Appelbaum FR, Spielberger RT, et al. Selective ablation of acute myeloid leukemia using antibody-targeted chemotherapy: a phase I study of an anti-CD33 calicheamicin immunoconjugate. Blood 1999 Jun 1; 93: 3678–84
Bernstein ID. Monoclonal antibodies to the myeloid stem cells: therapeutic implications of CMA-676, a humanized anti-CD33 antibody calicheamicin conjugate. Leukemia 2000 Mar; 14: 474–5
Naito K, Takeshita A, Shigeno K, et al. Calicheamicin-conjugated humanized anti-CD33 monoclonal antibody (gemtuzumab zogamicin, CMA-676) shows cytocidal effect on CD33-positive leukemia cell lines, but is inactive on P-glycoprotein-expressing sublines. Leukemia 2000 Aug; 14: 1436–43
Dowell JA, Korth-Bradley J, Liu H, et al. Pharmacokinetics of gemtuzumab ozogamicin, an antibody-targeted chemotherapy agent for the treatment of patients with acute myeloid leukemia in first relapse. Clin Pharm In press
Korth-Bradley JM, Dowell JA, Berger MS, et al. Assessment of the possible influence of patient demographics on the pharmacokinetics of a new antibody-chemotherapeutic agent for relapsed acute myelogenous leukemia [abstract]. Pharmacotherapy 1999 Oct; 19: 1217
Dowell JA, King SP, Liu H, et al. Assessment of the pharmacokinetics of gemtuzumab ozogamicin in pediatric patients with relapsed acute myelogenous leukemia [abstract]. Pharmacotherapy 2000; 20: 1256
Sievers EL, Larson RA, Stadtmauer EA, et al. Efficacy and safety of gemtuzumab ozogamicin in patients with CD33-positive acute myeloid leukemia in first relapse. J Clin Oncol 2001 Jul 1; 19: 3244–54
Voliotis D, Mineur P, Dombret H, et al. The efficacy and safety of gemtuzumab zogamicin (CMA-676) in patients with acute myeloid leukemia in first relapse [abstract]. 25th Congress of the European Society for Medical Oncology; 2000 Oct 13–16; Hamburg
Sievers EL, Berger M, Mallick R, et al. Outpatient dosing of gemtuzumab ozogamicin (CMA-676), a novel antibody-targeted chemotherapy, for relapsed acute myeloid leukemia (AML): comparison with current treatments [abstract]. 28th World Congress of the International Society of Hematology; 2000 Aug 26–30; Toronto (ON)
Mallick R, Ellis R, Berger M. Potential resource savings from reduced risk of chemotherapy hospitalization associated with gemtuzumab ozogamicin (CMA-676), a new antibody-targeted chemotherapy in treatment of relapsed acute myeloid leukemia (AML) [abstract]. EORTC European Conference on Economics of Cancer; 2000 Sep 3–5; Brussels
Sievers EL, Arceci R, Franklin J, et al. Preliminary report of an ascending dose study of gemtuzumab ozogamicin (Mylotarg™, CMA-676) in pediatric patients with acute myeloid leukemia [abstract 4663]. Blood 2000; 96 (Pt 2): 217b
Wyeth Laboratories D of W-API. Mylotarg®. Philadelphia, PA 19101, May 2000
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McGavin, J.K., Spencer, C.M. Gemtuzumab ozogamicin. Drugs 61, 1317–1322 (2001). https://doi.org/10.2165/00003495-200161090-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200161090-00007